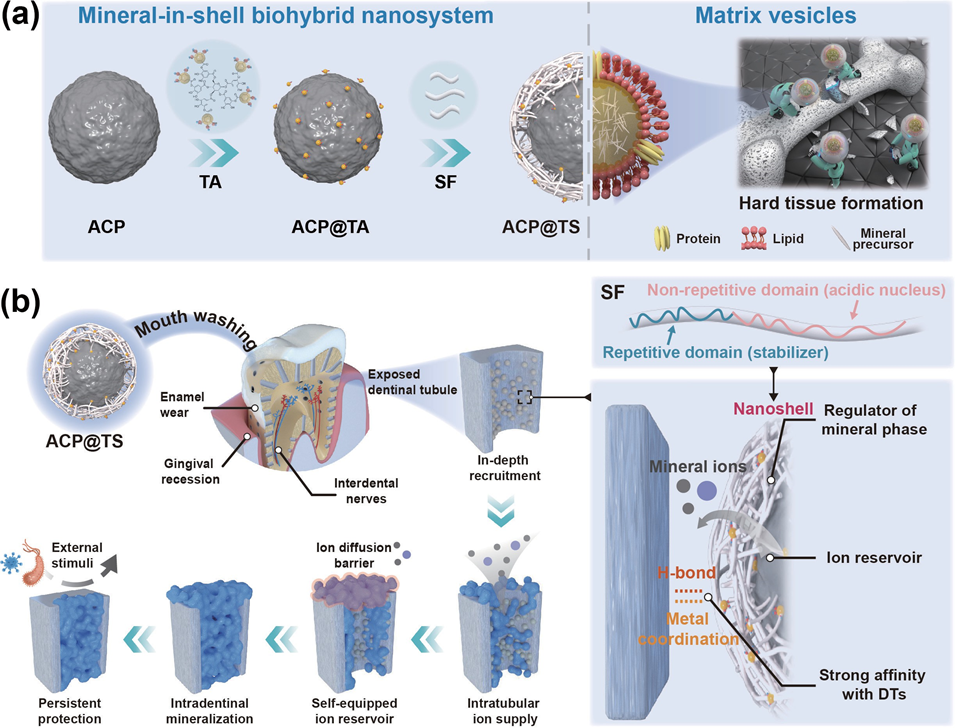
Bioinspired Mineral-in-Shell Nanoarchitectonics: Functional Empowerment of Mineral Precursors for Guiding Intradentinal MineralizationIssuing time:2025-04-01 15:53 Xiaoran Zheng, Yang Liu, Mingjing Li, Yuyan Li, Wanshan Gao, Rongmin Qiu, Jiaqi Xing, Jiaojiao Yang, Yantao Chen, Xinyuan Xu, Mingming Ding, Jun Luo (?), and Jianshu Lee (?) Abstract Effective mineralization of biological structures poses a significant challenge in hard tissue engineering as it necessitates overcoming geometric complexities and multistep biomineralization processes. In this regard, we propose “mineral-in-shell nanoarchitectonics”, inspired by the nanostructure of matrix vesicles, which can influence multiple mineralization pathways. Our nanostructural design empowers mineral precursors with tailorable properties through encapsulating amorphous calcium phosphate within a multifunctional tannic acid (TA) and silk fibroin (SF) nanoshell. The bioinspired nanosystem facilitates efficient recruitment of mineral precursors throughout the dentin structures, followed by large-scale intradentinal mineralization both in vitro and in vivo, which provides persistent protection against external stimuli. Theoretical simulations combined with experimental studies attribute the success of intradentinal mineralization to the TA-SF nanoshell, which exhibits a strong affinity for the dentin structure, stabilizing amorphous precursors and thereby facilitating concomitant mineral formation. Overall, this bioinspired mineral-in-shell nanoarchitectonics shows a promising prospect for hard tissue repair and serves as a blueprint for next-generation biomineralization-associated materials. Keywords mineral-in-shell, nanohybrids, self-assembly, biomineralization, dentinal tubule occlusion, dental hypersensitivity 1. Introduction Biomineralization is a fundamental process used by living organisms to create structural and functional minerals at different length scales, such as dentinogenesis, amelogenesis, and bone formation . Accordingly, numerous attempts with a major focus on regulating mineral crystallization have been made to duplicate the ingenious properties of biomineralization. However, biomineralization is a precise self-associating and self-organizing process in hierarchical biostructures, which includes ion accumulation, recruitment of mineral precursors to the site of mineralization, and morphogenesis of mineralized tissues, in addition to mineral crystallization. consequently, the aforementioned attempts to mimic biomineralization have only achieved effective mineralization in laboratory settings or have involved complex preparation and operational procedures, considerably hindering the development of clinically applicable methods for hard tissue engineering. Thus, it is necessary to develop a biocompatible system that spontaneously satisfies the multiple needs of mineralization in biological structures. Living organisms have developed sophisticated mechanisms that predictably and precisely regulate the process of biomineralization. Among them, matrix vesicles (MVs) are important nanovehicles for the fine-tuned morphogenesis of mineralized structures, such as skeletons and spicules. During these formation processes, the membrane of MVs functions as a confined space for concentrating mineral ions and prepacking mineral precursors, thereby avoiding the restriction from the solubility products of the mineral phase in biological fluids. Furthermore, MVs accompanied by intrinsically disordered proteins (IDPs) enable regulatory control of complex mineralization steps, such as the binding of metastable ionic cargos to mineralization fronts and the morphogenesis of mineralized tissues. Inspired by the unique mineral-in- shell nanoarchitectonics of MVs, grafting specific biomolecules onto mineral precursors to create a synthetic mineral-in-shell nanosystem, which represents a viable route for autonomously organizing multiple functions for efficient mineralization in biological structures. Based on these considerations, we propose bioinspired mineral- in-shell nanoarchitectonics and rationalize constitutive building blocks to accommodate multiple biomineralization pathways (Fig. 1(a)). Briefly, amorphous calcium phosphate (AcP) nanoparticles were encapsulated within a multifunctional shell comprising biocompatible silk fibroin (SF) via multiple interactions of tannic acid (TA) (termed AcP@TS). SF is composed of repetitive hydrophobic domains staggered by hydrophilic nonrepetitive acidic domains. These distinctive structural features of SF provide an opportunity to elegantly guide mineralization similar to IDPs, which can stabilize the amorphous precursors and also function as the critical nucleus for the growth of the polycrystalline phase. Moreover, as demonstrated by theoretical simulations, we found that the unique combination of TA and SF generated a strong affinity for mineralized structures via hydrogen bonding (H-bond). This affinity that stems from the TA-SF nanoshell confers AcP@TS a prolonged retention time in the dynamic physiological environment, thereby increasing the pool of prenucleation precursors available in the “hard-to-reach” regions. By merging these features from inorganic–organic modules and rationally investigating the mineralization of dentin structure, we found that AcP@TS exhibits broad bioactivity on a systemic level that influences multiple pathways involved in intradentinal mineralization, including ion accumulation, recruitment of precursor, and regulation of mineral formation (Fig. 1(b)). Experimental studies demonstrated that AcP@TS rapidly establishes mineralization frontiers on the interior of dentin (in 5 min) and exhibits remarkable intradentinal mineralization (over 50 μm), which is several times deeper than the majority of currently available treatments for dentin hypersensitivity (DH). The results allowed treated dentinal tubules (DTs) to remain compactly occluded in the presence of mechanical and acidic challenges, providing persistent protection against external stimuli. In summary, this facile supramolecular assembly of AcP@TS fulfills the needs of the multistep process involved in repairing dentin tissue and demonstrates that rationalizing the multifunctional-nanostructure design is essential for achieving effective mineralization within a biosystem.
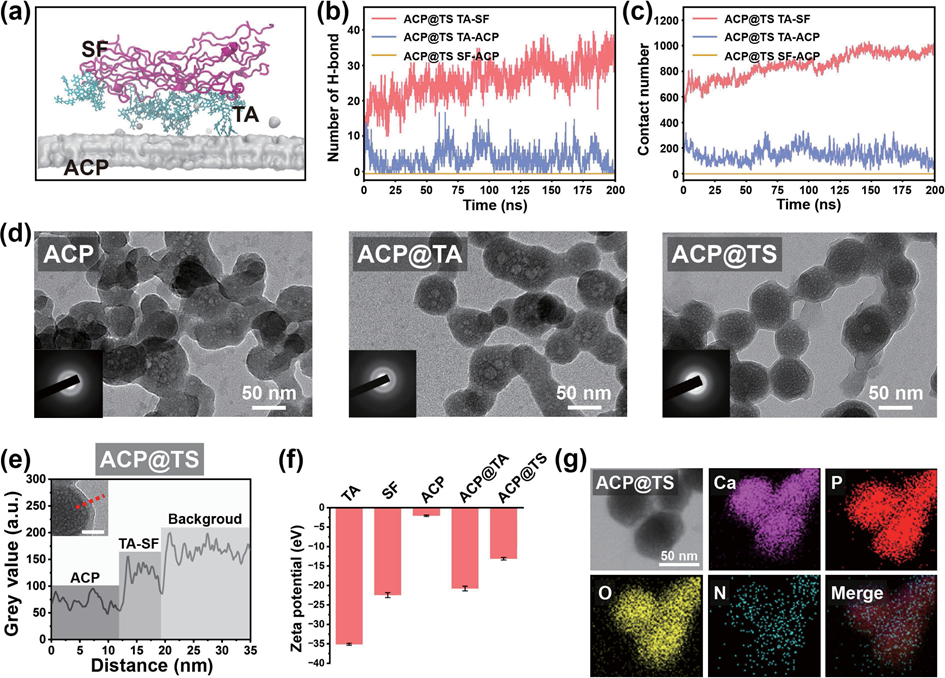
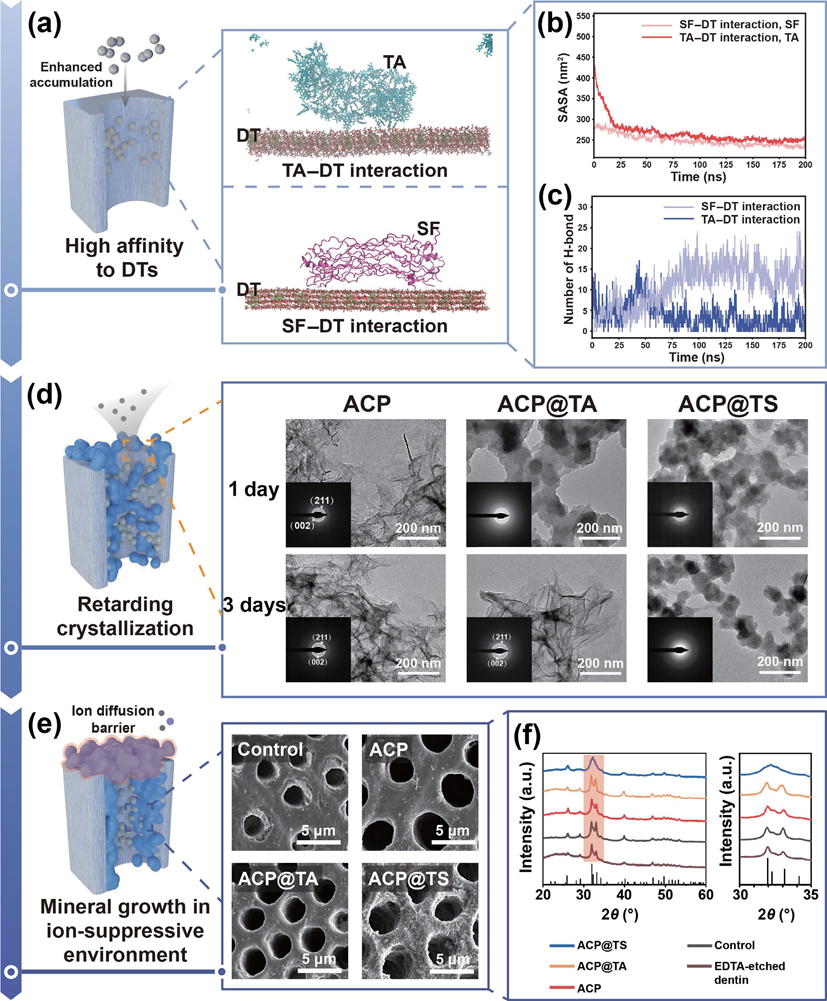
Figure 1 Schematic illustration of the inspiration of mineral-in-shell biohybrid nanosystem and its application for intradentinal mineralization and DH. (a) Functional empowerment of amorphous calcium phosphate by polyphenol-mediated protein assembly (designated as AcP@TS). (b) DH, one of the most prevalent chronic diseases in humans worldwide, is dependent on the severity of dentin exposure. In this context, enamel wear and gingival recession inevitably increased the exposure risk and concomitantly exposed the interdental nerves to external stimuli, causing transient and sharp pain. Herein, we proposed the modulation of the intradentinal mineralization process using multifunctional AcP@TS. Even if superficial layers were removed by bruxism or acidic diet habits, the compact and deep minerals provide persistent protection from external stimuli. 2. Results and discussion 2.1 Design and characterization of ACP@TS Although the mineral surface patchiness (e.g., morphology, roughness, and surface charge) is unfavorable for homogeneously and noncovalently assembling biomolecules at the nanoscale, polyphenol-mediated assembly via multiple supramolecular interactions offers new perspectives for mineral surface engineering. Firstly, the successful preparation of AcP was verified by X-ray diffraction (XRD). As shown in Fig. S1 in the Electronic Supplementary Material (ESM), no obvious diffraction peaks are observed, indicating that the product consists of amorphous calcium phosphate. In this sequential assembly process (Table S1 in the ESM), AcP nanoparticles were modified by TA (designated as AcP@TA) to bridge the gap between the SF protein and AcP mineral cores. Subsequently, the SF was coated to obtain a transparent protein shell on the surface of the mineral core (designated as AcP@TS), which acted as a stable emulsion (Fig. S2 in the ESM). We performed molecular dynamics (MD) simulations to analyze the supramolecular assembly of the AcP@TS system (Fig. 2(a)). We found that TA molecules readily form H-bonds with SF molecules to form TA-SF nanocomposites and also serve as H- bond bridges between AcP and SF (Fig. 2(b)). Furthermore, TA- SF nanocomposites within the AcP@TS nanosystem maintain close and stable contact with one another throughout the duration of the simulation. In stark contrast, during the 200-ns simulation, there was no contact between SF and AcP (Fig. 2(c)). These results suggest that the noncovalent H-bonds of TA-SF and TA- AcP comprise the energy source that initiates and stabilizes the self-assembly of the mineral-in-shell biohybrid nanosystem. Additionally, the calcium atoms in AcP may chelate to the phenolic ligands of TA, providing an additional energy source for the polyphenol-mediated assembly. However, due to the limitations of MD, charge redistribution among intramolecular atoms cannot be accomplished during the simulation. Thus, metal- phenol coordination was not considered, which may underestimate the force of attraction between AcP and TA molecules. Obtained AcP@TS exhibited diameters of ~ 70 nm and distinct amorphous phase diffraction patterns (Fig. 2(d) and the insets in Fig. 2(d)), demonstrating that surface engineering had a negligible influence on the crystallographic properties of AcP cores. Additionally, the size of AcP@TS was further verified through atomic force microscopy (AFM) (Fig. S3 in the ESM). Accordingly, TA was found to capture SF tightly in AcP@TS, leading to the formation of a well-defined nanoshell on the AcP surface (Fig. 2(e) and Fig. S4 in the ESM). Moreover, the zeta potentials of the sequential assemblies of AcP, TA, and SF (Fig. 2(f)), were found to shift correspondingly with the change in the outermost components. In addition, energy dispersive spectroscopy (EDS) mapping served as an intuitive proof of AcP@TS assemblies (Fig. 2(g)). The result demonstrated that the N distribution pattern matched well with the morphology of AcP@TS and the distribution patterns of ca, P, and O, thereby validating the continuous coating of SF on AcP. The TA-mediated assembly was further analyzed through Fourier transform infrared (FTIR) spectroscopy, X-ray photoelectron spectroscopy (XPS) and thermogravimetric analysis (TGA). Typical peaks at 1447 and 1362 cm–1, corresponding to the vibration of phenolic hydroxyl groups of TA and the amide ? peak at 1540 cm–1 in the FTIR spectra (Fig. S5 in the ESM) demonstrated the presence of TA and SF on AcP@TS. Similar results were confirmed via XPS (Fig. S6(a) in the ESM). Notably, the ca 2p3/2 and ca 2p1/2 peaks of AcP@TA and AcP@TS shifted to higher bind energies at 347.9 and 351.5 eV, respectively, as evidenced by the high-resolution XPS spectra (Fig. S6(b) in the ESM), which were caused by the coordination-induced electron transformation between TA and AcP. In addition, the new N 1s peak at 402 eV belongs to protonated NH3+ (Fig. S6(b) in the ESM), demonstrating that H-bonds exist between TA and SF in AcP@TS. As shown in Fig. S7 in the ESM, the TGA curve of AcP@TS can be divided into three stages. During the initial stage (30 to 155 oc), weight loss can be attributed to the vaporization of surface-bound and structural water, consistent with AcP-based materials. In the second stage (155 to 280 oc), a small weight loss occurred, possibly due to the decomposition of TA molecules. A similar trend was also observed in the TGA curve of AcP@TA. Finally, a significant combustion reaction of SF molecules (~ 10 wt.% of total weight) occurred from 280 to 440 oc. collectively, the branched TA molecule enables multiple binding sites in terms of ester groups and phenolic hydroxyls to maximally weave SF into a nanoshell on AcP cores, indicating that our protocol can be used to materialize the biohybrid mineral- in-shell nanostructure.
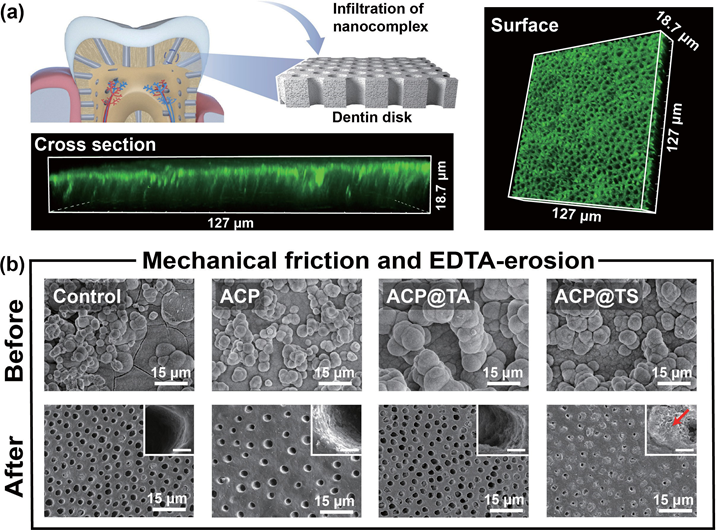
Figure 2 characterization of the bioinspired mineral-in-shell nanosystem. (a) Snapshots of AcP@TS after they reach equilibrium. (b) H-bond numbers and (c) contact numbers in the AcP@TS system. (d) TEM images of AcP, AcP@TA, and AcP@TS (insets: SAED patterns). (e) Line profile of the greyscale intensity for AcP@TS. (f) Zeta-potential values in the sequential assemblies. (g) TEM images and the corresponding EDS mappings show the elemental content of AcP@TS. 2.2 Constructionofmineralizationfrontiersand concomitant mineralization Highly organized mineralized tissues, including bone, dentin, and enamel, exhibit broad structural/functional activity. Among these structures, dentin is a rigid and durable mineral-rich composite and is generally characterized by oriented DTs (Fig. S8 in the ESM). Accordingly, dentin serves as the resilient support for the brittle enamel and a protective layer for the soft pulpal tissue and nerve endings. consequently, increasing levels of enamel wear or gingival recession inevitably increase the permeability of DTs, leading to the transmission of external stimuli to intradentinal nerves and ultimately causing dental hypersensitivity, which is one of the most prevalent chronic diseases. In this regard, intradentinal mineralization is essential for the maximum reduction of exposure risk and the alleviation of DH. To resolve these problems, traditional mineralization-associated materials, such as dendrimers, mineral nanoparticles, and protein/peptide-derived compounds, require a longer treatment time (1–12 h), which undoubtedly increases the burden on patients. Given the interwoven structure of TA-SF, it is anticipated that its strong interfacial adhesion capability will enable rapid adhesion between materials and substrates without the need for redundant pretreatments. The confocal laser scanning microscopic (cLSM) images (Fig. 3(a)) demonstrated that AcP@TS accumulates easily on the dentin surface and penetrates DTs below the surface (~ 15 μm) in only 5 min, exhibiting that AcP@TS could reduce treatment time and operational complexity during clinical treatment. To further elucidate the occlusion performance of the bioinspired nanosystem, we incubated dentin slices in simulated body fluid (SBF) following various treatments. The oversaturated ion environment facilitates the transformation of mineral precursors into hydroxyapatite (HAp), but also initiates a comprehensive crystallization process, including nucleation through monomer-by-monomer addition, precipitation, and ripening [6]. These factors caused the complete coverage of all dentin surfaces with comparable bulk minerals after 7 days of incubation (Fig. 3(b), upper panel, and Fig. S9 in the ESM). However, the superficial mineral layer was vulnerable to dietary acids and physical abrasion caused by tooth brushing, causing the relapse risk to inevitably increase once treatment has been discontinued. Herein, we conducted intensive brushing and ethylenediaminetetraacetic acid (EDTA)-erosion on remineralized dentin. Although superficial mineral layers of all groups were almost entirely eroded (Fig. 3(b), bottom panel), we found that minerals were still packed at the orifices of the AcP@TS-treated dentin and very few newly formed minerals can be found at the orifices of control group (i.e., without material treatment, AcP, and AcP@TA), similar to that in only TA- and SF-treated dentin disks (data not shown). In addition, the ca/P ratio of the remaining minerals was 1.57 (Fig. S10 in the ESM), which is close to the theoretical value of HAp. Notably, two commercial desensitizers, namely, Gluma and Tianfu, were utilized to compare the effectiveness of treatment. Although dentin was treated with these two commercial desensitizers, the sealing layers formed on the dentin surface were eroded in a similar manner (Fig. S11 in the ESM). Overall, this treatment required only 5 min to promote the formation of a deep mineralization frontier in DTs and significantly increase seal stability.
Figure 3 Formation of mineralization frontiers and matured minerals on dentin surface and within DTs. (a) AcP@TS rapidly spread and constructed the hybrid mineralization frontiers on dentin. (b) SEM images of remineralized dentin before and after tooth brushing and EDTA-etching (upper panel and bottom panel, respectively), revealing the resistance effect of different materials after brushing and acid attack. Insets: scale bar = 1 μm.
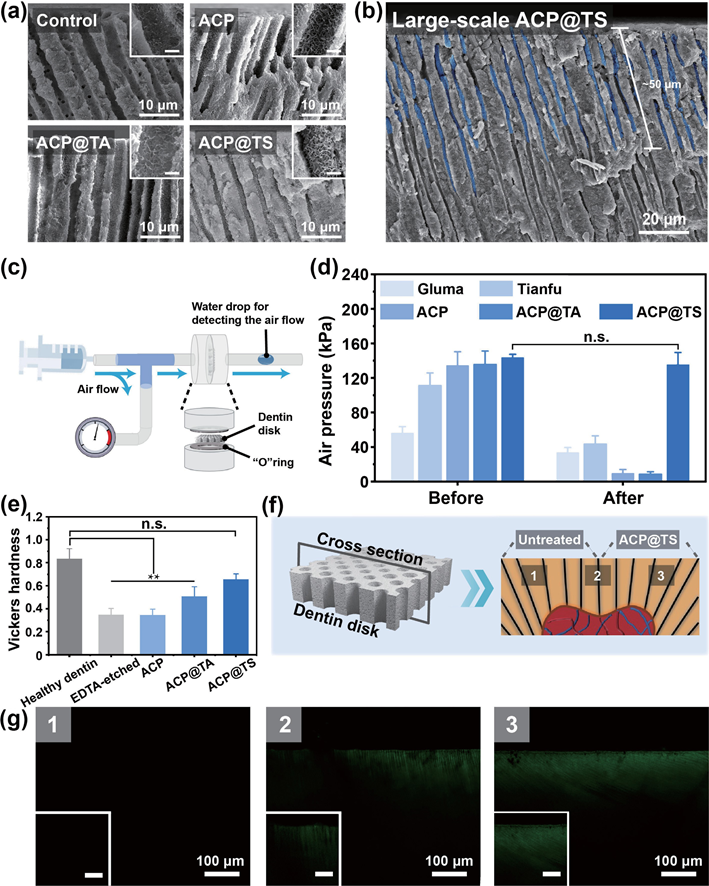
To thoroughly evaluate the mineralization capability, we expanded our studies to include the regeneration depth of minerals in DTs as well as the airtightness of remineralized dentin after AcP@TS rapidly established the mineralization frontier in the hierarchical structure of dentin. As shown in Fig. 4(a), the cross-sectional scanning electron microscopy (SEM) images of AcP@TS-treated dentin revealed that regenerated minerals (ca/P ratio = 1.74, Fig. S12 in the ESM) occluded the entire DTs, whereas no obvious mineral formation was observed in the other groups. To date, the commonly used materials are solely based on crystallization reactions, which renders them to be insufficient for meeting the complex requirements of intradentinal mineralization. In addition, we observed a relatively wide and uniform distribution of regenerated minerals throughout the DTs (> 50 μm) in the large- scale SEM image (Fig. 4(b)), which explains the excellent resistance to mechanical destabilization and acid erosion. Moreover, an airtightness test was conducted to determine the occlusion capability (Fig. 4(c)). As illustrated in Fig. 4(d), all dentin slices showed high airtightness prior to their exposure to abrasive and acidic conditions. However, the airtightness behavior of all groups changed significantly after the surface seal layer was removed by friction and erosion, with the exception of AcP@TS treated dentin, whose airtightness did not decrease significantly (i.e., from 143.5 ± 3.8 to 135.3 ± 14.1 kPa). Mechanical properties, which play a crucial role in evaluating dentin repair, were assessed using Vickers hardness tests (Fig. 4(e)). Healthy dentin showed optimal mechanical performance (0.83 ± 0.09 GPa). However, the mechanical properties of the dentin were weakened by EDTA etching (17 wt.%, 5 min). Notably, AcP@TS-treated dentin demonstrated a remarkable recovery in mechanical strength, with values of 0.66 ± 0.05 Gpa. Taking all of these results into consideration, the notable superiority of AcP@TS occludes the DTs along with its capacity to restore the chemical composition and mechanical properties of dentin, indicating AcP@TS has great potential for the management of DH. Furthermore, a visualized mineralization experiment was performed by monitoring the mineralization of whole dentin slices after different treatments and incubation in SBF (Fig. 4(f)). Accordingly, cLSM images were selected along the longitudinal section of the treated dentin. After 7 days of incubation, fluorescence was observed in the deep region of the AcP@TS-treated sample, confirming intradentinal mineral growth (Fig. 4(g)). In contrast, no fluorescence was observed in the untreated sample, which was further validated by the multiple length-scale SEM images (Fig. S13 in the ESM). In accordance with the previously mentioned findings, newly formed minerals were distributed in the deep region (~ 50 μm) of the AcP@TS- treated sample. correspondingly, we found that numerous spherical nanoparticles aggregated along the deeper wall of DTs, indicating that AcP@TS could accumulate in the dentin structures and facilitate intradentinal mineralization.
Figure 4 comprehensive evaluations of the mineralization capability. (a) cross-sectional SEM images of dentin after different treatments and 7 days of incubation. Insets: scale bar = 1 μm. (b) Large-scale SEM image of AcP@TS-treated dentin; dense minerals occluded the DTs (highlighted in blue). (c) Diagram of the airtightness test apparatus and (d) airtightness test results of different dentin samples before and after friction and erosion. (e) Vickers hardness of dentin after 7 days of incubation. (**p < 0.01). (f) Diagrams of a distinct visualized comparison between the untreated group and AcP@TS-treated group. (g) cross-sectional cLSM images were collected from the preestablished detection positions, i.e., 1, 2, and 3 in (f). Insets: scale bar = 25 μm. 2.4 Possible processes for intradentinal mineralization To understand the mechanism of intradentinal mineralization, the interactions between the DTs and the nanosystems were investigated via MD simulations using the GROMAcS 2021 software. Due to the high cost of simulation, the cores of the nanosystems were overlooked and only the outermost layers of the nanosystems (SF molecules in the AcP@SF system and TA molecules in the AcP@TA system) were considered. As shown in Fig. 5(a), both TA and SF molecules can bind to the surface of DTs. For a fair comparison of the interactions between TA molecules and DTs (TA-DT) and SF molecules and DTs (SF-DT), 40 TA molecules with comparable solvent-accessible surface area to the SF molecule were included in the system (Fig. 5(b)). Even though the solvent-accessible surface area of TA molecules was marginally higher than that of SF molecules, the contact number (Fig. S14(b) in the ESM) and hydrogen bond number (Fig. 5(c)) of TA-DT were lower than those of SF-DT. These results indicate that SF–DT interaction is stronger than TA–DT interaction. This is because the 40 TA molecules prefer self-interaction and assembly into stable aggregates, as shown in Fig. S14(c) in the ESM. conversely, the SF molecule could adjust its shape, thereby increasing the contact area and H-bonds with the deep walls of DTs, explaining the efficient recruitment of AcP@TS in dentin structures. Although several SF-TA-based macroscopic materials have shown universal adhesive behavior at the macro level, we are excited to observe our SF-TA nanoshell can generate sufficient H-bonds at the nano/micro level to immobilize mineral precursors onto the biological structures and facilitate the formation of mineralization fronts. Prevention of the rapid transformation from AcP to matured minerals and reduction of the relatively dense mineral layer that occludes orifices of DTs could ensure a continuous supply of mineral ions from the oral environment during the infant stage of biomineralization. Herein, we acquired the nanoscale transition process of our samples using TEM and selected area electron diffraction (SAED) (Fig. 5(d)). We immersed AcP, AcP@TA, and AcP@TS into aqueous solution and observed their morphological changes at pre-designed time intervals. Normally, the AcP phase exists in an aqueous solution for only a short period, consistent with our findings where both AcP and AcP@TA underwent rapid transformation into sheet-like crystals after 1 and 3 days, respectively. However, AcP@TS exhibited remarkable stability, retaining its amorphous and nanospherical structure even after 7 days, before undergoing a complete transformation into ribbon-like minerals in 14 days (Fig. S15 in the ESM). Two concentric rings appeared in the 1-day AcP, 3-day AcP@TA, and 14-day AcP@TS groups, corresponding to the crystallographic planes (211) and (002) of HAp (insets in Fig. 5(d)), indicating the transformation from AcP to HAp. Taken together, these morphology and crystallographic characterizations indicate that the TA-SF hybrid nanoshell in AcP@TS effectively retards the crystallization from AcP. One possible explanation for intradentinal mineralization is that TA molecules promote the accumulation of SF at the phase boundary of AcP, and subsequently, the formation of hybrid nanoshell confines the mineral phase, which inhibits the crystallization of the inner mineral core. To further categorize the pathway of intradentinal mineralization, an in vitro proof-of-concept study was designed to analyze the evolution of minerals in an ion-suppressive environment. In stark contrast to the other groups, only the AcP@TS-treated group (Fig. 5(e)) exhibited the presence of numerous needle-like minerals across the dentin surface and the tubule walls beneath the dentin surface. The confluence of findings from the broad amorphous hump (Fig. 5(f)) and the element composition in Fig. S16 in the ESM indicates the presence of amorphous calcium phosphate in the minerals associated with AcP@TS-treated dentin. The AcP@TS-induced mineral growth in a calcium ion-suppressive environment suggests that the encapsulated mineral precursors in AcP@TS can also self-supply sustained mineral ions (Fig. S17 in the ESM). Therefore, in the oral environment, AcP@TS is an ion reservoir that circumvents the aforementioned passive-diffusion barriers and self-supplies the required ions to synergistically promote intradentinal mineralization. We hypothesize the multistep mineralization processes of dentin structures based on the experimental studies and theoretical calculations described previously (Fig. 5). In this scenario, after AcP@TS diffuses into DTs, TA-SF nanoshell provides high-density noncovalent interactions (e.g., H-bond) with DTs and enhances the preferential recruitment of AcP@TS onto their wall. Although the high affinity of AcP@TS is not specific to DTs, this interaction may be advantageous for slowing the loss of AcP@TS from the interior of DTs and extending their retention time, thereby increasing the pool of prenucleation precursors in the deep region of DTs. Meanwhile, the nanoshell-retarded crystallization and the modest electrostatic repulsion between AcP@TS and DTs may prevent the formation of large aggregates from blocking the orifices of DTs when the mineralization process is in its infancy. consequently, the mineral- in-shell biohybrid nanosystem minimizes the influence of diffusion barriers for saliva into DTs to the greatest extent, ensuring that ionic calcium and phosphate are sufficiently saturated for mineral formation at a preliminary stage. Moreover, the ion-suppressing mechanism caused by the superficial mineral layer could be nullified by AcP@TS’s mineral precursor depot. Accordingly, the nonclassical nucleation theory is proposed for deep intradentinal mineralization. Briefly, encapsulated AcP cores either internally aggregate and rearrange to form matured minerals or accelerate heterogeneous recrystallization with the help of self-supplied mineral ions and an abundance of negatively charged groups in the TA-SF nanoshell. Therefore, the deep and compact occlusion in DTs is most likely attributable to the synergistic physicochemical influences that result from the mineral- in-functional-shell nanoarchitectonics, which program a series of mineralization steps in dentin structures.
Figure 5 Systematic characterizations for illustrating possible mineralization processes guided by AcP@TS. (a) Snapshots of the two systems after they reach equilibrium. The oxygen, phosphate, and calcium atoms in DT are colored red, purple, and green, respectively. Water and counter ions are not shown for clarity. (b) and (c) Solvent-accessible surface area and H-bond number as functions of time. The two columns in the legend represent the systems corresponding to the two panels A and the target component molecules in the corresponding systems. (d) TEM images and the SAED patterns of different groups stored in an aqueous solution. (e) SEM images of mineral growth in the ion-suppressive environment. (f) XRD spectra of the dentin disk after 7-day incubation in PBS (without calcium), following different treatments. The stick-figure spectrum for HAp is shown at the bottom. (powder diffraction file, hydroxyapatite, #86-0740).
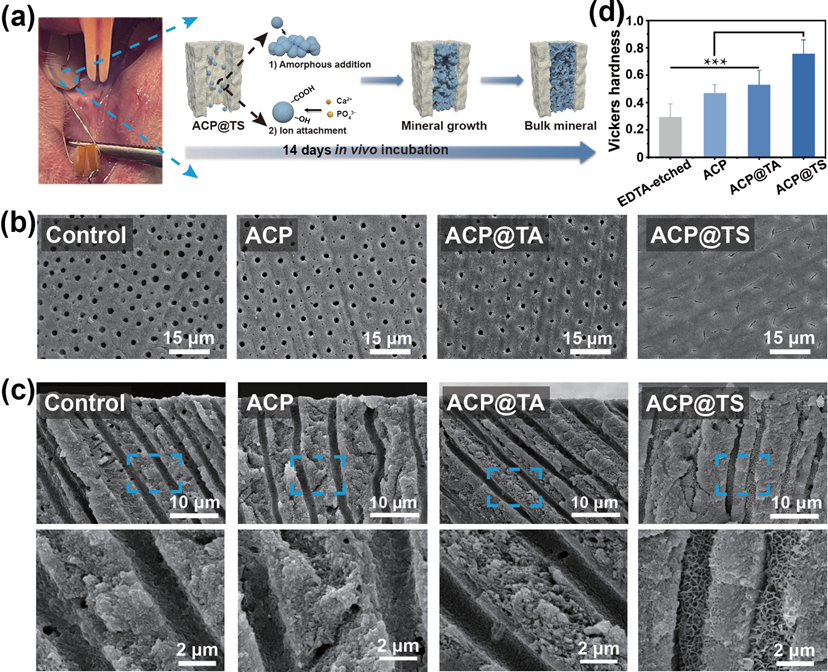
Based on the above results, we also investigated the biocompatibility of AcP@TS in the treatment of DH. First, we examined the in vitro cytotoxicity of dental pulp cells (DPcs) (Figs. S18 and S19 in the ESM). As expected, all groups exhibited good cell viability. consistently, the cell morphology confirmed the cytocompatibility of AcP@TS. Further in vivo experiments were conducted to evaluate the performance of the bioinspired nanosystem (Fig. 6(a)). compared to other groups, only a continuous bulk mineral layer was observed on the surface of AcP@TS-treated dentin (Fig. 6(b)). Moreover, the cross-sectional SEM images confirmed the in vitro findings, which demonstrate that newly formed calcium phosphate minerals were packed in AcP@TS-treated DTs (Fig. 6(c) and Fig. S20 in the ESM). In contrast, we did not observe any mineral formation in the other groups, which included untreated dentin, AcP-treated dentin, and AcP@TA-treated dentin. Additionally, mechanical properties behavior is essential in dentin repair evaluation. As shown in Fig. 6(d), after 14 days of incubation in the oral cavity of rats, the Vickers hardness of the dentin surfaces treated with AcP@TS has recovered to the level of healthy dentin, with values of 0.75 ± 0.10 GPa. Overall, the exceptional in vivo mineralization capacity of AcP@TS reflected a promising clinical potential for hard tissue repair.
Figure 6 AcP@TS induced in vivo intradentinal mineralization. (a) Photograph of the dentin disk fixed in the oral cavity (left) and the proposed multiple biomineralization process occurring within DTs (right). The protein shell of AcP@TS confines the amorphous mineral phase, allowing crystallization by the addition of nanoparticles, additionally, the organic molecules in the protein shell provide additional nucleation sites for mineral growth [6]. (b) SEM images of dentin surfaces and (c) cross-sectional SEM images of dentins after 14 days of in vivo incubation. (d) Vickers hardness of dentin after 14 days of incubation in the rat oral cavity. (***p < 0.001) 3. Conclusions The functions of MVs in the natural biomineralization process inspired us to design the mineral-in-shell biohybrid nanoarchitectonics. By rationalizing multifunctional-structural self- assembly, AcP@TS fulfills the needs in the multi-factors of mineralization. It functions as a mineral-precursor regulator and serves as an ion reservoir, while demonstrating a high affinity to mineralization fronts. Our findings demonstrate that AcP@TS rapidly constructs mineralization frontiers in DTs within 5 min, facilitates intradentinal mineralization, and increases the occlusion stability of DTs. The remarkable biocompatibility and expeditious self-assembly processes inherent in our nanohybrids present a promising way for fabricating highly exquisite nanosystems, whose properties can be finely regulated by functional constituents such as DNA, enzymes, and genetically engineered proteins. We envision that materials constructed using “mineral-in-shell nanoarchitectonics” could be further explored to precisely mimic the natural biomineralization process for various hard tissue applications, including intrafibrillar and extrafibrillar mineralization of collagen scaffolds, mineralized tissue morphogenesis such as enamel epitaxial growth and bone healing. 4. Experimental sections 4.1 Materials TA was purchased from Sigma-Aldrich (USA). calcein and lithium bromide (LiBr) were purchased from Aladdin (china). Polyethylene glycol (PEG, MW = 20,000) was purchased from Macklin (china). Hydrogen chloride (Hcl), EDTA, and all salts were purchased from chron chemicals (china). The calcium assay kit and phosphate assay kit were purchased from Nanjing Jiancheng Bioengineering Institute (china). Phosphate buffered saline (PBS without calcium and magnesium, pH = 7.4), fetal bovine serum (FBS), penicillin-streptomycin (P/S), and a- minimum essential media (a-MEM) were purchased from Gibco (USA). cell counting kit-8 (ccK-8) was purchased from MedchemExpress (USA). Fluorescein diacetate/propidium iodide (FDA/PI), FITc-Phalloidin, and 4‘,6-diamidino-2-phenylindole dihydrochloride (DAPI) were purchased from Solarbio (china). 4.2 Extraction of SF solution The purified SF solution was obtained based on a previously reported method [32]. Briefly, Bombyx mori cocoons were cut into pieces and then boiled in 0.02 M Na2cO3 for 1 h to remove sericin. This step was repeated three times. The degummed fibers were washed with Milli-Q water three times and dried overnight. Subsequently, the dry fibers were dissolved in a 9.3 M LiBr solution for 4 h at 60 oc. The resulting solution was dialyzed against Milli-Q water for 3 days and then concentrated against a 10-wt.% PEG (MW = 20,000) aqueous solution to yield a 10-wt.% SF aqueous solution. Finally, the concentration of SF aqueous solution was determined by weighing the residual solids after drying the solution. 4.3 Preparation of mineral-in-shell nanosystem A 0.004 M solution of Na2HPO4 (25 mL) was rapidly mixed with a 0.006 M solution of cacl2 (25 mL) for 2 min to yield an AcP suspension. Subsequently, AcP@TA suspension was prepared by adding 100-μL TA solution (8 mg/mL) to 50 mL of AcP suspension, followed by stirring for 5 min. Subsequently, 20-μL SF solution (10 mg/mL) was added to the AcP@TA suspension; this step was repeated at different times (Table S1 in th ESM) (each time stirred for 5 min). Particularly, the AcP@TS suspension comprised 100-μL TA and 100-μL SF. All preceding procedures were conducted in an ice incubator at 4 oc. Additionally, all suspensions were photographed to analyze their stability (Fig. S2(a) in the ESM). Specifically, for fluorescence imaging, 50-μL calcein (1 mg/mL) was added to 50-mL-extracted AcP suspension and stirred for 30 min. The calcein-labelled suspension was then dialyzed against Milli-Q water for 4 h to remove unbound compounds before TA-SF nanoshell formation. The same protocol was used to prepare calcein-AcP@TS. 4.4 Characterization of mineral-in-shell nanosystem The morphology of the nanosystem was determined via TEM (JEOL, JEM-2100F, Japan) and AFM (AIST-NT, SMART SPM, USA). Additionally, their amorphous characteristics were demonstrated through SAED. To confirm the sequential assembly of TA and SF on AcP surfaces, the changes in their surface zeta potentials were observed using a Zetasizer Nano zeta-potential analyzer (Malvern, Z90, UK). Moreover, FTIR (Nicolet 6700, Thermo Scientific, USA) and XPS (K-α, Thermo Scientific, USA) were employed to analyse the chemical composition of nanosystems. The binding energies were calibrated using the c 1s peak (c–c and c–H) of hydrocarbons at 284.6 eV. The elemental compositions of the nanosystem were detected via EDS (Aztec X- Max80, Oxford, UK). The transformation process of AcP into HAp was monitored by TEM. In brief, 25 mL of the aqueous suspensions of different samples were stored at 4 oc, respectively. At pre-designed time points (i.e., 1, 3, 5, 7, and 14 days), TEM specimens were prepared by applying a 5 μL drop of the dispersion onto a carbon-coated copper grid and drying. The thermal stability of various nanosystems was assessed using a thermogravimetric analyzer (NETZScH STA 449 F3, Germany). Approximately 3 mg of each sample was used for each measurement, and all measurements were conducted under a nitrogen atmosphere with a gas flow, heating up to 800 oc at a rate of 20 oc/min. 4.5 Preparation of human dentin disk Human teeth were obtained from the West china Hospital of Stomatology, Sichuan University. After carefully cleaning the tooth, dentin disks were prepared using a saw by separating the root from the crown, as reported previously. To simulate DH, dentin disks were immersed in 17-wt.% EDTA solution (pH = 7.4) for 5 min to ensure that all DTs were exposed. EDTA-etched dentin disks were ultrasonically treated in Milli-Q water for 10 min and then dried with nitrogen gas. The study was approved by the Medical Ethics committees of West china Hospital of Stomatology (WcHSIRB-D-2021-078). 4.6 Formation of mineralization frontier EDTA-etched dentin disks were immersed in 10-mL AcP, AcP@TA, or AcP@TS suspensions for 5 min to form a mineralization front. The AcP, AcP@TA, and AcP@TS-treated dentin disks were washed three times with Milli-Q water to remove unbound components. For cLSM (A1R+, Nikon, Japan) experiments, calcein-AcP@TS-treated dentin was observed through cLSM. The fluorescent images were obtained via excitation using a 488 nm laser. The emission wavelength was 515 nm. 4.7 In vitro occlusion of mineral-in-shell nanosystem According to the established method, 1.5 x SBF (pH = 7.4) was prepared using the following ionic salts: Nacl (137.5 mM), NaHcO3 (4.2 mM), Kcl (3 mM), K2HPO4?3H2O (1.5 mM), Mgcl2?6H2O (1.5 mM), cacl2 (3.75 mM), and Na2SO4 (0.5 mM). The AcP, AcP@TA, and AcP@TS-treated dentin disks were then incubated in 10-mL 1.5 x SBF for 7 days. Throughout the process, SBF was replaced every 24 h. For the control groups, dentin disks without material treatment, only 100-μL TA (8 mg/mL) or only 100-μL SF (8 mg/mL)-treated dentin disks were selected, and identical incubation conditions were applied, respectively. Remineralized dentin disks were washed with Milli-Q water three times and air-dried for further experiments. Additionally, commercial desensitizers (Gluma@ and Tianfu@) were used to treat EDTA-etched dentin disks according to the manufacturer’s instructions. Briefly, 1) Gluma desensitizer was applied to the dentin disk, left undisturbed for 60 s, and then rinsed with Milli-Q water. 2) The dentin disk was brushed with Tianfu desensitizer for 3 min and then rinsed with Milli-Q water. To simulate additional abrasive and acid challenges, the dentin disks were intensively brushed for 20 min. Subsequently, they were immersed in 50-mL 2-wt.% EDTA solution for 1 min. Finally, dentin disks were thoroughly rinsed with Milli-Q water and air-dried. Field emission scanning electron microscopy (FE-SEM, Apreo S, Thermo Scientific, USA) was used to observe the morphological changes of dentin surface minerals. XRD (Ultima IV, Japan) analysis was used to determine the crystal phase of dentin surface minerals. With a step size of 0.02o and a scan speed of 10.0 o/min, the data was evaluated. EDS was used to detect the elemental compositions of remaining minerals after brushing and EDTA-etching. 4.8 Airtightness According to the previous protocol [34], a quantitative airtightness test was conducted to evaluate the occlusion capability. Treated dentin disks were fixed between a pair of O-shaped rubber rings and fixed in the chamber. The critical air pressure was the air pressure at which the water droplet begins to move in the tube. To better observe the occlusion depth, remineralized dentin disks were directly cut along the longitudinal direction. All cross- sectional images were captured via FE-SEM. The elemental composition of minerals grown on the AcP@TS-treated DTs was analyzed via EDS. 4.9 Microhardness measurements The dentin slices from in vitro/vivo mineralization experiments were collected for further measurement by Vickers hardness test. Vickers hardness measurements were performed with a microhardness tester (TMVS-1, Times High Tech, china) at 25 g load for 10 s at 5 random locations on each dentin slice specimen (n = 5). 4.10 Fluorescence imaging The dentin disk for cLSM was prepared following a previous report. Briefly, the dentin disk was divided into two portions with different treatments, namely, the untreated portion (i.e., covered with acid-resistant nail varnish) and the calcein–AcP@TS- treated portion. This dentin disk was then washed three times with Milli-Q water to remove unbound components and then immersed in 1.5 x SBF. After 7 days of incubation, a disk of mineralized dentin was rinsed with Milli-Q water and air-dried before the test. The as-obtained dentin disk was excited using a 488 nm laser, and the cross-sectional fluorescence images were captured by cLSM. 4.11 In vitro ion release Subsequently, we investigated the in vitro ion release behavior of AcP@TS. Briefly, a 1-mL suspension of AcP@TS was placed inside a dialysis bag (MW = 3500 Da). The dialysis bag was then submerged in 10-mL Milli-Q water at 37 oc. At the predetermined time point, 200 μL of the solution was collected, followed by the addition of 200 μL of fresh Milli-Q water. The cumulative release curves were analyzed by calcium and phosphate assay kit according to the manufacturer’s operating protocol. 4.12 Mineralization in the calcium-free buffer To study the mineralization process of AcP@TS in calcium-free buffer, AcP, AcP@TA, and AcP@TS-treated dentin disks were incubated in PBS (without calcium and magnesium, pH = 7.4) for 7 days, which is identical to the protocol of incubation in SBF; for the control group, the treatment of materials was omitted and the same incubation protocol was applied. Throughout the process, PBS was replaced every 24 h. Before testing, mineralized dentin disks were washed three times with Milli-Q water and air-dried. The morphologies of dentin disks were investigated via FE-SEM. The crystalline properties and the elemental compositions were detected via XRD and EDS, respectively. 4.13 Molecular dynamics simulations All simulations were conducted using the GROMAcS 2021 software. The TA molecules were simulated using the cHARMM general force field, and the remaining molecules were simulated using cHARMM 36 force field. Langevin Integrator was applied, and the temperature was maintained at 298 K. Lennard–Jones interactions were smoothly switched off between 10 and 12 ? by a forced-based switching function. Long- range electrostatic interactions were calculated using the particle- mesh Ewald method with an error tolerance of 10–5. We successfully simulated and tested the SF model and its interaction with TA in our previous publication. The AcP or DT model parameters were used according to the previous study. Surface models for common (001) facets at a pH of 5 were applied. The configuration of a TA molecule was optimized with the approximate normal coordinate rational function optimizer (ANcopt) using the xTB 6.3 program. The Packmol package was then used to dissolve 16 or 40 configurationally optimized TA molecules and SF and AcP (or DT) in ~ 12,000 TIP3P water molecules. Further, 0.15 M of sodium chlorides were added to neutralize the simulation box. During equilibration, the X and Y dimensions of the simulation box were fixed by setting the compressibility to zero, whereas the pressure in the Z dimension was maintained by the Parrinello–Rahman barostat [52] at 1 bar. A time step of 1 fs was used, and the simulations lasted for more than 30 ns until the Z dimension of the simulation box was stable. In the production run, constant NVT (canonical ensemble) dynamics with a 2-fs time step was applied for over 200 ns. The first 5 ns of simulations were discarded during analysis.
DPcs were employed to evaluate cell compatibility in vitro. DPcs were cultured in a complete a-MEM medium (supplemented with 10% FBS and 1% penicillin-streptomycin solution). The ambient conditions were set to 37 oc with 5% cO2. The ccK-8 assay was employed to investigate the effect of each group on the cytocompatibility of DPcs. A total of 1 x 104 cells were seeded per well, and then, AcP, AcP@TA, and AcP@TS suspensions were added to a 96-well plates at the ratio of 10 μL/90 μL (Volumesuspension/Volumemedium). After 1-day incubation, the cells were stained with FDA/PI or FITc-phalloidin/DAPI according to the manufacturer’s instructions. The staining cells were imaged using a fluorescence microscopy (Olympus IX71, Japan). After 1- day incubation, a-MEM medium containing 10% ccK-8 was added to each well, followed by 3 h of incubation. The optical density at 450 nm was measured using a microplate reader (KHB ST-360, Kehua Bio-Engineering, china). The cell viability of different samples was determined by the following Eq. (1) Cell viability = (ODtest ? ODblank)/(ODcontrol ? ODblank) × 100% (1) The test groups include wells containing both cells and nanoparticles. In contrast, the control or blank groups do not include the addition of nanoparticles or cells, respectively. 4.15 In vivo occlusion of mineral-in-shell nanosystem The animal experiments were approved by the Animal Ethics committee of chengdu Dossy Experimental Animals co., Ltd, china (dossy20210509001). Female Sprague-Dawley rats (8 weeks old, 260–300 g, one sample per rat, and six rats per group) were used to conduct the animal experiments. The human dentin was sliced into rectangular disks with dimensions of 6 mm x 4 mm x 1 mm, and all disks were treated according to the aforementioned in vitro treatments. In accordance with the previous method [34], two holes were punched to fix the dentin disk in the oral cavity of rats; sequentially, a 0.20-mm stainless steel ligature wire was inserted into the interproximal space between the first and second molars, and then subsequently, the punched dentin disk was connected to the maxillary central incisors of the rat. The dentin samples were completely exposed to saliva in the oral cavity of living rats, making the occlusion process similar to that of humans. Additionally, the animals’ food supply was ground up to prevent potential appliance damage. After 14 days, the disks were removed, washed with deionized water, and air-dried. The morphology and elemental composition of each group were measured using FE-SEM and EDS. 4.16 Statistical analysis All the data were presented as the mean ± standard deviations. The statistical analyses were performed using the Origin 2018 software (OriginLab, USA). The P-values were determined by one- way ANOVA and paired samples T-tests were used to analyze the statistical significance between the two groups by using IBM SPSS Statistics 23 (IBM corporation, USA). Acknowledgements We are grateful for the financial support provided by the National Natural Science Foundation of china (Nos. 52273135, 51925304, and 52203180). We thank L. Z. Sun, L. Zhen, and S. Y. Tao for the technical assistance and valuable discussions. Electronic Supplementary Material: Supplementary material (Table S1, the stoichiometric ratios of different nanocomplexes; Figs. S1–S17, results of two studies that be performed to screen for a suitable stoichiometric ratio, AFM images of AcP, AcP@TA, and AcP@TS, TEM images and line profiles of the greyscale intensity of AcP and AcP@TA, FTIR spectra of AcP, AcP@TA, and AcP@TS, XPS spectra of AcP, AcP@TA, and AcP@TS, SEM images of EDTA-etched dentin, XRD spectra of dentin after different treatments, EDS spectrum of remaining minerals in the orifice of DTs, SEM images of commercial desensitizers treated dentin before and after tooth-brushing and EDTA-etching, EDS spectrum of the mineral grown in the interior of AcP@TS treated DTs, cross-sectional SEM images of untreated and AcP@TS treated dentin at multiple length scales, configuration analysis in MD simulation, EDS spectrum of the rod-like mineral on AcP@TS treated dentin, ion release curves of AcP@TS, cell viability of dental pulp cells which was cultured with all groups, representative live/dead and morphology cell images, and in vivo mineralization EDS spectrum of AcP@TS group) is available in the online version of this article at https://doi.org/10.1007/s12274- 023-6336-0. Source: www.thenanoresearch.com |