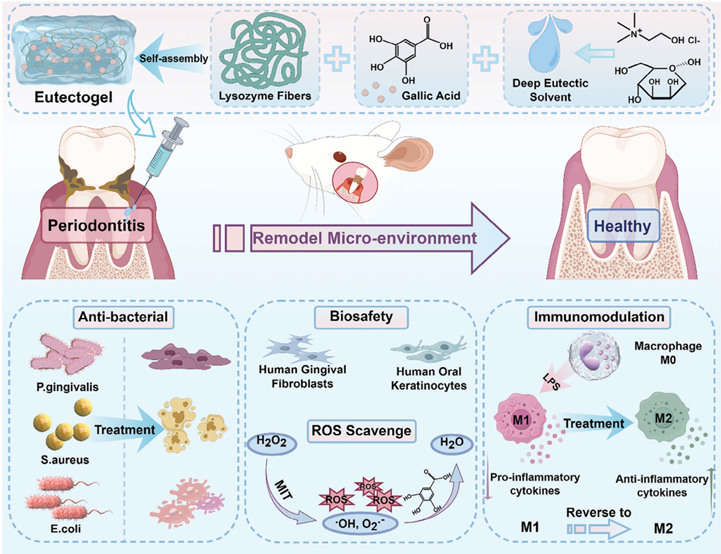
Self-Assembled Eutectogel with Cell Permeation and Multiple Anti-Inflammatory Abilities for Treating Chronic PeriodontitisIssuing time:2025-03-26 09:46 Ziying Cheng, Mengyan Kang, Xu Peng, Li Ren, Jing Xie, Quan Yuan, Xinyuan Xu, and Jianshu Lee Eutectogels represent an attractive option for various industrial applications that use deep eutectic solvents (DESs) as effective liquid active ingredients and offer remarkable stability, cost-effectiveness, and environmental friendliness. However, the biological applications of these compounds are limited. DESs are highly structurally tunable and exhibit remarkable biofunctionality and biocompatibility, conferring substantial benefits in the treatment of diseases. In this study, choline-chloride and mannose are used to fabricate ChCl/M DES, followed byintroduction of lysozymefibersandgallicacidforself-assembly into injectable eutectogels through hydrogen bonding and hydrophilic/hydrophobic interaction interactions. The eutectogels demonstrate almost 100% bactericidal activities against three strains and significant immune-regulation. This is supported by a decrease in the proportion of CD86-expressing cells from 64.02% to 18.17% and an increase in CD206-expressing cells from 2.53% to 29.96% through flow cytometry experiments. The eutectogels effectively inhibit alveolar bone loss and all eviated local inflammation in a rat model of chronic periodontitis owing to the promotionofgallic acid in thecell membrane by the ChCl/M DES. Hence, self-assembled eutectogels exhibit the potential to enhance the efficacy if treatments/therapies against inflammatory diseases by facilitating bacterial control, reactive oxygen species scavenging, and the regulation of macrophages by promoting cell permeation of small-molecule drugs. 1. Introduction Deep eutectic solvents (DESs) are composed of a minimum of one hydrogen-bond acceptor (HBA) and one hydrogen-bond donor (HBD), which exist predominantly in a eutectic mixture through hydrogen bonds when these compounds are mixed at specific molar ratios. Water is also used as a component in DES with the primary purpose of reducing the density and viscosity of DES. Researchers use hydration as a tool to overcome limitations of DESs, which will contribute to the development of advanced DESs as more environmentally friendly processing and reaction media. Over the past decades, DESs have received significant interest across diverse industries for example, materials synthesis, catalytic reactions, electrochemistry, biomaterial separation, and extraction, owing to their unique properties, such as exceptional thermal stability, modifiable structural characteristics, and high solubility for many solutes, along with facile preparation methods, economies of scale, and environmental sustainability. Therefore, the eutectogels containing DESs as their effective liquid active ingredients have been widely distributed in the fields of sensing, and interfacial electrical lubrication. conductivity, ditionally, AdDESs exhibit enhanced biodegradability 1. Introduction Deep eutectic solvents (DESs) are composed of a minimum of one hydrogen-bond acceptor (HBA) and one hydrogen-bond donor (HBD), which exist predominantly in a eutectic mixture and biocompatibility as well as a superior ability to accelerate the solubility and permeability of drugs, thereby improving the bioavailability of many pharmaceuticals. The intrinsic properties of DESs can potentially address the limitations with traditional hydrogels in terms of diffusivity and permeability, particularly for molecules of low water solubility. Therefore, the application of eutectogels containing DESs with biofunctions in inflammatory diseases is highly warranted, as their research potential has not been fully explored. Chronic periodontitis is a globally prevalent oral disease caused by the synergistic action of multiple pathogenic factors. Bacteria such as Porphyromonas gingivalis (P.g) is one of the primary factors contributing to excessive inflammation, leading to elevated local levels of reactive oxygen species (ROS) and overexpression of M1 macrophages. Modulation of the periodontal microenvironment through antibacterial action, ROS scavenging, and attenuation of M1 macrophage expression could serve as an efficacious therapeutic approach for the treatment of chronic periodontitis. Gallic acid (GA) is a small molecule with good antioxidant, free radical-scavenging, and anti-inflammatory properties; however, its hydrophilic and strong polarity present formidable obstacles to its transmembrane absorption. Enhancing the intracellular permeability of gallic acid is essential, and DESs offer an excellent solution. The natural sugar mannose is abundant in various fruits, and its primary function is the glycosylation of specific proteins. In recent years, mannose has emerged as a promising and effective agent for promoting immune tolerance and suppressing inflammatory diseases associated with autoimmunity and allergy. Gan et al. discovered that mannose-modified SiO2-NPs accelerated wound healing in diabetic mice by binding to mannose receptors on macrophages, triggering the activation of the ERK pathway and facilitating the transition from phenotypic M1 macrophages to M2. Therefore, mannose was chosen as the hydrogen bond donor (HBD), and choline chloride ([Ch]Cl) was selected as the hydrogen bond acceptor (HBA) because of its biodegradability, non-toxicity and cost-effectiveness. Additionally, the utilization of gallic acid in liquids leads to its rapid metabolism and high elimination rates in vivo. However, enhancing bioavailability while decreasing biotoxicity remains a significant challenge. Injectable gels are highly suitable for irregular damage and for enhanced localized drug retention. Lysozyme, which is primarily derived from egg whites, is an FDA approved alkaline protein with biocompatible and antibacterial properties. It achieves this by breaking down the 1, 4--linkages of peptidoglycan and chitodextrins present in bacterial cell wall. Amyloidogenic fibers are self-assembled ordered aggregates of relatively straight and unbranched nanostructures. Lysozyme fibers formed through heat-induced degeneration in an acidic solution show remarkable mechanical properties, including strength and strain at break. One article described a physically self-assembled hydrogel composed of lysozyme and gallic acid, which was formed through – interactions and hydrogen bonding. However, it was observed in this study that GA (60–90 mg mL?1) required heating at temperature exceeding 70 °C for more than 60 min to achieve complete dissolution. Moreover, hydrogels exhibit poor shear stability and bioadhesion, making it difficult to meet the diverse clinical demands. The introduction of ChCl/MDES and lysozyme fibers was significant for reducing the gel-forming temperature, simplifying gelforming procedures, facilitating gel preservation, and improving gel properties. In this study, an injectable eutectogel was fabricated via the self-assembly of ChCl/M DES (10 wt% in H2O), gallic acid 2412866 (2 of 17) www.advmat.de (GA), and lysozyme fibers (FLy), and its therapeutic efficacy was evaluated in a rat model of chronic periodontitis as shown in Scheme 1. First, eutectogels were prepared using a one-pot method, and their physicochemical characteristics, such as mechanical properties and adhesion, were evaluated. Second, the bactericidal activity and duration of action of the gels with different components were tested against P. gingivalis, the main pathogenic bacterium causing chronic periodontitis. Additionally, cytotoxicity toward human gingival fibroblasts (HGF) and normal human oral keratinocytes (HOK) was assessed. Furthermore, the ROS-scavenging ability and immune regulatory effects on macrophages were investigated in vitro. Finally, an experimental model involving rats with chronic periodontitis induced by the application of silk thread around teeth was used to evaluate the anti-inflammatory properties of the gels and their ability to reduce bone loss. Moreover, the distribution of the bacterial flora in the oral cavity of the rats was analyzed.
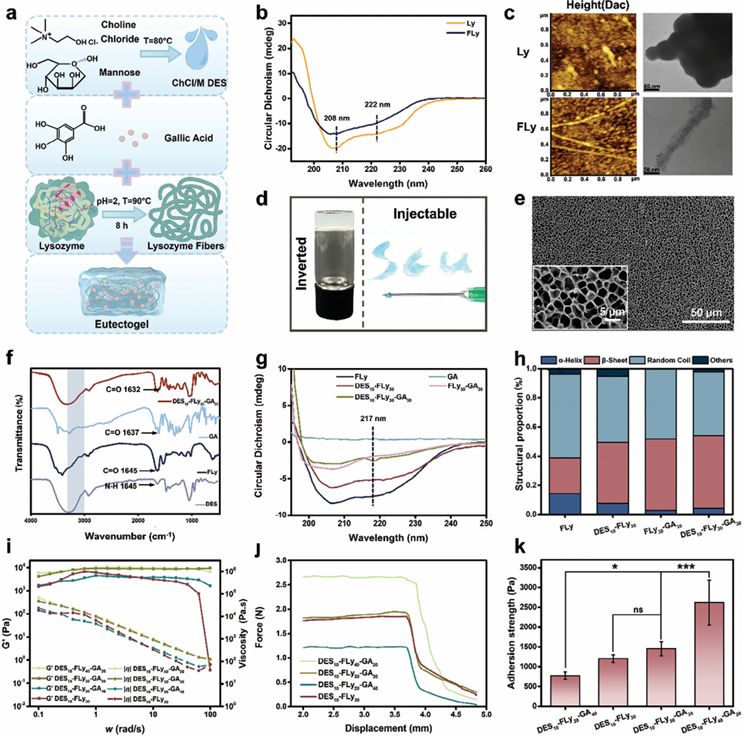
Scheme 1. Illustration of the self-assembly eutectogel and its therapeutic principle for chronic periodontitis (MIT: mitochondria). 2. Results and Discussion 2.1. Preparation and Characterizations of Eutectogels Eutectogels were fabricated through the self-assembly of lysozyme fibers(FLy), gallic acid (GA), and ChCl/MDES (10wt% in H2O) (Figure 1a). Egg lysozyme undergoes hydrolysis at low pH and high temperatures to form polypeptides, which are further assembled to form lysozyme fibers. FLy formation was initially confirmed using circular dichroism (CD) spectroscopy, atomic force microscopy (AFM), and transmission electron microscopy (TEM) (Figure 1b,c). Purified lysozyme protein (Ly) exhibit characteristic circular dichroism negative peaks of -helix peaks at 208 and222nm, whereas the -helical negative peaks of lysozyme fibers (Fly) show significant attenuation. According to calculations of the CDpro software, the secondary structure of Ly has a proportion of 0.252 -helices, 0.4 -folds with 0.386 randomclusters; the secondary structure of FLy has a proportion of 0.172 -helices, 0.512 -folds with 0.281 random clusters. This alteration in secondary structure is a distinctive feature of protein f ibrillation. The elongated fibrillar morphology observed in the AFM and TEM images further confirmed the formation of fibers. Incontrast, the crystalline Ly is characterized by a small-scale particle size ranging from0.1to0.3μminAFM,anditformsaspherical multimer composed of several clusters of round-like crystals with an approximate diameter of 50 nm in TEM. The chemical structures of Ly and FLy were further determined using ultraviolet spectrophotometry and Fourier-transform infrared spectrometry (Figure S1a,b, Supporting Information). UV absorbance at 240 and 280 nm was observed for both Ly and FLy, indicating the predominant absorption of phenylalanine (Phe), tyrosine (Tyr), and tryptophan (Trp) residues. It was demonstrated that the f iber assembly process of FLy did not alter the amino acid composition of lysozyme. The FLy absorption peak of the amide I bandat 1652cm?1 isslightly shifted to the right and has a significant growth compared to Ly, which indicates an increase in the folding content of the characterization. Collectively, these findings suggest that lysozymeproteins undergo fibrillar transformation under high temperature and acidic conditions. TheChCl/MDES, a clear and transparent viscous liquid, were obtained by stirring a mixture of choline chloride and mannose at a constant temperature of 80 °C for 2h. The formation mechanism was analyzed using Fourier transform infrared spectroscopy (FT-IR) (Figure S1c, Supporting Information). During the ChCl/M DES formation process, the telescopic vibrational absorption peak corresponding to-OH groups in ChCl/M DES is redshifted to lower wavenumbers within the range of 30003500 cm?1, suggesting the formation of amounts of hydrogen bonds between the HBA [Ch]Cl and HBD Mannose. The formation of hydrogen bonds reduces the lattice energy of [Ch]Cl, and the melting point of the [Ch]Cl and mannose mixture also decreases. The Fly, GA and ChCl/M DES mixture was sonicated at 40 °C to generate a homogeneous solution, which was subsequently cooled to room temperature resulting in the formation of an opaque and non-flowing eutectogel (Figure 1d). The formulations and nomenclature of the two- and three-component eutectogels are showninTableS1,Supporting Information.Asshown, the cryo-SEM images in Figure 1e revealed that the DES10-FLy30- GA30- gels exhibits a consistent pore morphology, characterized by uniform pores ranging from 3 to 6 μm in size. The FT-IR spectra in Figure 1f and Figure S1d, Supporting Information demonstrate a reduction in the intensity of the absorption peak corresponding to–OH in the DES10-FLy30-GA30 gels, along with a tendency for the peak to shift toward lower-frequency bands compared to FLy and GA. Conversely, the C═O peaks of FLy at 1645 cm?1 andgallic acid (GA) at 1637 cm?1 shifted to 1632 cm?1 upon gel formation, indicating the presence of hydrogen bonds during the eutectogel assembly. To study the secondary structure of FLy after eutectogel formation, the circular dichroism (CD) spectra of FLy, each two-component solution, and the DES10-FLy30-GA30 eutectogel were measured and compared. The DES10-FLy30-GA30 gels, as depicted in Figure 1g, exhibit a negative peak at 217–218 nm and a strong positive peak at 195198 nm compared to the other groups, indicating a significant enhancement of the -sheet conformation. This observation is consistent with the quantitative calculations performed using CDpro shown in Figure 1h. Eutectogel preparation was confirmed from the aforementioned data, and the potential mechanism of gel formation was analyzed as follows. This eutectogel is constituted with peptides to form hydrogelated -sheet peptides (HPs). Self-assembly of HPs is driven by the interaction between hydrophobic and hydrophilic residues of ChCl/M DES. Hydrophobic groups avoid the solvent medium, whereas complementary polar residues interact with water molecules, stabilizing peptide assemblies through hydrogen bond formation and other intermolecular interactions. The conformational stability of the ChCl/M DES for FLy was precisely achieved through the effective entanglement of FLy in the solvent, which enhanced the strength of hydrogen bonding. Furthermore, the rheological properties of the gels were assessed using shear rheology experiments, and the mechanical properties of the gels with varying concentrations of FLy and GA were examined to ascertain the influence of the components (Figures 1i). The viscosity (||) of the gel gradually decreases with increasing , indicating that the gels exhibit shear thinning behavior. Additionally, as the FLy content increases, both G′ and || of the gel also increase, further confirming that FLy provides network entanglement support for the eutectogels. The injection force test conducted using a texture analyzer demonstrated that all forces measured were below 3N, which is advantageous for clinical use. The injection force increased proportionally with FLy content, similar to the findings of the rheological examination (Figure 1j and Figure S2a, Supporting Information). The adhesion of the eutectogel to the tissues was evaluated, as shown in Figure 1k and Figure S2b, Supporting Information. Eutectogels have a certain adhesion strength to biological tissues, which enables them to effectively adhere and facilitate their pharmacological effects. The adhesion strength increased with increasing FLy content, which was attributed to the synergistic effect of a range of electrostatic interactions, hydrogen bonding, and surface hydrophobic interactions occurring between various functional groups (-NH2,-COOH,-OH,-SH,-(CH)n, and-CH3) on the surfaces of eutectogels and biological tissues.
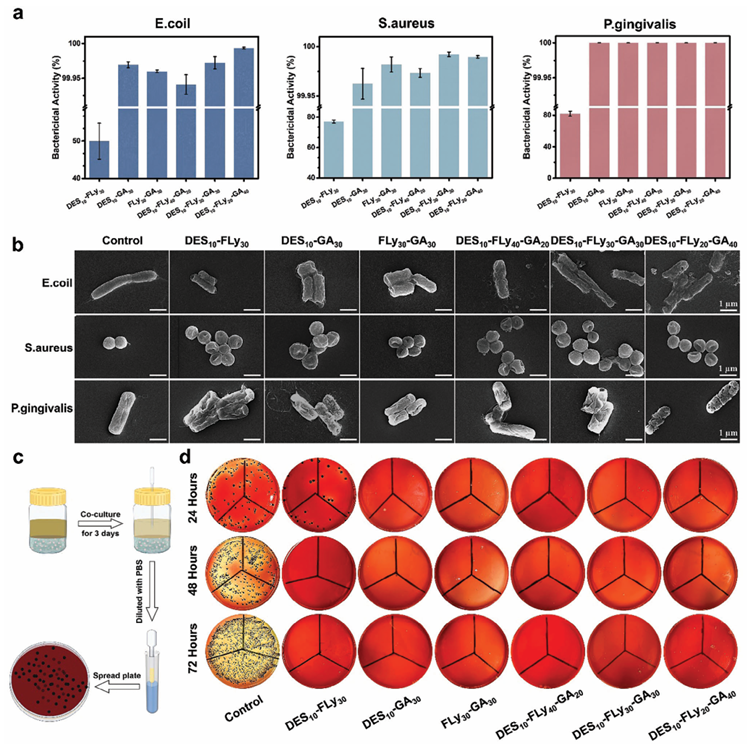
Figure 1. a) Schematic illustration of the formation of a eutectogel. b) Circular dichroism images of lysozyme proteins (Ly) and lysozyme fibers (FLy). c) AFM (Height) and TEM images of LyandFLy. d) The optical photo of eutectogel. e) Cyro-SEM images with a poresize of about 3–6 μm of the eutectogel (DES10-FLy30-GA30). f) FTIR spectra of ChCl/M DES, FLy, GA and eutectogel (DES10-FLy30-GA30). g) Circular dichroism images and h) Quantitative data for CDpro of FLy, DES10-FLy30,DES10-GA30,FLy30-GA30,DES10-FLy30-GA30. i) Rheological testing of eutectogels with G’ and || following velocity changes. j) Injection force test for eutectogels. k) Adhesion test of eutectogels to pig skin. 2.2. Antibacterial Properties of Eutectogels The eutectogels were evaluated for their broad-spectrum antimicrobial properties using the plate spreading method with various strains of bacteria, including Escherichia coli (E. coli), Staphylococcus aureus (S. aureus) and Porphyromonas gingivalis (P. gingivalis). Gels containing GA exhibited ≈100% antimicrobial efficacy. The DES10-Fly30 group without GA exhibited less than 50% bactericidal activity against E. coli and ≈80% bactericidal activity against S. aureus and P. gingivalis (Figure 2a). Although FLy demonstrates some antibacterial properties, it fails to meet clinical antimicrobial requirements. The SEM images presented in Figure 2b illustrate the cellular membrane morphology of the bacteria in the various treatment groups. The cell membranes of normal cultured bacteria exhibit a rounded appearance and are devoid of cracks or cellular debris. In contrast, bacterial cell membranes treated with GA-containing groups displayed a notable increase in the number of cracks, accompanied by extensive membrane fusion, agglomeration, disintegration, and air leakage. Cell fragmentation and lysis are also observed. Conversely, the bacterial cells treated with the DES10-FLy30 group display a wrinkled appearance but no signs of cell membrane perforation. GA can alter the membrane permeability of bacteria, disrupt membrane adhesion, cause membrane perforation, and cause irreversible changes in the hydrophobicity and physicochemical properties, thereby inhibiting bacterial growth and leading to bacterial death. The triple-component eutectogels exhibited satisfactory results in the 24-h antibacterial trials, prompting us to conduct a 3-day long-lasting antibacterial experiment against P. gingivalis, the primary etiological agent of periodontitis. The experimental procedure is shown in Figure2c.The untreated group exhibited a remarkable proliferation rate of P. gingivalis, simulating the initial stages of periodontitis characterized by bacterial colonization of the periodontal pockets and alveolar bone. Following drug application, no colonies were observed in any of the groups containing GA over the course of 3 days, indicating the excellent long-lasting antibacterial properties of the eutectogel groups (Figure2d). Blackcolonies appeared on the first day in the DES10-FLy30 group, but no colonies were detected on the second and third days. This can be attributed to the bactericidal mechanism of FLy, which causes cell membrane disintegration through a synergistic combination of positive charge and increased hydrophobic -sheet. However, when dealing with P. gingivalis possessing an outer membrane, the bactericidal efficacy of FLy needed a long exposure duration. The long-lasting antibacterial properties were attributed to the release of GA in the solution environment, with the release ratio reaching up to 80% by the third day, according to the UV absorbance test (Figure S3,Supporting Information). To further validate the bactericidal properties of the different groups, the gel-to-bacterial ratio was decreased (Figure S4, Supporting Information). Subsequent analysis revealed that the DES10-FLy40-GA20 group only achieve a bactericidal activity of only 60%, rendering it unsuitable for further applications. 2.3. Cytotoxicity, ROS Scavenging and Immunomodulation of Eutectogels 2.3.1. Cytotoxicity of Eutectogels The cytotoxicity of the eutectogels on human gingival fibroblasts (HGF)andhuman oral keratinocytes (HOK) was evaluated over a 7-day period using a cell counting kit-8 (CCK-8) assay, live/dead staining, and morphological staining (Figures S5 and S6,Supporting Information). Cell viability, measured by the CCK-8 assay, showed no significant difference from the control group in all groups on the first day. Most groups exhibited an unexpected rate of cell proliferation on the first day, except for DES10-FLy20GA40. The relative viability of cell proliferation after 7 d was detected using the CCK-8 assay. Except for the DES10-FLy20-GA40 and DES10-GA30 groups, all the other groups exhibited cell proliferation rates that were not lower than those of the control group for both cell types. In contrast, the HGF proliferation ratio of DES10-FLy20-GA40 on the seventh day was only 50% that of the control group, whereas the HOK proliferation ratio in this group was only 20% that of the control group. It is hypothesized that this is due to the formation of hydrogen bonds between the hydroxyl groups in GA and FLy, thereby reducing the potential of GA to form hydrogen bonds with phospholipids in cell membrane liposomes, which ultimately results in a reduction in GA cytotoxicity. However, when the concentration of GA was 40 mg mL?1, hydrogen bonding between GA and the cellular liposomes was sufficient to cause cell membrane rupture and cell death. In the fluorescence images, HGF and HOK cells incubated with the DES10-FLy20-GA40 groups how significantly lower cell survival and fewer live cells (green fluorescent dots). In contrast, cells treated in the other groups retained their spread growth morphology and hadanextremely high cell survival ratio. Due to the relatively high cytotoxicity of the DES10-FLy20-GA40 group, this group was excluded from subsequent experiments. The DES10-FLy30GA30 group was selected for further studies owing to its satisfactory cyto compatibility and previously demonstrated bactericidal properties.
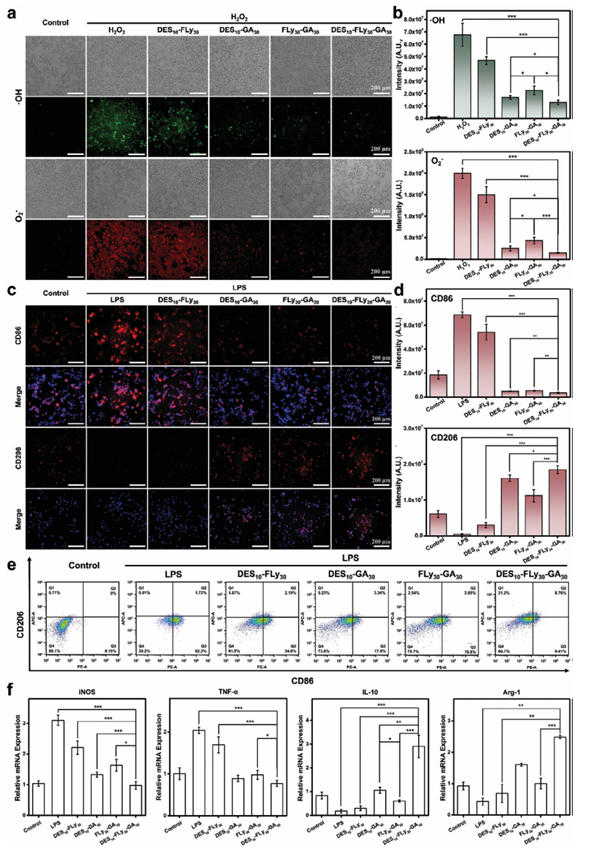
Figure 2. a) Bactericidal activity of eutectogels after co-culture with 106 CFU mL?1 of bacterial fluids for 24 h at a volume ratio of 1:1. b) SEM images of bacteria after co-culturing with different samples. c) Schematic diagram of the test of eutectogels co-cultured with 106 CFU mL?1 P. gingivalis at a 1:1 volume ratio for 72 h. d) Photographs of agar plates at 24, 48, and 72 h for this test. 2.3.2. ROS Scavenging of Eutectogels Excessive accumulation of reactive oxygen species (ROS) produced by the mitochondria (MIT) leads to oxidative stress, which contributes to chronic inflammation. Hence, efficient ROS scavenging is important for the treatment of inflammatory diseases. A 1,1-diphenyl-2-trinitrohydrazine (DPPH) freeradical scavenging assay demonstrated that the DPPH scavenging efficiency is influenced by the concentration of GA. The DPPH scavenging rate of DES10-FLy40-GA20 reached 70% and the DPPH scavenging ratio of DES10-FLy30-GA30 with a higher GA concentration, exceeded 80%, indicating its exceptional ability to scavenge free radicals (Figure S7, Supporting Information). Treatment of macrophages with H2O2 induces the generation of excessive intracellular ROS, which can serve as a model for evaluating the intracellular ROS-scavenging capacity of eutectogels. The intracellular ROS levels are assessed using a dichlorodihydro-fluorescein diacetate (DCFH-DA) probe, which exhibits green fluorescence upon labelling ·OH, and a dihydroethidium (DHE) probe, which emits red fluorescence upon labelling O2 ?. As shown in Figure 3a, the H2O2 group without treatment showed robust·OH green fluorescence and O2 ? red fluorescence, providing evidence for the induction of excessive ROS production by macrophages upon H2O2 stimulation, whereas the control group without H2O2 stimulation displayed minimal fluorescence. Compared to the H2O2 group, the number of fluorescent dots for ·OH and O2 ? in macrophages stimulated by H2O2 was significantly reduced following GA treatment. Quantitative data revealed that theDES10-FLy30-GA30 eutectogels exhibited remarkable efficacy in reducing the fluorescence intensity of ·OH from 6.75 × 107 in the H2O2 group to 1.29 × 107, as well as that of O2 ? from 2.00 × 108 to 0.15 × 108, representing the highest efficiency in ROS scavenging (Figure 3b). Conversely, in the absence of GA, only a marginal decline in the fluorescence intensity was observed in the DES10-FLy30 group compared to that in the H2O2 group. The antioxidant activity of GA has been demonstrated in numerous studies and can be attributed to the presence of three phenolic hydroxyl groups in its chemical structure. Specifically, the p-OH group is recognized for its pivotal role in facilitating free radical scavenging. Several reaction enthalpy studies have determined that GA undergoes a hydrogen-atom transfer (HAT) mechanism to effectively scavenge ROS in solvent media. The DES10-GA30 solution and DES10-FLy30-GA30 eutectogel groups, both incorporating ChCl/M DES as a solvent, exhibited significantly enhanced ROS scavenging efficacy compared to FLy30-GA30 with water as a solvent. It is hypothesized that the enhanced cell membrane permeability induced by the ChCl/M DESresults in increased GA bioavailability and antioxidant effects. The disparity in the intensity of both ·OHandO2 ? between DES10-FLy30-GA30 and DES10-GA30 can be attributed to the presence of FLy. FLy acts as a scaffold for the eutectogels, ensuring their structural integrity and preventing their complete hydrolysis in solution. Hence, DES10-FLy30-GA30 eutectogels exhibited an effective scavenging capacity for both ·OH and O2 ?, thereby inhibiting uncontrolled oxidative stress and laying the foundation for the treatment of chronic inflammatory sites.
Figure 3. a) Bright field and fluorescent images of H2O2-stimulated Raw 264.7 macrophagesco-cultured with different samples; the green fluorescence of DCFH-DA staining indicates ·OH in the cells, and the red fluorescence of DHE indicates O2? in the cells. b) Quantitative data of scavenge intracellular ROS of different samples calculated using ImageJ. c) The fluorescence images of LPS-stimulated Raw 264.7 macrophages co-cultured with different samples; the blue fluorescence of DAPI staining indicates nuclei, and red fluorescence indicates CD86/CD206 in the cells. d) Quantitative data of CD86 and CD206 of different samples calculated using ImageJ. e) Flow cytometry results of macrophages expressing CD86 and CD206 after treatment with different groups. The antibodies of CD86 (red, PE channel) and CD206 (green, APC channel) were employed to specifically label M1-like macrophages and M2-like macrophages, respectively. f) The qPCR results of inflammation-related cytokines and anti-inflammation-related cytokines of Raw264.7 cells treated with different groups. 2.3.3. Immunomodulation of Eutectogels Among the various immune cells involved in immunomodulation, macrophages have been shown to play a pivotal role in chronic inflammation. Macrophages polarize into different phenotypes in response to their surrounding microenvironment. In the context of infection, macrophages initially polarize toward the pro-inflammatory M1 phenotype, which releases pro-inflammatory cytokines to facilitate antigen and necrotic cell clearance. During the repair phase, M1 macrophages undergo polarization to become M2 macrophages, which secrete anti-inflammatory cytokines to suppress inf lammation and promote tissue repair and remodeling. LPS-stimulated macrophages were utilized to analyze the in vitro phenotypic shift effects of samples by immunofluorescence staining and quantification of CD86 M1-type and CD206 M2-type markers, while simultaneously confirming the cellular content through flow cytometry (Figure 3c,d,e). Following LPS stimulation, macrophages exhibited a significant fluorescence intensity of CD86 (M1Φ maker) staining and minimal CD206 (M2Φ maker) fluorescence in the LPS group. Additionally, flow cytometry analysis revealed that the proportion of cells labeled with CD86 reached 64.02%, whereas only 2.53% of the cells were labeled with CD206, indicating a predominantly M1-type pro-inflammatory phenotype following LPS stimulation. After treatment with GA-containing groups, the fluorescence intensity of CD86 in the macrophages was significantly reduced, whereas the fluorescence intensity of CD206 increased. Flow cytometry analysis revealed an ≈65% decrease in the proportion of CD86positive macrophages in the GA-containing groups compared to that in the LPS-stimulated group. The efficacy of GA in promoting the M1-M2 transition in macrophages by scavenging ROS and inhibiting NF-B activation has been demonstrated, thereby offering potential benefits in attenuating the inflammatory response associated with chronic periodontitis. The introduction of ChCl/M DES resulted in the DES10-FLy30-GA30 eutectogel exhibiting a higher fluorescence intensity and a larger proportion of CD206-labeled macrophages than FLy30-GA30. This phenomenon can be attributed to two factors. First the ChCl/M DES enhanced GA penetration across cell membranes and increased the efficiency of GA entering the cell interior, thus exerting its immunomodulatory effects. Second, CD206, known as the mannose receptor, is a transmembrane protein expressed on macrophages with an intracellular antibody-binding site that can be activated by mannose within the ChCl/M DES. The discrepancy in the proportion of CD206 cells between the DES10FLy30-GA30 andDES10-GA30 groups can be attributed to the target and aggregation effects of FLy. The DES10-FLy30-GA30 eutectogel was anchored around the cells, so that the local concentration of GA around the cells was high, and the osmotic pressure generated by the concentration gradient allowed GA to easily pass through the cell membrane with increased permeability into the cell interior. Additionally, some immunomodulatory effects were observed in the DES10-FLy30 group owing to the modulatory effect of mannose on inflammation. The immunoregulatory capacity of different samples was further evaluated using real-time polymerase chain reaction (RTPCR) to quantify the concentration of the extracellular secreted product region (Figure 3f). M1 phenotype macrophages are capable of secreting a variety of pro-inflammatory cytokines, including TNF-,iNOS,IL-6,andIL-1.These cytokines arebelieved to play crucial roles in promoting inflammatory responses. In contrast, M2 macrophages possess the ability to suppress inflammation and facilitate tissue repair by producing anti-inflammatory cytokines, such as IL-10 and Arg-1. The relative mRNA expression of iNOS and TNF- was significantly upregulated in the LPS-stimulated group, whereas a decrease was observed in the groups treated with GA-containing compounds. Compared to DES10-GA30 and FLy30-GA30 groups, the expression levels of iNOS and TNF- cytokines were further lower in DES10-FLy30GA30 group.Furthermore, treatment with the eutectogel resulted in significant upregulation of both IL-10 and Arg-1 expression compared to that in the LPS-stimulated groups, indicating an increase in M2 phenotypic macrophages. These results suggested that the DES10-FLy30-GA30 eutectogel exhibited remarkable efficacy in modulating the phenotypic transition of macrophages from M1 to M2, thereby effectively suppressing the expression of pro-inflammatory factors and enhancing the expression of anti-inflammatory factors. Consequently, it can be concluded that the DES10-FLy30-GA30 eutectogel possesses a favorable immunomodulatory effect, which is crucial for treating chronic periodontitis.
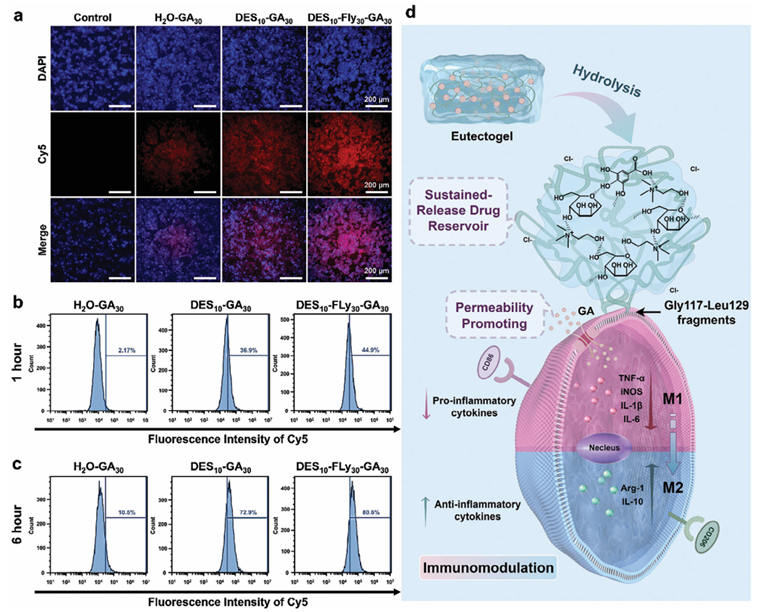
Figure 4. a)The fluorescence images of Raw 264.7 macrophages cells co-cultured with different samples, the blue fluorescence of DAPI staining indicates nuclei, and red fluorescence indicates cy5-GA in the cells. Flow cytometry results of macrophages containing cy5-GA after treatment with different groups of samples for b) 1 h and c) 6 h. d) Schematic illustration of effective cellular internalization of GA by eutectogels containing FLy and ChCl/M DES for immunomodulation. 2.3.4. Intracellular Uptake of the Gallic Acid The ROS scavenging efficacy, proportion of cells labeled with CD206 markers, and expression levels of Arg-1 and IL-10 cytokines collectively indicated that the DES10-FLy30-GA30 group exhibited superior performance compared to the two-component sample, despite having the same concentration of GA, whereas the FLy30-GA30 combination was found to be inferior to the DES10-GA30 group.Thecy5-GA was utilized to fabricate materials for investigating the intracellular uptake of GA in macrophages using fluorescence microscopy and flow cytometry to validate the potential mechanism underlying the enhanced antiinflammatory properties of the eutectogel. The fluorescence intensity of Cy5 in H2O-GA30 when incubated with macrophages for 1 h as depicted in Figure 4a, was significantly lower than that in the other two experimental groups with ChCl/M DES as the solvent. Flow cytometry analysis in Figure 4b revealed that after a 1-h incubation, the percentage of cells containing cy5GA in H2O-GA30 was only 2.17%. In contrast, the DES10-GA30 groupexhibited a significantly higher proportion of cells containing cy5-GA (36.9%) compared to the aqueous group. Remarkably, the presence of FLy increased the number of cells expressing cy5-GA by 44.9%. As depicted in Figure 4c, the macrophage uptake of cy5-GA was time-dependent. After a 6-h incubation period, the H2O-GA30 group showed an increase in cells containing GA to 10.5%, while this proportion rose substantially to 72.9% and 80.6% in the DES10-GA30 and DES10-FLy30-GA30 groups, respectively. This provided compelling evidence that the presence of ChCl/M DES and FLy facilitated the efficient internalization of GA by macrophages (Figure 4d). Osmotic pressure is generated by differences in chemical potential of ions and molecules between the cytoplasm and extracellular fluid. Difference in solute concentration is one of the sources of osmotic pressure gradients. As a small molecule drug, GA can take advantage of concentration-induced osmotic pressure effects for intra- and extracellular diffusion. In aqueous solutions, the ChCl/M DES may undergo partial dissociation, leading to the release of cations (choline), anions (Cl?), and mannose (HBD) for hydrogen bonding and electrostatic interactions with the cell membrane, resulting in partial distortion of the cell membrane. Partial damage to the plasma membrane leads to increased cell membrane permeability, thereby facilitating the transport of GA across the cell membrane is facilitated. Therefore, compared to H2OGA30, DES10-GA30 and DES10-FLy30-GA30 with ChCl/M DES as the solvent showed a higher efficiency of cellular internalization of GA. The disparity in the GA endocytosis efficiency between DES10-FLy30-GA30 and DES10-GA30 could be attributed to the target and aggregation effects of FLy. FLy is charged positively at physiological pH, allowing it to be attached to the anionic membrane surface of macrophages. The unfolding and reorganization of the Fly polypeptide chain exposes additional disulfide bonds, which then bind to sulfhydryl groups on cell membrane proteins. The Gly117-Leu129 fragments of FLy anchor it to the lipid matrix in the cell membrane, while its core is located on the outer surface. Consequently, the eutectogels with FLy are immobilized outside the cell and function as a sustained-release drug reservoir. Due to the presentation of eutectogel, the local concentration of GA outside the cell could be higher than that in the overall fluid environment, causing high osmotic pressure. However, the hydrogen bonding between the phenolic hydroxyl of GA and carboxyl groups of ChCl/MDES was completely dissociated due to the absence of a stable aggregate in DES10-GA30, resulting in the dispersion of GA throughout the solution environment. The concentration gradient of GA between the intra- and extracellular fluid is minimal causing low chemical potential and osmotic pressure. Therefore, despite the fact that ChCl/M DES causes partial distortion of the cell membrane and enhances cell permeability, a proportion of GA is unable to penetrate cells because of the small osmotic pressure exerted in DES10-GA30. In conclusion, the combined effect of ChCl/M DES's cell membrane permeability and the highly localized extracellular concentration of GA synergistically enhances GA endocytosis efficiency in DES10-FLy30-GA30-.
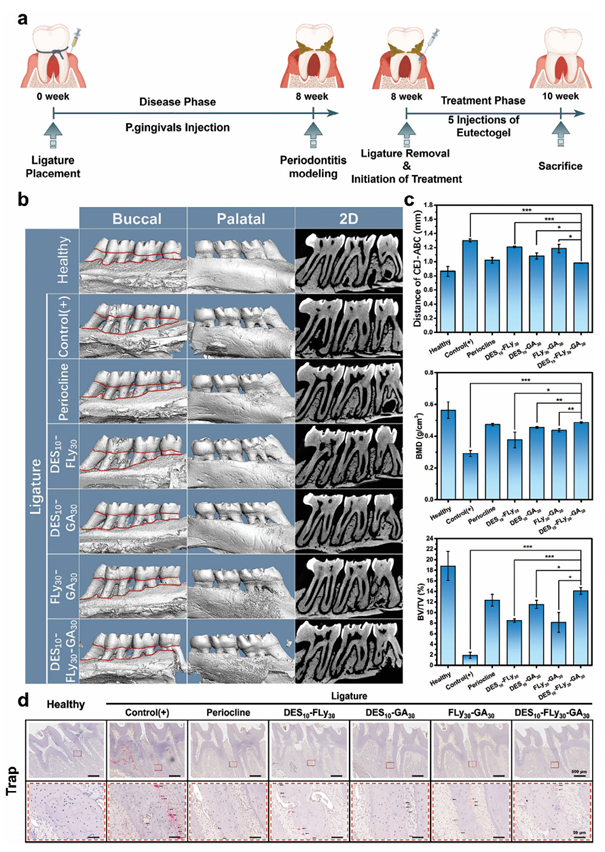
Figure 5. a) Schematic representation of the comprehensive timeline for the treatment of periodontal tissues in SD rats. b) Assessment of alveolar bone loss from micro-CT results. Representative 2D sagittal X-ray images and 3D-constructed images viewed from the buccal and palatal sides of the maxillary alveolar bone after 2 weeks of treatment. c) Statistical analysis of the average linear distance of CEJ-ABC, bone mineral density (BMD) and bone volume/total volume (BV/TV). d) Histological examination was performed using tartrate-resistant acid phosphatase (TRAP) staining of the interdental areas around the upper first and second molars. Black arrows indicate TRAP-positive (stained red) cells. 2.4. Treatment of Chronic Periodontitis 2.4.1. Inhibition of Alveolar Bone Resorption A Sprague-Dawley (SD) rat model of periodontitis was established by ligating the maxillary first molar for eight consecutive weeks and injecting P.gingivalis fluids. Subsequently, the ligature wire was removed and various samples were injected into the periodontal pockets. Following a 2-week treatment period, euthanasia was performed on the rats were euthanized for subsequent analysis. The overall timeline of animal experiments is shown in Figure 5a. Representative morphological variations in the alveolar bone were assessed using 2D and 3D-constructed images obtained by micro-CT (Figure 5b). Bone volume data, including the average distance from the cemento-enamel junction to the alveolar bone crest (average distance of CEJ-ABC), bone mineral density (BMD), and bone volume/total volume (BV/TV), were acquired to analyze the extent of alveolar bone loss (ABL), as depicted in Figure 5c.Thecontrol(+) group (ligated without treatment) exhibited significant bone loss on the buccal and palatal aspects of the first molar (M1) and second molar (M2). The sample exhibited the largest CEJ-ABC distance (1298 ± 21.42 μm), the lowest BMD value (0.292 ± 0.019 g cm?3), and the lowest BV/TV value (1.896±0.561%), indicating successful induction of chronic periodontitis. Additionally, the mean CEJ-ABC distance in the healthy group is ≈866 ± 57.5 μm, which significantly surpasses the diameter of the ligated wire (200 μm), thereby suggesting that alveolar bone loss cannot be attributed to mechanical damage resulting from wire insertion. The eutectogel DES10FLy30-GA30 demonstrated the most favorable outcomes after a 2-week treatment period. In the eutectogel group, the CEJ-ABC distance was 75.5% of that in the periodontitis group, indicating a notable improvement in alveolar bone resorption. Additionally, the BMD value was 166.44% and the BV/TV value was 743.20% of those in the periodontitis group, demonstrating a considerable increase in bone mineral density and bone volume. The therapeutic efficacy of the eutectogel group was better than that of the commercially available periodontitis drug Periocline. The extent of bone loss in the DES10-FLy30 group was significantly more severe than that in the other experimental groups, indicating that this group had no beneficial effect on preventing bone resorption or in vivo anti-periodontitis effects. It can be inferred that GA is an essential component for the effective management of periodontitis. The therapeutic efficacy of FLy30-GA30 was significantly lower than those of DES10-FLy30 GA30 and DES10-GA30, consistent with the results obtained from the aforementioned in vitro experiments. This discrepancy can be attributed to the enhanced permeability of ChCl/M DES in GA. Furthermore, DES10-GA30 exhibited inferior therapeutic eff icacy compared to that of DES10-FLy30-GA30. Apart from its diminished impact on macrophage M2 phenotype transformation, as previously mentioned, DES10-GA30 failed to maintain sufficient retention in the gingival tissues for the required 72-h period, thereby compromising its intended drug retardation and sustained bactericidal and anti-inflammatory activity. These findings indicated that the eutectogel DES10-FLy30-GA30 exhibited the most satisfactory inhibition of bone resorption in chronic periodontitis. The pathogenesis of bone loss is characterized by an imbalance between osteoblastic bone formation and osteoclastic bone resorption, necessitating the modulation of osteoclast activity in the treatment of bone diseases. Figure 5d shows the evaluation of osteoclast activity and alveolar bone resorption by tartrateresistant acid phosphatase (TRAP) staining, revealing the presence of numerous TRAP-positive cells (stained red, indicated by black arrows) that formed resorption cavities along the surface of the alveolar bone surrounding the intermolar region of the upper f irst and second molars in the control(+) group. Both the Periocline and DES10-FLy30-GA30 groups exhibited a near-complete absence of osteoclasts, which corroborated the findings of micro-CT analysis. These results demonstrate that the DES10-FLy30-GA30 eutectogel has the potential to attenuate osteoclast activity and inhibit bone resorption caused by periodontal inflammation, thereby providing an effective approach for managing chronic periodontitis.
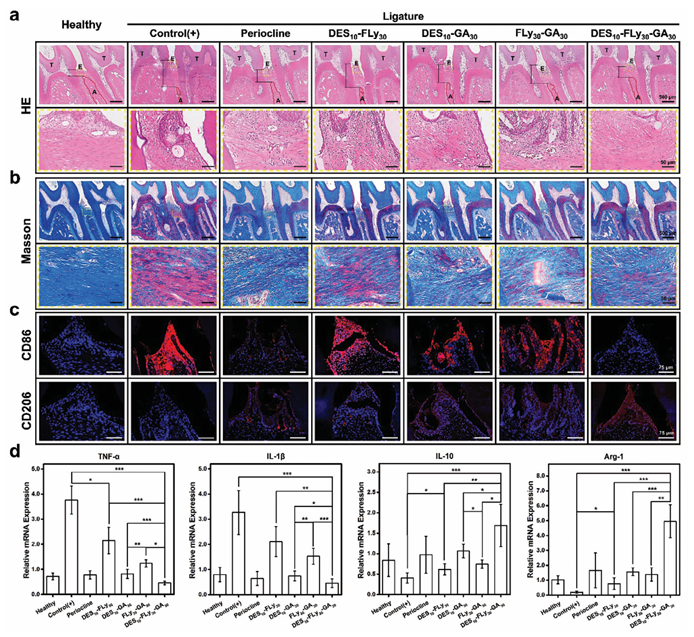
Figure 6. Histological examination using a) H&E staining and b) MASSON staining of the interdental area around the upper first and second molars. Black arrows in (a) indicate cross-section CEJ-ABC distances. c) Immunofluorescence images of gingival tissue between the first and second molars in rats, DAPI-stained blue fluorescence indicates nuclei and red fluorescence indicates CD86/CD206 in cells. d) The qPCR results of inflammation-related cytokines and anti-inflammation-related cytokines of periodontal tissues treated with eutectogels. 2.4.2. Anti-Inflammatory Action In Vivo The H&E-stained images in Figure 6a show that the control(+) group exhibited severe bone loss, severe alveolar breakdown, and irregular bone crest surfaces, as evidenced by the cross-sectional CEJ-ABC distances (black arrows). The cross-sectional CEJ-ABC distance was shorter than that of the periocline group, yet greater than that of the healthy group for the DES10-FLy30-GA30 eutectogel group. Furthermore, the cross-sectional CEJ-ABC distance in the experimental group treated with GA was smaller than that in the DES10-FLy30 group, suggesting the potential efficacy of GA in treating periodontitis. This observation is consistent with previous findings from micro-computed tomography and quantitative bone data. Hematoxylin and eosin (H&E)-stained images of healthy periodontal tissues demonstrated a regular-thickness epithelial layer, dense connective tissue, organized arrangement of collagen fibers, and smooth bone crest surfaces. The control(+) group (no treatment following ligation) exhibited evident thickening and structural disarray of the epithelial layer along with notable infiltration of inflammatory cells, including purple-stained eosinophils and red-stained neutrophils, indicating the onset of severe periodontal inflammation. Compared with the control(+) group, the DES10-FLy30-GA30 group showed a notable reduction in inflammatory cell in filtration,mild tissue destruction, reduced alveolar protrusion erosion, and a significant improvement in periodontitis; inflammatory progression was significantly slower than that in the healthy group. Gingival epithelial tissue also showed a significant reduction in the number of inflammatory cells following periocline treatment. The other experimental groups also showed some therapeutic effects, with a reduction in inflammatory cell infiltration of the tissues; however, their therapeutic effects were poorer than those of the DES10-FLy30-GA30 and periocline groups. Masson's trichrome staining as shown in Figure 6b, revealed degeneration of collagen fibers and a large number of disorganized and sparsely arranged muscle fibers (stained red) in the control(+) group. This observation served to quantify the severity of the inflammatory response in the CP microenvironment of chronic periodontitis. Following treatment withDES10-FLy30-GA30 eutectogel orperiocline, the numberofirregular muscle fibers was significantly reduced. Simultaneously, dense and well-aligned collagen fibers (stained blue) appeared, indicating optimal periodontal tissue healing and diminished inflammation. Furthermore, treatment with DES10-GA30 and FLy30-GA30 improved collagen fiber density and the regularity of gingival tissues, highlighting the pivotal role of GA in managing inflammatory processes. However, the DES10-FLy30 group exhibited inadequate treatment outcomes, as evidenced by the persistently degenerated collagen fibers, suggesting that mannose alone lacks sufficient anti-inflammatory capacity to treat chronic periodontitis. The macrophages in vivo in the periodontal tissues between the first and second molars were immunostained using CD86 and CD206 markers (Figure 6c). The fluorescence intensity of CD86 (red) was markedly elevated in the periodontal tissues of rats in the control(+) group, whereas that of CD206 (red) was nearly undetectable. These findings indicated that in the gingival tissues of rats with periodontitis, macrophages predominantly exhibit a pro-inflammatory M1 phenotype. The red fluorescence of CD86 was nearly absent in the DES10-FLy30-GA30 eutectogel group, indicating its highly effective anti-inflammatory properties. In contrast, the DES10-FLy30-GA30 group exhibited the highest intensity of CD206 fluorescence, suggesting its superior ability to induce macrophage polarization from the M1 to M2 phenotype in vivo. The outstanding permeability achieved by the ChCl/M DES is responsible for this phenomenon. It is noteworthy that the periocline exhibits a diminished fluorescence intensity of CD86, but lacks the expression of CD206, indicating that it solely targets inflammation associated with the M1 phenotype without inducing further immune modulation toward the M2 phenotype. The presence of GA in the DES10-GA30 and FLy30GA30 groups lead to a decreased number of cells exhibiting the protein CD86. However, the decrease in the proportion of M1 phenotype macrophages was not as effective as that of DES10FLy30-GA30 because of inadequate tissue retention in their liquid state. The fluorescence intensity of CD86 remained largely unaltered in the DES10-FLy30 group, indicating a limited anti- inflammatory effect in the absence of GA.These findings are consistent with the results of the immunofluorescence staining for LPS-induced inflammation in vitro. The levels of inflammatory factors in the gingival tissues surrounding the rat molars were further quantified using qPCR (Figure 6d and Figure S8, Supporting Information). In the control (+) group, there was an upsurge in the expression of pro-inflammatory factors, including TNF-, iNOS, IL-1,andIL6, as well as minimal expression of M2 anti-inflammatory cytokines, IL-10 and Arg-1. Conversely, the DES10-Fly30-GA30 eutectogel group demonstrated a marked reduction in M1 proinf lammatory cytokine expression and elevated levels of M2 antiinflammatory cytokine expression. In addition to H&E, MASSON, and immunofluorescence staining, the DES10-Fly30-GA30 eutectogel showed excellent immunomodulatory effects and can effectively treat chronic periodontitis. Periocline exhibited a comparable capacity to scavenge pro-inflammatory cytokines similar to DES10-Fly30-GA30. However, it displayed considerably lower levels of anti-inflammatory cytokines than the eutectogel, which is consistent with previous results of immunofluorescence staining. Moreover, compared to the DES10-Fly30GA30 eutectogel group, all two-components ample-treated groups showed higher expression levels of pro-inflammatory factors and lower expression levels of anti-inflammatory factors. These observations were primarily attributed to the deletion of GA, reduced membrane permeability, or impaired retention ability. These results suggest that DES10-Fly30-GA30 eutectogels have the potential to mitigate inflammatory cell infiltration, enhance collagen fiber density, and modulate the transition of periodontal macrophages from the M1 to M2 phenotype, thereby influencing the remodeling of the periodontal inflammatory microenvironment. 2.4.3. Biosafety Assessment In Vivo The long-term biocompatibility of eutectogels administered topically through periodontal pockets is crucial for their clinical application. Therefore, histological examination of the visceral organs using H&E staining (Figure S9, Supporting Information) and blood analysis (Figure S10, Supporting Information) were conducted to assess the potential effects. No significant alterations in the behaviors of the experimental rats were observed throughout the treatment period. Histological examination of the heart, liver, spleen, lungs, and kidneys using H&E staining revealed that the topical application of the eutectogel did not induce any morphological changes or indications of inflammation. Specifically, the cardiac muscle fibers exhibited a trapezoidal shape and well-organized arrangement with no evidence of inflammatory cells. The hepatocytes were arranged in rows and no histopathological changes were detected. The red and white bone marrow within the spleen displayed clear structural integrity without any abnormalities. The alveoli maintained a clear structural appearance and exhibited no signs of edema or inflammatory cell infiltration. The glomeruli exhibited uniformsize, normalshape,and distinct borders, indicating normal renal physiology. The results of routine blood tests indicated that the indicators for all groups were within the normal range. In conclusion, local injection of eutectogels exhibited good biocompatibility and biosafety.
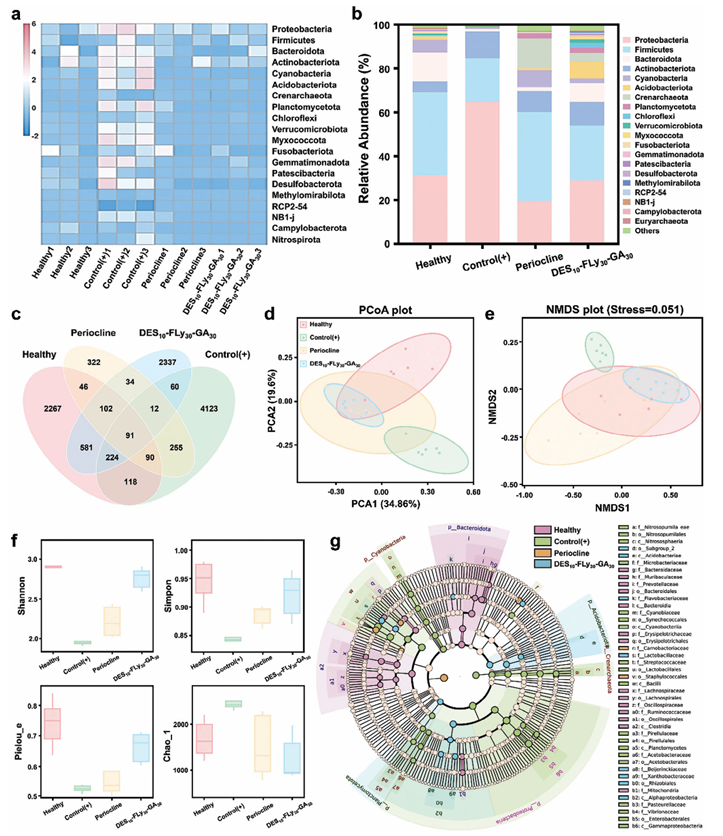
Figure 7. Assessment of oral microbiota in chronic periodontitis using 16S rRNA high-throughput amplicon target sequencing. a) Heat map of the oral microbiota of healthy, control (+), Periocline and DES10-FLy30-GA30 groups. b) Relative abundance bar graph of the oral microbiota of healthy, control (+), Periocline and DES10-FLy30-GA30 groups. c) Venn diagram of the oral microbiota of the oral microbiota of healthy, control (+), Periocline and DES10FLy30-GA30 groups. d,e) PCoA and NMDS analyses of the oral microbiota of the oral microbiota of healthy, control(+), Periocline and DES10-FLy30-GA30 groups f) Shannon, Simpon, Pielous_e and Chao_1 richness index of the oral microbiota of the oral microbiota of healthy, control (+), Periocline and DES10-FLy30-GA30 groups. g) LEfSe analysis identifying the dominant bacteria of healthy, control (+), Periocline and DES10-FLy30-GA30 groups. 2.5. Oral Microbiota in Chronic Periodontitis Oral saliva samples were collected by scraping the oral cavity using a sterile swab, transferred into sterile centrifuge tubes, and sequenced to target the highly variable region of the bacterial 16S rRNA gene. Differences in the oral microbiota of healthy, control(+), periocline, and experimental groups were investigated using species clustering analysis, alpha diversity analysis, beta diversity analysis, and statistical tests (Figures 7 and S11, Supporting Information). The heatmap based on the top 20 most abundant species at the phylum level revealed a significant deviation in the colony structure between the healthy and control(+) groups. The latter harbors numerous pathogenic bacteria, such as Proteobacteria, Firmicute and Bacteroidota and other phyla. The level of consistency observed in the DES10-FLy30GA30 and the Periocline groups was more similar to that of the healthy group, indicating satisfactory antipathogenic qualities. A more comprehensive visualization of the relative abundance of the species is shown in Figure 7b. The healthy group exhibited high relative species abundance, whereas the periodontitis group was predominantly characterized by three pathogenic bacterial phyla, leading to a substantial imbalance within the oral environment. The species abundance in the DES10-FLy30-GA30 group wascomparable to that in the healthy group. As illustrated in Figure S11b, Supporting Information, the relative abundance of DES10-FLy30 group exhibited a similarity to that of the periodontitis group, indicating that DES10-FLy30 was insufficient to eradicate the complex pathogenic flora within the oral cavity. The distribution of amplicon sequence variants (ASVs) shows that the healthy and DES10-FLy30-GA30 groups shared 998 ASVs, whereas the control(+) group shared only 387 ASVs with the DES10-FLy30GA30 group(Figure 7c). Importantly, the Periocline group shared only 329 ASVs with the healthy group. The Venn diagram suggests that DES10-FLy30-GA30 has a greater potential than periocline to induce a shift in the oral microenvironment toward a state similar to that of the healthy group. Subsequent analysis used principal component analysis (PCoA) and non-metric multidimensional scaling (NMDS) based on the unweighted UniFrac distance to assess sample similarity. The primary determinant in PCoA and NMDS within the confidence ellipse, as depicted in Figure 7d,e, Supporting Information, is the interdot distance. A shorter distance signifies decreased dissimilarity. The distance between the healthy group and DES10-FLy30-GA30 group was significantly shorter than that between the DES10-FLy30-GA30 and periodontitis groups, demonstrating a smaller difference between the healthy group and DES10-FLy30-GA30 group than in the control(+) group. The averages of the Shannon, Simpson, Pielous_e, and Chao_1 richness indices are shown in Figure 7f. The Shannon and Simpson diversity indices were used to assess the abundance of species within the samples. An elevated level of community diversity, coupled with a more even distribution of species, was associated with an augmented value for both the Shannon and Simpsonindices. The periodontitis group exhibited low values for both Shannon and Simpson indices, indicating diminished community diversity and microbial community imbalance. These findings were consistent with the results depicted in the relative abundance bar graph. The Shannon and Simpson indices were approximated for both the DES10-FLy30-GA30- and healthy groups, indicating nearly identical levels of community evenness and diversity between the two groups. The Pielou’s evenness index and Chao’s richness estimator are quantitative measures of species diversity. The Pielou_e index was more reflective of the diversity of relatively high-abundance communities, whereas the Chao_1 index was more indicative of the diversity in communities with low abundance. Among the experimental groups, the control(+) group exhibited the lowest Pielou_e and the highest Chao_1 indices. This indicated that the control(+) group had few high-abundance communities, whereas the low-abundance communities were more abundant. The results revealed that in the periodontitis group, the proliferation of pathogenic bacteria encroached on the low-abundance community and impinged on the survival of other microorganisms,thereby leading to an acute imbalance in the oral microenvironment. Notably, the results obtained for the DES10-FLy30-GA30 group closely resembled those observed in the healthy group regardless of whether the communities were high or low in abundance. This finding suggested that the DES10-FLy30-GA30 eutectogel effectively preserved community richness comparable to that of the healthy group in both low- and high-abundance oral communities, altering the imbalance in the oral microenvironment caused by periodontitis. The bacterial species from different groups at the family level were analyzed using the linear discriminant analysis effect size (LEfSe) test, as depicted in Figure 7g.In the healthy group, the predominant strains were identified as belonging to the Bacteroidota within the intestinal microflora. Conversely, the periodontitis group exhibited a notable prevalence of pathogenic bacteria, including members of the Vibrionaceae, Enterobacterales, Gammaproteobacteria, and Streptococcaceae families, along with inflammatory bacteria, such as Pasteurellaceae and Bacilli. In addition, pathogenic bacteria were scarcely detected in the Periocline and DES10-FLy30-GA30 groups, suggesting asignificant reduction in periodontitis-associated pathogenic bacteria has been achieved. The application of the DES10-FLy30GA30 eutectogel demonstrated remarkable efficacy in eradicating periodontitis-associated pathogenic bacteria, thereby restoring an imbalanced oral microenvironment. 3. Conclusion In conclusion, the advantages of high biocompatibility, prosolubilization, and pro-permeability of the ChCl/M DES were combined to develop an injectable DES10-FLy30-GA30 eutectogel using a one-pot method. The eutectogel exhibited remarkable broad-spectrum antimicrobial properties in vitro because of GA’s ability to perforate the bacterial periplasm and excellent antiinflammatory effects by effectively scavenging ROS and inducing the polarization of macrophages from the M1 phenotype to the M2phenotype. Additionally, the eutectogels demonstrated the capacity to suppress alveolar bone resorption in ligation-induced chronic periodontitis, reducing the CEJ-ABC value of 1298 ± 21.42 μm in periodontitis group to 980 ± 4.22 μm, while also attenuating osteoclast activity and decreasing the inflammatory response. Furthermore, the 16S RNA test revealed that the eutectogels showed excellent efficacy in eradicating pathogenic bacteria while simultaneously restoring the balance of the oral microflora and preserving homeostasis within the oral microenvironment. This exceptional performance can be attributed to the ChCl/M DES and the macromolecular peptide FLy in this eutectogel, which effectively enhanced the dissolution rate and permeability of the small-molecule drug GA. Consequently, the cellular uptake efficiency and bioavailability of GA were significantly improved. Therefore, the incorporation of eutectogels into biology presents a novel concept for enhancing drug utilization and establishing a straightforward, yet efficacious, antibacterial and anti-inflammatory defense platform for managing inflammatory diseases. Supporting Information Supporting Information is available from the Wiley Online Library or from the author. Acknowledgements This work was supported by the National Natural Science Foundation of China (Grant Nos. 51925304, 52203180, and 52073191). We thank Dr.Yani Xie from the Analysis and Testing Center of Sichuan University for helping us perform CD studies. Part of the schematic was drawn by FIGDRAW. Conflict of Interest The authors declare no conflict of interest. Data Availability Statement The data that support the findings of this study are available from the corresponding author upon reasonable request. Keywords anti-inflammation, cell permeation, chronic periodontitis, eutectogel, selfassembly Source: www.advmat.de |