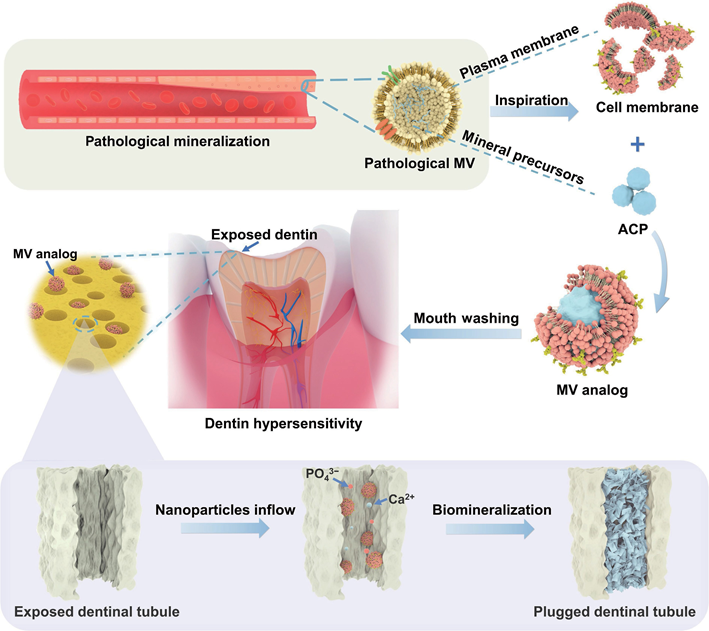
Ectopic Mineralization-Inspired Cell Membrance-Based Matrix Vesicle Analogs for In-Depth Remineralization of Dentinal Tubules for Treating Dentin HypersensitivityIssuing time:2025-04-11 10:25 Mingjing Li, Xiaoran Zheng, Zhiyun Dong, Yuyue Zhang, Wei Wu, Xingyu Chen, Chunmei Ding, Jiaojiao Yang4, Jun Luo, and Jianshu Lee Abstract The invasion of etched dentinal tubules (DTs) by external substances induces dentin hypersensitivity (DH). The deep and compact occlusion of DTs is highly desirable for treating DH but still challenging due to the limited penetrability and mineralization capacities of most current desensitizers. Matrix vesicles (MVs) participate in the regulation of ectopic mineralization. Herein, ectopic MV analogs are prepared by employing natural cell membranes to endow mineral precursors with natural biointerfaces and integrated biofunctions for stimulating dentin remineralization. The analogs quickly access DTs (> 20 μm) in only 5 min and further penetrate deep into the interior of DTs (an extraordinary ~ 200 μm) in 7 days. Both in vitro and in vivo studies confirm that the DTs are efficiently sealed by the newly formed minerals (> 50 μm) with excellent resistance to wear and acid erosion, which is significantly deeper than most reported values. After repair, the microhardness of the damaged dentin can be recovered to those of healthy dentin. For the first time, cell membrane coating nanotechnology is used as a facile and efficient therapy for in-depth remineralization of DTs in treating DH with thorough and long-term effects, which provides insights into their potential for hard tissue repair. Keywords bioinspiration, dentin remineralization, cell membrane coating nanotechnology, ectopic matrix vesicles, mineral precursors 1. Introduction Matrix vesicles (MVs), a kind of extracellular vesicles, are membrane-bound microparticles secreted by cells that act as initiators for biomineralization in both physiological and pathological mineralization processes. Mineral precursors and plasma membranes are the key components of MVs for biomineralization. The plasma membrane, as the outer membrane of MVs containing functional proteins and lipids, possesses the potential to induce hydroxyapatite formation in vivo. For example, alkaline phosphatase (ALP) tethered to the plasma membrane can promote the hydrolysis of both pyrophosphate (an effective calcification inhibitor) and phospholipids, and the released phosphate from phospholipids can bind calcium ions. During the physiological mineralization process, MVs originating from the plasma membrane of mineral-forming cells (i.e., osteoblasts, chondrocytes, and odontoblasts), consisting of mineral precursors surrounded by plasma membranes, can promote the biomineralization of bones, cartilages, and teeth. During the ectopic mineralization, MVs play a similar role; however, they mainly originate from nonmineral-forming cells (e.g., macrophages and smooth muscle cells). They can actively regulate the mineralization process during ectopic mineralization, resulting in mineral formation in inappropriate positions, e.g., in blood vessels. MVs originating from macrophages and vascular smooth muscle cells can reach the vascular matrix to facilitate the ectopic deposition of apatite minerals in blood vessel wall cells and in extracellular matrices, which can cause vascular calcification. MVs also play a key role in the budding stage of dentin formation, where odontoblasts secrete MVs as the mineral initiator and template to induce biomineralization . However, in mature dentin, due to the lack of odontoblasts, few MVs are present. Therefore, the mature dentin cannot be recovered via spontaneous biomineralization after being damaged. Because of gingival recession or enamel thinning caused by overbrushing and erosion, dentinal tubules (DTs) are easily exposed, and the fluid flow within the exposed tubules is sensitive to external stimuli, which causes dentin hypersensitivity (DH) accompanied by transient and severe pain. Currently, the most effective DH treatment strategy is dentin desensitization by occluding exposed DTs. Dentin adhesives, sealing resins, remineralization coatings, and mineral nanoparticles (NPs) are the most commonly used desensitizers to block DTs. However, traditional clinical treatment methods are inclined to slough off due to their limited sealing capacity, e.g., dentin adhesives can only seal the dentin surface, and therefore, the sealing layer tends to slough off due to acid etching or mechanical friction (e.g., tooth brushing). In addition, complex synthesis methods, complicated operations, lengthy courses of treatment, and the potential biosafety hazards of synthetic desensitizers significantly affect their clinical outcomes. Thus, it is imperative to develop easy and secure methods for the rapid and deep occlusion of DTs. The recent development of cell membrane coating nanotechnology directly leverages natural cell membranes to endow nanoparticles with the inherent biofunctions of cell membranes. This nanotechnology can also ensure a rightside-out cell membrane orientation, leading to the exposure of more functional proteins on the outside of nanocomposites. In addition, the cell membrane coating can protect the inner nanoparticles and facilitate their dispersal to achieve uniformly small particle sizes and long-term stability. Among the various types of cell membranes, red blood cell membranes (RBcMs), the most widely used ones, display several advantages: widespread sources, ease of collection, low immunogenicity, and rich in functional proteins. More importantly, there are many functional proteins on the RBcMs that favor biomineralization. For example, calmodulin on RBcMs can regulate the intracellular calcium concentration under normal physiological conditions, and maintain continuous calcium transport under pathological conditions. In particular, a recent research demonstrates that intraplaque hemorrhaging promotes the pathological calcification of atherosclerosis, which alludes to the contribution to the calcification potential of RBcMs. Therefore, in this work, inspired by the roles of MVs originating from nonmineral-forming cells during pathological ectopic mineralization, ectopic mineralization-inspired MVs are designed for dentin remineralization by directly leveraging the RBcMs to coat a classical type of mineral precursor (i.e., amorphous calcium phosphate (AcP) nanoparticles) (denoted RBcM@AcP NPs) (Scheme l). AcP NPs, as the core of RBcM@AcP NPs, are employed to provide continuous calcium and phosphate ions for biomineralization. Additionally, with liquid-like fluidity, moldability, and excellent biocompatibility, AcP NPs could be the foundation of structuring sophisticated minerals in the microstructure for hard tissue repair. Moreover, AcP NPs are flexible, which would benefit their reorganization and fusion in the cell membrane coating process. Besides, in the process of dentin mineralization, AcP NPs could penetrate into the collagen matrix and facilitate the formation of biominerals in situ. RBcMs with potential mineralization ability, as the membrane of RBcM@AcP NPs, can hinder premature crystallization from AcP NPs, facilitate the penetration of the nanoparticles to the deep regions of DTs, and act as a biomineralization template in situ. RBcM@AcP NPs can rapidly and deeply penetrate into DTs (> 20 μm) in 5 min and induce effective remineralization in situ to achieve deep sealing (> 50 μm) both in vitro and in vivo. The formed minerals in DTs, tightly bound to the collagen matrix, have excellent resistance to wear and acid erosion. In vivo studies also show that the microhardness of dentin can recover to its healthy state after l4 d of treatment. This bioinspired method can overcome the drawbacks of existing clinical treatment methods because it involves facile material preparation and operation, shows excellent biocompatibility, and achieves rapid and deep occlusion of DTs. In addition, for the first time, cell membrane coating nanotechnology is applied for biomineralization, which provides a new avenue for future research into cell membranes.
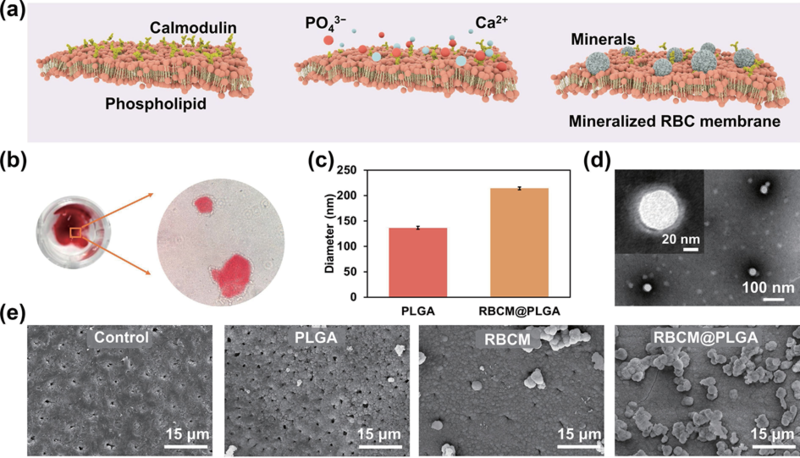
Scheme 1 Schematic of ectopic mineralization-inspired MV analogs consisting of AcP cores surrounded and stabilized by RBcMs for deep mineralization and occlusion of DTs. 2. Discussion 2.1 Characterization of RBCM nanofragments and RBCMs-coated nanoparticles To validate the potential of RBcM nanofragments for biomineralization (Fig. l(a)), RBcMs after ultrasonication were cultured in a calcium-containing medium for 7 days and stained with alizarin red to mark calcium salts. The RBcMs turned red (Fig. l(b)), indicating that the RBcM nanofragments could combine with calcium and promote mineralization. This may be due to the abundant functional proteins (e.g., calmodulin) and phospholipids presenting on the erythrocyte membranes. Coating RBcMs on the surface of nanoparticles can enhance the colloidal stability of the particles and ensure a right-side-out configuration with more functional proteins exposed to the outside [3l]. Therefore, we assessed the mineralization capacity of RBcMs when coated onto nanoparticles. Poly(lactic-co-glycolic acid) nanoparticles (PLGA NPs) were selected as the core particles since they are hydrophobic and biocompatible, but they have no ability to promote mineralization. RBcMs-coated PLGA NPs (RBcM@PLGA NPs) were prepared by a coextrusion method. After coating with the erythrocyte membranes, the average hydrodynamic size of the PLGA NPs increased to approximately 2l0 nm compared with approximately l40 nm for the bare PLGA NPs (Fig. l(c)), indicating the successful coating of the erythrocyte membrane. The shell–core structure of the RBcM@PLGA NPs is also clearly observed in Fig. l(d) compared with the bare PLGA NPs (Fig. Sl in the Electronic Supplementary Material (ESM)). 2.2 Biomineralization potential of RBCM nanofragments and RBCMs-coated nanoparticles in vitro Then, an in vitro human dentin model was used to explore the biomineralization potential of the RBcM@PLGA NPs. After incubation in simulated body fluid (SBF) for 7 days, dentin slices with different treatments were observed with scanning electron microscopy (SEM) (Fig. l(e)). The dentin slices treated with water (the control group) and PLGA NPs (the PLGA group) were not completely blocked. In contrast, in both the RBcMs (the RBcM group) and RBcM@PLGA NPs-treated dentin slices (the RBcM@PLGA group), the DTs were sealed with new mineral layers. Notably, more mineral crystals were observed in the RBcM@PLGA group than in the RBcM group, which may be due to stable nanostructures and abundant exposed functional proteins, making the RBcM@PLGA NPs more suitable as nucleation sites and for ca2+ binding than RBcMs alone.
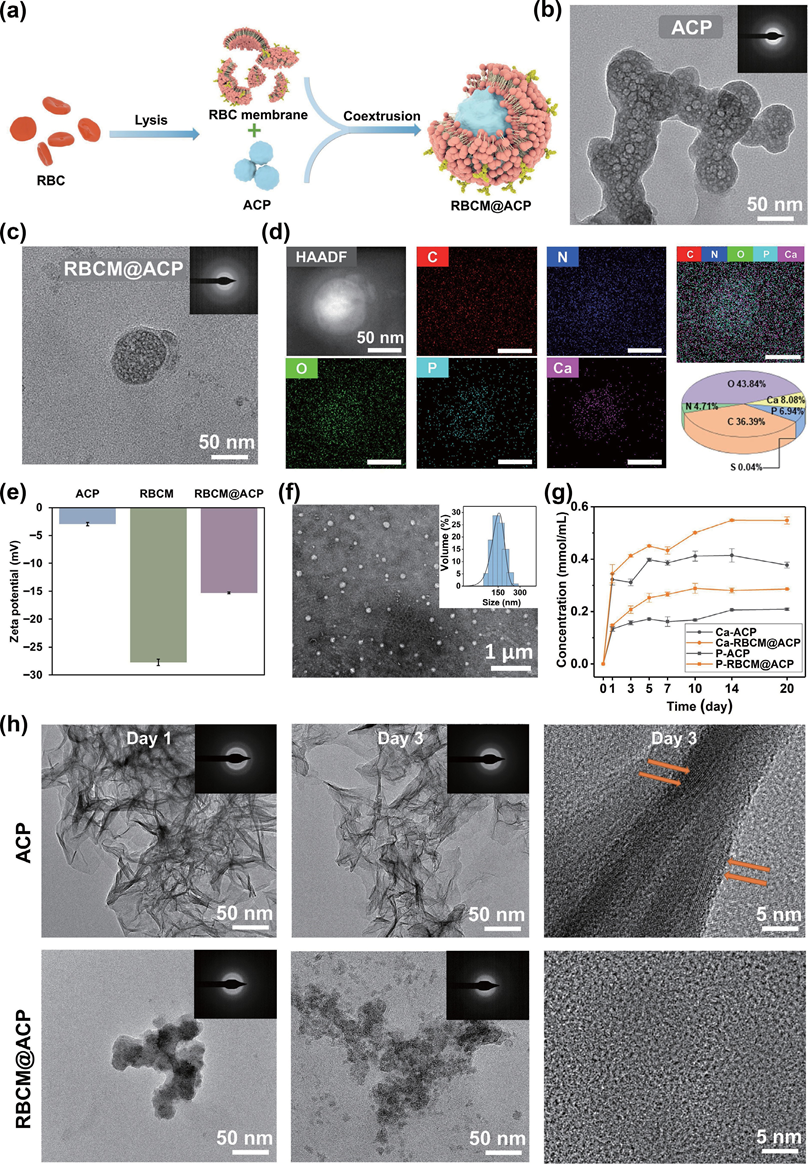
Figure 1 Preparation and application of RBcM@PLGA NPs. (a) Diagram of the mechanism of cell membrane-induced mineralization. (b) Alizarin red staining of mineralized RBcMs. (c) Hydrodynamic size (diameter) of PLGA NPs and RBcM@PLGA NPs. (d) TEM images of RBcM@PLGA NPs. (e) SEM images of remineralized DTs treated with different materials after incubation in SBF for 7 days. 2.3 Characterization of ectopic MV analogs (RBCM@ACP NPs) Dentin is composed of a mineralized extracellular matrix with 70% inorganic minerals and 20% organic matrix. To simulate the MVs present during the ectopic mineralization, we coated AcP NPs, widely used mineral precursors and effective sources of calcium and phosphate ions, with RBcMs for the first time to obtain ectopic MV analogs (RBcM@AcP NPs) with similar dentin organic–inorganic components for the aim of promoting the mineralization effect (Fig. 2(a)). After coating with the cell membrane, a distinct cell membrane layer was observed in the transmission electron microscopy (TEM) image (Fig. 2(c)) compared with the AcP NPs (Fig. 2(b)). An enrichment in N, O, ca, and P was also seen in the mapping images (Fig. 2(d)), which further indicated RBcMs coating. The zeta potential of the AcP NPs dramatically decreased to –l5 mV (Fig. 2(e)) After coating with the RBcMs in the RBcM@AcP group, which further affirmed the formation of a biomimetic surface. The results of dynamic light scattering (DLS) measurements showed that the hydrodynamic diameter of the RBcM@AcP NPs was approximately l60 nm (Fig. 2(f)), which was similar to that of the MVs. No valid size data was obtained from DLS measurements for the AcP NPs because of their aggregation. This result revealed that the RBcMs coating benefits the stabilized dispersion of AcP NPs (Figs. S2 and S3 in the ESM). This may be due to the strong bonding between the phosphate groups on the cell membrane and the calcium ions on the surface of the AcP NPs. The release profiles of calcium and phosphate ions from the AcP NPs and RBcM@AcP NPs showed that more calcium and phosphate ions were released from the RBcM@AcP NPs (Fig. 2(g)), confirming that the RBcMs coating can retard the crystallization process of AcP NPs. After incubation in an aqueous solution for 3 days, the distinct crystal orientation growth of the AcP NPs, which was due to the metastable property of AcP NPs, was visualized by high resolution transmission electron microscope (HRTEM). In contrast, at the same time point, the AcP cores in the RBcM@AcP group still maintained small particle sizes, and the crystallization from the AcP NPs was noticeably retarded (Fig. 2(h)). This revealed that the coating of the cell membrane can not only facilitate the dispersion of AcP NPs but also reduce the rate of crystallization of AcP NPs, which would be beneficial for the storage, transport, and mineralization of these materials.
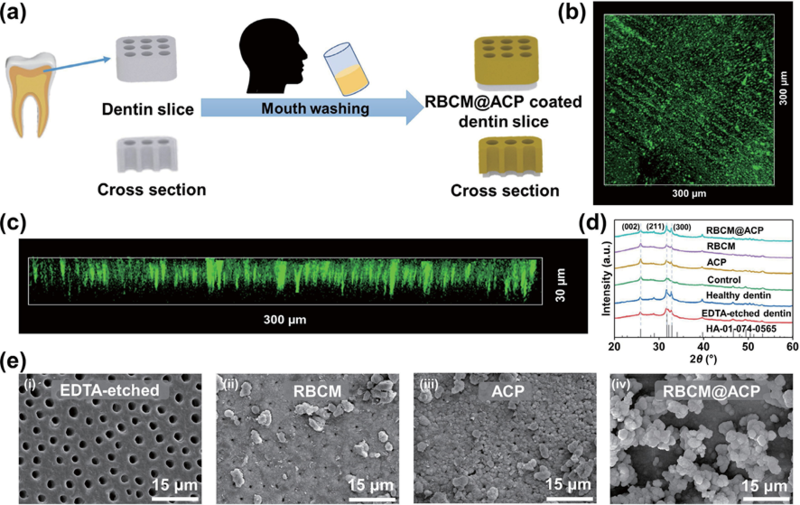
Figure 2 Preparation and characterization of RBcM@AcP NPs. (a) Preparation chart of RBcM@AcP NPs. TEM images of (b) AcP NPs and (c) RBcM@AcP NPs. (d) The results of high-angle annular dark field (HAADF) image, EDS, and elemental mapping analysis of RBcM@AcP NPs. (e) Zeta potential of AcP NPs, RBcMs, and RBcM@AcP NPs. (f) The hydrodynamic diameter distribution of RBcM@AcP NPs from DLS tests (insert) and their negatively stained TEM image. (g) Release curves of calcium and phosphate ions from AcP NPs and RBcM@AcP NPs. (h) TEM, SAED, and HRTEM images of AcP NPs and RBcM@AcP NPs stored for l and 3 days at 4 oc. 2.4 Biomineralization capacity of RBCM@ACP NPs in vitro Typical DH treatments generally require a long treatment time, and they still do not achieve satisfactory outcomes due to the shallow occlusion of DTs, thus lacking long-term stability. To achieve the deep plugging of DTs for the DH treatment, the material needs to not only cover the surface of the dentin but also access the deep regions of the DTs to induce mineralization in situ and therefore achieve effective and stable occlusion (Fig. 3(a)). The confocal laser scanning microscopy (cLSM) images (the RBcMs were dyed with 3,3′ -dioctadecyloxacarbocyanine perchlorate (Dio)) showed that the RBcM@AcP NPs could cover the dentin surface and enter the DTs at a depth of more than 20 μm within 5 min (Figs. 3(b) and 3(c)), which is much deeper than the penetration depth of the RBcM vesicles (~ l0 μm, Fig. S4 in the ESM). The RBcM vesicles (Fig. S5 in the ESM) have a relatively stable particle structure and wider particle size distribution than RBcM@AcP NPs. This revealed that the stable nanoparticle structure of the RBcM@AcP NPs may benefit their rapid and deep penetration in the DTs. To further verify the mineralization effect of the nanoparticles, ethylenediaminetetraacetic acid (EDTA)-etched dentin slices treated with different materials were incubated in SBF for 7 days. The dentin slices treated with RBcM@AcP NPs could rapidly mineralize and completely block DTs with a denser mineral layer compared with the other groups (Fig. 3(e)). X-ray diffraction (XRD) and energy-dispersive spectroscopy (EDS) results showed that the main component of the new mineral layer was hydroxyapatite, which was similar to the inorganic substance in healthy dentin (Fig. 3(d) and Fig. S6 in the ESM).
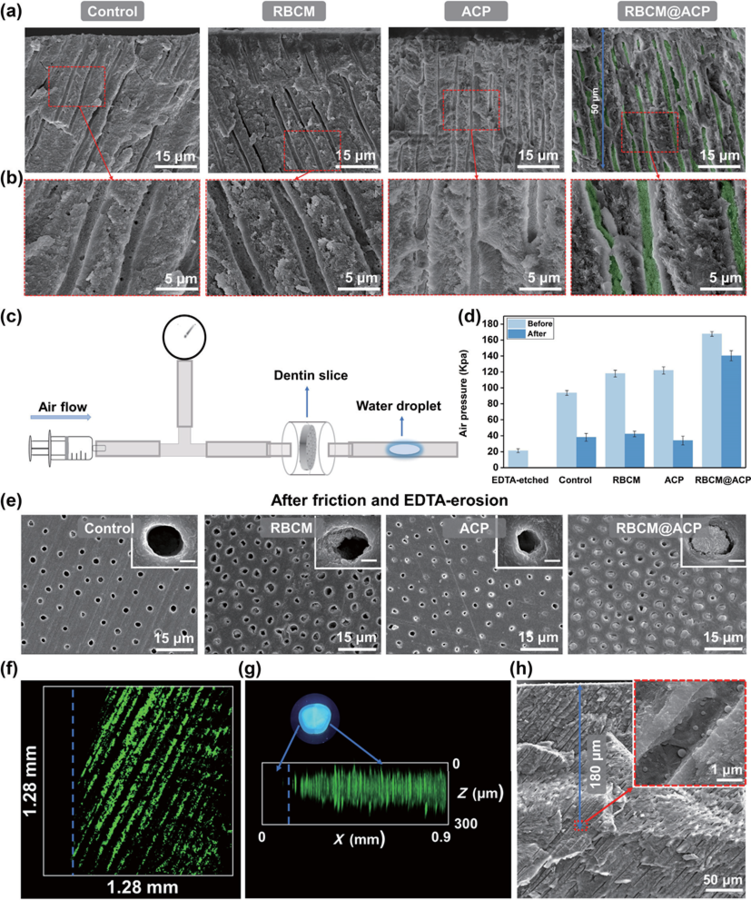
Figure 3 (a) Schematic illustration of RBcM@AcP NPs covering and penetrating DTs. (b) Top and (c) side views of cLSM images of dentin slices after soaking in a Dio-stained RBcM@AcP NPs solution for 5 min. (d) XRD curves of dentin slices handled with different methods. (e) SEM images of (i) acid-dentin, (ii) RBcMs solution-soaked dentin, (iii) AcP NPs solution-soaked dentin, and (iv) RBcM@AcP NPs-treated dentin incubated in SBF for 7 days. 2.5 In-depth remineralization of DTs via RBCM@ACP NPs in vitro As the average diameter of DTs is approximately l.7 μm, materials with nanoscale sizes are suitable for penetrating DTs. As the deep occlusion of DTs with long-term stability is crucial for DH treatment, the efficacy of deep mineralization of DTs by nanoparticles was assessed. After incubation in SBF for 7 days, the dentin slices were quenched in liquid nitrogen, and their internal structures were observed by SEM (Figs. 4(a) and 4(b)). New flake minerals were observed in the DTs in each group except for the control group. compared with the remineralized dentin slices treated with AcP NPs and the RBcMs, the internal minerals in the DTs of the dentin slices treated with RBcM@AcP NPs were clearly denser. In addition, a clear and deep plugging of DTs to a mineral depth of more than 50 μm was achieved in the RBcM@AcP group. The EDS results show that the new minerals are calcium-phosphate composites (Fig. S7 in the ESM). To evaluate the macro sealing effect of DTs, we adopted the airtightness test (Fig. 4(c)). compared with the dentin slices etched with EDTA, after incubation in SBF for 7 days, all the dentin slices had higher maximum air pressures due to the formation of new minerals in the DTs (Fig. 4(d)). The dentin slices in the RBcM@AcP group had higher airtightness than those in the other groups, indicating that these nanoparticles with stable dispersion may play an important role in promoting deep mineralization. In daily life, chewing and food residues can wear and erode tooth mineral layers, causing further damage, which also makes the DH treatment more difficult. Therefore, we used mechanical friction and acid etching simulations to explore the persistence of the new mineral layer. After removing the mineral layer from the surface of the dentin slices, only the air pressure of the RBcM@AcP NPs-treated dentin slices did not strongly decrease and was significantly higher than that of the other groups (Fig. 4(e)), even compared with the dentin slices treated with commercially available products (i.e., Gluma and Tianfu) (Fig. S8 in the ESM). The SEM images of the dentin slices before and after friction and EDTA erosion also confirmed that the dentin slices treated with RBcM@AcP NPs had better wear and acid resistance, as almost all the DTs were still blocked even after friction and EDTA erosion, which was much better than that observed in the other dentin slices (Figs. 3(e) and 4(e) and Fig. S9 in the ESM). In comparison, the two commercially available products cannot block the DTs after friction and EDTA erosion (Fig. S9 in the ESM). Mechanical friction and acid etching can only remove minerals from the dentin surface rather than removing mature minerals from the deep region of DTs, further indicating the importance of deep mineralization for long-term occlusion during DH treatment. To further explore the deep penetration potential of RBcM@AcP NPs in the DTs, fluorescent-labeled AcP NPs were employed to prepare fluorescent-labeled RBcM@AcP NPs. The cLSM results showed that the boundary between the fluorescent- labeled RBcM@AcP NPs-treated and acid-etched dentin sections (protected by nail polish during the treatment) was clear, and fluorescence was detected at a depth of more than 200 μm for the RBcM@AcP NPs-treated area (Figs. 4(f) and 4(g)). Notably, nanoparticles with particle sizes similar to those of the RBcM@AcP NPs on the walls of the DTs were also found approximately l80 μm beneath the dentin surface (Fig. 4(h)). This shows that this material has good wettability and can diffuse into the deep region of DTs, which may be due to the superhydrophilic and antiadhesion characteristics of the cell membrane and the Brownian motion of nanoparticles.
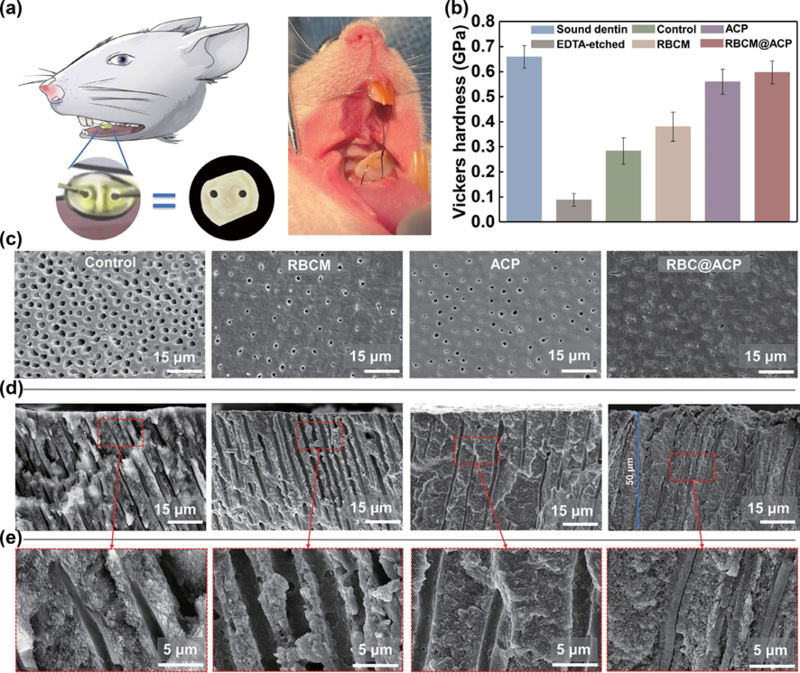
Figure 4 (a) and (b) cross-sectional SEM images of dentin slices treated with different materials after soaking in SBF for 7 days. (c) Schematic diagram of the air pressure test. (d) Air pressure graph of dentin slices treated with different methods before and after friction and EDTA erosion. (e) SEM images of remineralized dentin slices after mechanical friction and EDTA erosion. cLSM images of (f) the surface and (g) cross-section of remineralized dentin dyed with calcein, in which the left area of the blue dotted line (displayed as dark) was protected with nail polish. (h) SEM images of RBc@AcP NPs-treated dentin slices incubated in SBF for 7 days. Scale bars, l μm ((e), inset). 2.6 In-depth remineralization of DTs via RBCM@ACP NPs in vivo Both the endogenous RBcMs and AcP NPs have superior biocompatibility. To further investigate the potential of RBcM@AcP NPs to be applied in the oral environment, we chose human dental pulp cells (HDPcs) and human umbilical vein endothelial cells (HUVEcs) to evaluate the biocompatibility of RBcM@AcP NPs. As expected, all groups (i.e., RBcM vesicles, AcP NPs, and RBcM@AcP NPs) co-cultured with both HDPcs and HUVEcs, respectively, exhibited good cell viability (Figs. Sl0 and Sll in the ESM). Furthermore, as shown in the results of the live/dead assays (Fig. Sl2 in the ESM) and the cell morphologies (Fig. Sl3 in the ESM) of HDPcs, all groups had virtually no dead cells and the cells were intact with consistent cell morphologies, indicating excellent cytocompatibility of RBcM@AcP NPs. Encouraged by the results of the in vitro experiments, we used a rat oral model to further evaluate the practical value of RBcM@AcP NPs in an actual oral environment (Fig. 5(a)). We placed dentin slices into SD rat oral cavities to simulate mineralization in real cases. After 7 days of incubation in the oral cavity, intrafibrillar mineralization was only found in the RBcM@AcP group (Fig. Sl4 in the ESM). The tight bonding between the newly formed minerals and the collagen matrix gave the remineralized dentin excellent resistance to wear and acid erosion. After l4 d of incubation in the oral cavity of rats, the Vickers hardness of the dentin surfaces treated with the RBcM@AcP NPs was similar to that of healthy dentin (Fig. 5(b)). However, only DTs in the RBcM@AcP NPs-treated dentin sheets were efficiently blocked by newly formed minerals (Fig. 5(c)). In addition, in the cross-sectional SEM images (Figs. 5(d) and 5(e)), we observed that the newly formed minerals had penetrated into the DTs to a mineral depth of more than 50 μm, which was also consistent with the results of the in vitro experiments. The EDS results showed that the main components of the new minerals were calcium and phosphorus (Fig. Sl5 in the ESM). Interestingly, although the RBcM@PLGA NPs had a good mineralization effect in SBF (Fig. l(e) and Fig. Sl6 in the ESM), they could not efficiently block DTs in the oral environment (Fig. Sl7 in the ESM), which may be due to the low calcium and phosphorus content in the real oral environment. This result was consistent with the results of the mineralization experiment in a calcium-free environment, as some minerals only formed on the wall of DTs in the RBcM@AcP NPs-treated dentin slices (Figs. Sl8 and Sl9 in the ESM). In the RBcM@AcP group, the cell membrane coating stabilized the AcP NPs and therefore facilitated the release of calcium and phosphate ions from the AcP NPs. Thus, RBcM@AcP NPs can act not only as growth sites for hydroxyapatite but also serve as a continuous source of calcium and phosphate ions for self-mineralization in situ, which is similar to the mineralization mechanism of MVs during ectopic mineralization. Above all, the results of the in vivo experiments further verified the potential of RBcM@AcP NPs for the DH treatment.
Figure 5 (a) Schematic of the in vivo experiment. (b) Vickers hardness of dentin slices after soaking in SBF for 7 days. Measurements were taken from distinct samples (n = 5). (c) SEM images of remineralized dentin surfaces after mechanical friction and EDTA erosion. (d) and (e) SEM images of a cross section of dentin. 3. Conclusion In conclusion, we developed ectopic mineralization-inspired MV analogs (RBcM@AcP NPs) based on natural cell membranes (i.e., RBcMs) and mineral precursors (i.e., AcP NPs) for the DH treatment. RBcMs provide an ideal nucleation site for crystallite deposition and ensure better dispersal and stability of the AcP cores. The continuous release of calcium and phosphate ions originating from the stabilized AcP cores can facilitate self- biomineralization in situ. Therefore, the MV analogs can quickly penetrate the deep region of DTs, acting as mineral nucleation sites and efficiently providing calcium and phosphate ions, fulfilling the need for deep mineralization to block DTs. Moreover, remineralized dentin is wear-resistant and acid-resistant, which can effectively block DTs to achieve long-term stability. As an emerging type of anti-immunogenic material, cell membrane-based nanomaterials have not yet been utilized for dentin mineralization. These strategies may open new avenues for their application to hard tissue repair. 4. Experimental section 4.1 Materials PLGA (MW = 90,000, 50/50) was purchased from Meilun Biotechnology co., Ltd. (Dalian, china). Dimethylformamide (DMF), EDTA, calcium chloride (cacl2), disodium hydrogen phosphate (Na2HPO4), and hydrogen chloride (Hcl) were purchased from chron chemicals co., Ltd. (chengdu, china). Dio was purchased from Beyotime Biotechnology co., Ltd. (Jiangsu, china). Alizarin red S solution (l%, pH = 4.2), calcein, fluorescein diacetate/propidium iodide (FDA/PI), fluorescein isothiocyanate (FITc)-phalloidin, and 4’,6-diamidino-2- phenylindole dihydrochloride (DAPI) were purchased from Solarbio Science & Technology co., Ltd. (Beijing, china). calcium assay kits and phosphate assay kits were purchased from Jiancheng Bioengineering Institute co., Ltd. (Nanjing, china). Phosphate buffered saline (PBS) was purchased from Labgic Technology co., Ltd. (Beijing, china). β-Glycerophosphate disodium salt hydrate (β-GP) was purchased from Macklin Biochemical co., Ltd. (Shanghai, china). Dulbecco’s modified Eagle’s medium (DMEM), fetal bovine serum (FBS), and a penicillin-streptomycin solution were purchased from Gibco Life Technologies co., Ltd. (Grand Island, USA). cell counting kit-8 (ccK-8) was purchased from MedchemExpress co., Ltd. (State of New Jersey, USA). 4.2 Preparation of RBCMs The animal study was approved by the Animal Ethics committee of chengdu Dossy Experimental Animals co., Ltd., china. Whole blood collected from rabbits by cardiac puncture was transferred into tubes with EDTA for anticoagulation, and then centrifuged (3000 rpm, 5 min) to remove the plasma and leukocytes. After breaking the RBcs with 0.25x PBS at –20 oc, the released hemoglobin was removed by centrifugation (l2,000 rpm, 5 min), and the membranes were washed 2–3 times with ultrapure water until the supernatant was colorless. Then, the RBcMs were stored at –80oc. 4.3 Preparation and characterization of PLGA NPs, ACP NPs, RBCM@PLGA NPs, and RBCM@ACP NPs PLGA NPs were synthesized using a solvent displacement technique. PLGA was dissolved in DMF (4 mg/mL), and then the solution was added dropwise to 4 mL of ultrapure water while stirring at 200 rpm. The PLGA NPs were obtained by dialysis in water for 4 h, with the water changed every 0.5 h. Solutions of 0.006 M cacl2 and 0.004 M Na2HPO4 were mixed at a ratio of l:l for 2 min at 4 oc to obtain the AcP NPs. To form NPs coated with the RBcMs, after 5 min of ultrasonication, 5 mL NPs dispersion and RBcM vesicles extracted from l mL of whole blood were serially extruded through 0.2 μm porous membranes. The prepared RBcM@PLGA NPs and RBcM@AcP NPs were used immediately. Residual AcP NPs and RBcM@AcP NPs were reserved at 4 oc for the stability test. 4.4 Characterization of NPs The hydrodynamic diameter (Dh) and zeta potential (?) of all prepared NPs were characterized in a Zetasizer Nano (Malvern, ZS90, UK). Each group was tested three times to ensure accuracy. To determine the morphology of the NPs, the solution was dropped onto a TEM support grid and dried. Then, the NPs were observed with TEM (Talos F200S, Thermo Fisher Scientific, USA), and the NPs coated with the RBcMs were negatively stained with phosphotungstic acid before analysis. The elemental composition of the RBcM@AcP NPs was determined by EDS (SUPER X, Thermo Fisher Scientific, USA). The crystal morphology was verified by selected area electron diffraction (SAED). To evaluate long-term stability, the sample solutions of groups AcP NPs and RBcM@AcP NPs were stored at 4 oc for l and 3 days, respectively. Then each sample solution was dropped on a TEM support grid and air dried before TEM analysis. The Tyndall effect of groups AcP NPs and RBcM@AcP NPs stored at 4 oc was observed on day l, day 3, day 5, and day 7, respectively. To verify the mineralization capacity of RBcM nanofragments, the RBcMs after ultrasonication for 5 min were incubated with DMEM (l0% FBS, l% double, l0 mmol β-GP) for 7 days in 48- well plates. RBcM nanofragments were dyed with alizarin red solution for 3 min, washed with ultrapure water, centrifuged 3 times, and then observed under a microscope. The prepared solutions of the RBcM@AcP and AcP NPs were placed in glass bottles (5 mL in each bottle), sealed with a dialysis bag, and then placed in a centrifuge tube containing 20 mL of ultrapure water. We collected 500 μL of liquid after a certain time interval and added 500 μL of ultrapure water. The liquid was added to calcium and phosphate kits and analyzed by a MicroplateReader (Spectra Plus, Tecan, Zurich, Switzerland). Measurements were taken from distinct samples (n = 3). 4.5 Preparation of hypersensitive dentin Permanent teeth were acquired from Huaxi Stomatological Hospital of Sichuan University. After the teeth were cleaned, the root and crown were cut off with a cutting and grinding machine to obtain dentin slices. The dentin slices were polished with 600, 800, l000, l500, and 2000 mesh sandpaper to obtain smooth surfaces without scratches. Then, the dentin was etched with EDTA for l0 min to fully expose the DTs and simulate DH. The study was approved by the Medical Ethics committees of Huaxi Stomatological Hospital of Sichuan University (WcHSIRB-D- 202l-078). 4.6 RBCM@ACP NPs as interfacial templates for the occlusion of DTs For the in vitro experiment, acid-etched and nitrogen-dried dentin slices were soaked in 5 mL of RBcM@AcP NPs for 5 min. After the dentin was cleaned with ultrapure water, the treated dentin slices were incubated in l.5x simulated body fluid (SBF: l37.5 mM Nacl, 4.2 mM NaHcO3, 3 mM Kcl, l.5 mM K2HPO4?3H2O, l.5 mM Mgcl2?6H2O, 3.75 mM cacl2, and 0.5 mM Na2SO4) for 7 days. l.5x SBF was replaced every 24 h. Dentin slices were reserved after cleaning and drying in a vacuum. The surface morphology of the dentin slices was studied by SEM (Apreo 2c, Thermo Fisher Scientific, USA) with gold sputtering. To observe the DTs, cross-sections of dentin were observed after the sample was fractured. EDS (Ultim Max65, Oxford Instruments, UK) was used to measure the calcium/phosphorus ratio of the regenerated minerals. XRD (Ultima IV, Rigaku, Japan) was applied to analyze the crystalline form of each sample. Two commercial products, Gluma? and TianFu herbal desensitizer?, were applied to the dentin following the manufacturers’ specifications. And then these dentin slices were incubated in l.5x SBF for 7 days, and l.5x SBF was replaced every 24 h, which was consistent with the RBcM@AcP group. As only unblocked DTs can allow for gas flow, the macroscopic sealing effect can be evaluated by assessing permeability with an air pressure infiltration setup. As shown in Fig. 4(c), the dentin slices were attached to the tee, the gas was blown, and we recorded the barometer readings as the water drops moved [25]. To simulate friction and etching in an oral environment, remineralized dentin slices were treated with a 2% EDTA aqueous solution for 2 min after mechanical friction was applied. Finally, the sealing effect was detected by air pressure testing. Measurements were taken from distinct samples (n = 4). 4.7 Depth characterization of RBCM@ACP NPs entering DTs The RBcMs were resuspended in a l0 μM Dio dye solution for l0 min in the dark and then cleaned by centrifugation with ultrapure water. After the stained RBcMs were coextruded with AcP NPs, the EDTA-etched dentin slice was immersed in a 5 mL solution. The treated dentin slices were observed with cLSM (LSM700, ZEISS, Germany) under a 488 nm laser, and the fluorescence depth was scanned in Z-stack mode. To explore the potential depth of dentin remineralization induced by the RBcM@AcP NPs, we blocked half of the EDTA- etched dentin slice with varnish. Then, 5 μL of l mg/mL calcein was added to 5 mL of the AcP NPs solution, dialyzed in the dark at 4 oc for 4 h, and then coextruded with RBcMs. The half- blocked dentin slice was dipped in a calcein-dyed RBcM@AcP NPs solution for 5 min and incubated in SBF for 7 days. The fluorescence depth of the remineralized dentin slices was detected by cLSM under a 488 nm laser. 4.8 In vitro cytotoxicity assay To evaluate the biocompatibility of RBcM@AcP NPs, HDPcs and HUVEcs were employed to assess the cellular viability by a ccK-8 test. HDPcs and HUVEcs were cultured in a DMEM complete medium consisting of l0% FBS and l% penicillin- streptomycin solution, respectively. Each type of the cells (approximately l05 cells/mL, l00 μL) were seeded in a 96-well plate for 24 h at 37 oc. Then the DMEM medium was removed and l00 μL of the DMEM medium with l0% AcP NPs, RBcM NPs, and RBcM@AcP NPs was added and incubated with each type of the cells for another 24 h, respectively. Meanwhile, cells in the DMEM medium were served as the control group. Afterward, l00 μL of the DMEM medium with l0% ccK-8 solution was added. After 2 h of incubation, the optical density (OD) at 450 nm was tested by a microplate reader (KHB ST-360, Kehua co., Ltd., china). Besides, the live/dead cells and cell morphologies of HDPcs were recorded by a fluorescence microscope. HDPcs (approximately 5 x l04 cells/mL, 500 μL) were seeded in a 24-well plate for 24 h. Then the DMEM medium was removed and l00 μL of the DMEM medium with l0% AcP NPs, RBcM NPs, and RBcM@AcP NPs was added and incubated with the cells for another 24 h, respectively. After washing with PBS, HDPcs were stained with FDA/PI for l5 min. After washing with PBS again, the cells in each group were observed with an inverted fluorescence microscope (IX7lSlF-3, OLYMPUS, Japan). HDPcs were fixed by 4% paraformaldehyde for l5 min and then stained with FITc-phalloidin and DAPI before observing their morphologies via the inverted fluorescence microscope. 4.9 Evaluation of remineralization effect in vivo The animal study was approved by the Animal Ethics committee of chengdu Dossy Experimental Animals co., Ltd., china. Eight- week-old male SD rats weighing 220–300 g were selected for animal experiments (5 rats in each group). Two holes were symmetrically punched in dentin slices, and the other treatment was consistent with the in vitro experiments. The rats were anesthetized with pentobarbital sodium (l%). A low-speed straight machine was used to punch and penetrate the distal middle of the two maxillary central incisors near the center of the gingival end. Root canal files were used to enlarge the gap between the first and second molars. Then, the steel spring wire from the dentin slices was passed through the gap between the first molar and the second molar, and the wire was passed through the middle incisor of the rat, and the wire was twisted and fixed to cut off the superfluous wire. The rats were fed soft food. After l4 d, the rats were sacrificed, and samples were collected for further analysis. The Vickers hardness was measured by a microhardness tester (TMVS-l, Time High Technology, china) by using a load of 25 g for l0 s in 5 random places on the dentin slices [5l]. The surface and cross section of the samples were tested by SEM and EDS as described above. Acknowledgements We are grateful for the financial support of the National Natural Science Foundation of china (Nos. 5l925304, 5l903l75, and 5l973l33). Sources: www.theNanoResearch.com | www.Springer.com/journal/12274 | Nano Research |