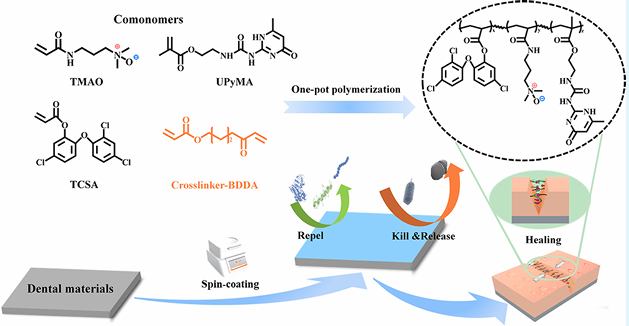
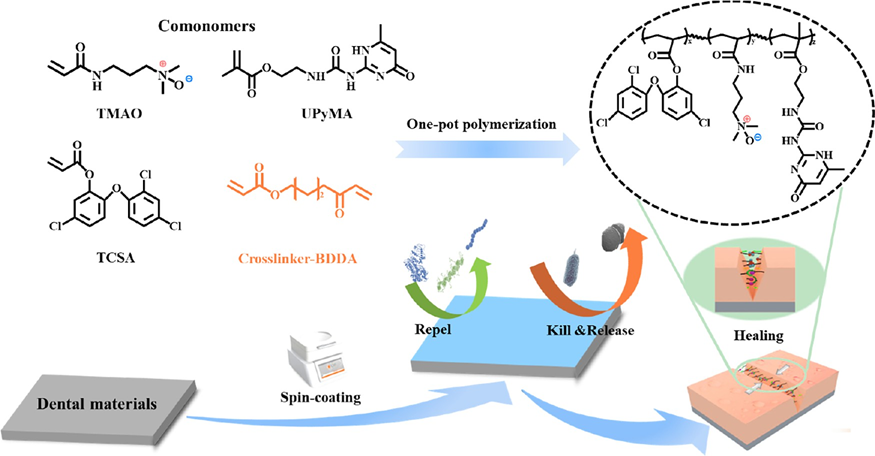
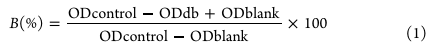Supramolecular Self-Healing Antifouling Coating for Dental MaterialsIssuing time:2025-05-26 11:46
Abstract In orthodontic treatment, orthodontic appliances are prone to bacterial infections,which pose a risk to oral health. Surface modification of orthodontic appliances has been explored to improve their antifouling properties and impart antibacterial capabilities, inhibiting initial bacterial adhesion and biofilm formation. However, coatings are susceptible to damage in the complex oral environment, leading to a loss of functionality.Here, we have prepared an antifouling self-healing coating based on supramolecular bonding by employing a simple spin coating method. The presence of the hydrophilic zwitterionic trimethylamine N-oxide (TMAO) and the hydrophobic antimicrobial moieties triclosan acrylate (TCSA) imparts to the polymers an amphiphilic structure and enhances the interaction with bacteria, resulting in excellent antimicrobial activity and surface antifouling properties. The multiple hydrogen bonds of ureido-pyrimidinone methacrylate (UPyMA) and ionic interactions contained in the polymers not only increased the adhesion of the coating to the material substrate (approximately 3 times) but also endowed the coating with the intrinsic self-healing ability to restore the antibiofouling properties at oral temperature and humidity. Finally, the polymer coating is biologically safe both in vitro and in vivo, showing no cytotoxic effects on cells and tissues. This research offers a promising avenue for improving the performance of orthodontic appliances and contributes to the maintenance and treatment of oral health. KEYWORDS: surface coating, dental appliance, antibacterial, antifouling, supramolecular self-healing
1. Introduction With increasing oral health awareness, various dental materials such as metals, ceramics, and polymers are being developed for implant treatment and orthodontics. However, these dental materials are susceptible to the accumulation of bacteria, proteins, and food residues due to the absence of metabolic renewal function on their surfaces, leading to the formation of plaque retention sites in the intricate oral environment. Persistent plaque retention promotes the rapid proliferation of bacteria, which not only fostering the development of bacterial infectious oral diseases such as dental caries and periodontitis but also lead to mechanical deterioration of the materials and affect the service life of the materials. Consequently, there is a significant opportunity for the development of novel dental biomaterials that exhibit both functional and aesthetic properties, which holds promising implications in the field of dentistry. To inhibit bacterial adhesion and proliferation on the surface of orthodontic appliances, physical coatings or chemical grafting of inorganic/organic antibacterial agents and hydro- philic materials such as polyethylene glycol (PEG) and zwitterionic compounds have been employed. PEG forms a dense hydration layer on the material surface by binding with water molecules through hydrogen bonding, creating a physical barrier against protein adsorption and exhibiting antifouling properties. However, due to its molecular structure, PEG is susceptible to auto-oxidation, forming aldehydes or ethers in the presence of oxygen and metals, leading to the loss of its antiprotein adsorption functionality. Zwitterionic compounds, on the other hand, bind with water molecules through strong ion?solvent interactions. Studies have shown that zwitterionic compounds exhibit superior antifouling performance compared to PEG. However, when commonly used in physiological environments such as the human body, zwitterionic compounds experience charge shielding due to the abundance of salt ions and various biological molecules in bodily fluids, which weakens their hydration capacity and reduces their antifouling properties. Research has reported that the closer the distance between positive and negative charges in zwitterionic compounds, the stronger their hydration capacity. The novel zwitterionic compound trimethylamine N-oxide (TMAO), with its positive and negative charges in close proximity, demonstrates superior hydration and antifouling capabilities compared to PEG, as well as lower immunogenicity. Furthermore, TMAO molecules exhibit a smaller dipole moment, with methyl groups surrounding the N+ ion and only a portion of the O? exposed in solution. The short distance between O? and N+ groups results in strong repulsion between N+ and other ions like Na+, thereby resisting ion-induced damage in saline solutions and exhibiting excellent antifouling properties in physiological environments. During usage, damage or defects may occur, which can affect the functionality of the material. Endowing antifouling coatings with self-healing capabilities can reduce costs, improve the lifespan of materials, and achieve long-term antifouling functionality. Compared to exogenous self-healing approaches that have limited repair cycles and difficulty in achieving functional restoration, intrinsic self-healing materials based on supramolecular noncovalent bonds (such as hydrogen bonding, host?guest interactions, and ionic interactions) can repair damaged areas through dynamic recombination of supramolecular bonds. The continuous breaking and rearrangement of supramolecular noncovalent bonds theoret- ically enable unlimited healing. By selection of suitable supramolecular units, self-repair can be achieved at room temperature, leading to functional restoration of the damaged region. Quadruple-hydrogen-bonding moieties ureidopyrimi- dinone (UPy), which possesses quadruple hydrogen bonds, find wide application in the field of self-healing materials. It can also enhance the adhesion to the substrate through hydrogen bonding interactions, thereby improving the stability of the coating. To impart excellent antifouling properties to the surface of the dental materials, a novel hydrophilic self-healing multi- functional coating with excellent antiadhesion and bactericidal properties was developed in this work. This coating was prepared by the simple spin-coating of polymers on the surface of various dental materials. The polymers, named as PTTU, were synthesized by the feasible one-pot free radical polymer- ization of TCSA, TMAO, and UPyMA. The presence of the amphiphilic hydrophilic monomer TMAO and hydrophobic antimicrobial unit TCSA imparts the PTTU polymer with an amphiphilic structure and enhances the interaction with bacteria, resulting in excellent antimicrobial activity and surface antifouling properties. The multiple hydrogen bonds and ionic interactions contained in the PTTU endowed the coating with intrinsic self-healing ability to improve the durability of coatings at oral temperature and humidity. This multifunctional self-healing coating provides long-lasting prevention of related oral diseases and has a valuable reference for the development of new dental materials (Scheme 1).
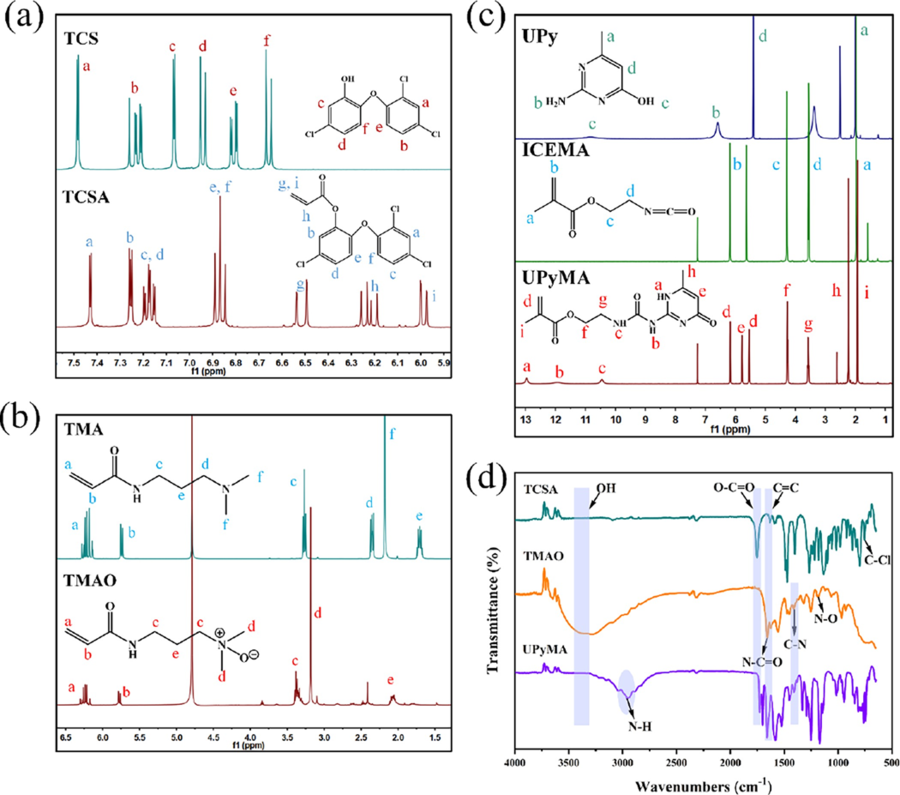
Scheme 1 Schematic Diagram of the Synthetic Process and the Preparation and Application of PTTU Coating 2. Materials and Methods 2.1 Materials Triclosan (TCS), acryloyl chloride, dichloro-methane (DCM), triethylamine (TEA), petroleum ether (PE), acetone, anhydrousethyl ether, methanol, dimethyl sulfoxide (DMSO), trichloromethane, and other chemical solvents and reagents were purchased from Haihong Reagent Co.,Ltd. (Chengdu,China). 2-Amino-4-hydroxy-6-methylpyrimidine, 2-isocyanatoethyl methacry- late (ICEMA), N-[3-(dimethylamino) propyl] acrylamide), 1,4-bis- (acryloyloxy) butane diethylenetriaminepentaacetic acid 2,2′-azobis (2-methylpropionitrile), 4 methoxyphenol (MEHQ), and hydrogen peroxide (H2O2) were bought from Shanghai Aladdin Biochemical Technology Co., Ltd. (Shanghai, China). Bovine serum albumin(BSA), lysozyme (Lz), phosphate buffered solution (PBS), penicillin, streptomycin, and trypsin were commercial lyavailable from Chengdu Bosco Biotechnology Co., Ltd. (Chengdu, China). Fluorescein isothiocyanate (FITC) was purchased from Beijing Solarbio Science & Technology Co., Ltd. (Beijing,China). Cell Counting Kit-8 (CCK- 8) was obtained from Apexbio Co., Ltd. (USA). Fluorescein diacetate (FDA) was bought from Sigma(USA). The BCA protein assay kit, propidium iodide (PI), and 2-(4-amidinophenyl)-6-indolecarbami- dine dihydrochloride (DAPI) were obtained from Beyotime Biotechnology Co., Ltd.(Shanghai,China).The brain heart infusion medium (BHI), nutrient agar, LIVE/DEAD BacLight bacterial viability kit (L7012), Gibcoalpha-modified Eagle medium (alpha- MEM), and Gibco fatal bovine serum (FBS) were purchased from Thermo Fisher Scientific Co., Ltd. (USA). High-purity oxygen was provided by Chengdu Hengyuan Gas Co., Ltd. (Chengdu, China). 2.2 Characterization 1H NMR and molecular weights of the synthetic products were examined by Bruker NMR spectrometry (400 MHz) and high-resolution mass spectrometry (Element XR/Thermo Fi). The FTIR spectra were examined by Fourier transform attenuated total reflectance infrared spectroscopy (ATR-FTIR, Thermo Fisher Corp., USA) in the wavelength range 600?4000 cm?1 with a resolution of 4 cm?1 and 64 scans. The elemental and chemical composition of the surface were performed on an X-ray photoelectron spectrometer (XPS, Thermo Scientific K-Alpha+, USA) from 0 to 1200 eV. The water contact angle (WCA) was carried out using a DSA 25 contact angle goniometer (KRUSS, Germany). Surface morphology, roughness, and adhesion of proteins and bacteria were observed by scanning electron microscopy (SEM, FEI/ INSPECT F50, USA), fluorescence microscopy (IX71S1F-3, OLYMPUS, Japan), atomic force microscope (AFM, Bruker, USA), and confocal laser scanning microscope (CLSM, LSM 700, ZEISS, Germany). The fluorescence intensity of proteins and bacteria on surfaces was analyzed using ImageJ. Transmittance and UV absorption spectra were examined by a UV?vis?NIR spectropho- tometer (UV-3600). The thermal properties of the polymer in the temperature range of ?50 to 150 °C were investigated by differential scanning calorimetry (DSC 25 TA Instruments, USA) at a rate of 10°C/min under a nitrogen atmosphere. 2.3 Synthesis of PTTU 2.3.1 Synthesis of TCSA. The synthesis of TCSA was carried out as follows: 6 mmol (1.74 g) of TCS was dissolved in 15 mL of anhydrous DCM, followed by the addition of 18 mmol of TEA as a deacid reagent (2.5 mL). The reaction mixture was kept at 0 °C, and then 18 mmol (1.44 mL) of acryloyl chloride was added dropwise over 30 min. The progress of the reaction was monitored by thin-layer chromatography on silica gel plates. After the mixture was stirred at room temperature for 6 h, the reaction was stopped. The crude product was obtained by rotary evaporation under reduced pressure, extracted with a large amount of DCM, and dried with anhydrous Na2SO4. After another round of rotary evaporation under reduced pressure, a yellow, viscous liquid was obtained and stored at ?20 °C for further use. NMR results are shown in Figure 1a. 1H NMR (400 MHz, CDCl3) δ 7.43 (d, J = 2.5 Hz, 1H), 7.25 (d, J = 2.5 Hz, 1H), 7.20?7.15 (m, 2H), 6.87 (t, J = 8.9 Hz, 2H), 6.51 (dd, J = 17.3, 1.1 Hz, 1H), 6.22 (dd, J = 17.3, 10.5 Hz, 1H), 5.99 (dd, J =10.5, 1.1 Hz, 1H). 2.3.2 Synthesis of UPyMA. Four grams (32 mmol) were dissolved in 50 mL of anhydrous DMSO. The mixture was heated in an oil bath at 170 °C for 10 min until the solid was completely dissolved, and then the oil bath was removed. Immediately, 5.5 g (35.2 mmol, 1.1 equiv) of ICEMA was added to the solution and vigorously stirred for 30 s. To prevent monomer self-polymerization, the mixture was rapidly cooled in an ice bath at 0 °C with continuous stirring, resulting in the gradual precipitation of a white solid. The white solid was washed three times by repeated centrifugation (8000 rpm) with acetone, followed by vacuum drying at 40 °C. After being ground in a mortar, the product was stored at ?20 °C for later use. NMR result is shown in Figure 1b. 1H NMR (400 MHz, CDCl3) δ 12.97 (s, 1H), 11.90 (s, 1H), 10.42 (s, 1H), 6.17 (s, 1H), 5.78 (s, 1H), 5.56?5.51 (m, 1H), 4.26 (t, J = 5.6 Hz, 2H), 3.57 (dd, J = 11.1, 5.5 Hz, 2H), 2.23 (s, 3H), 1.93 (s, 3H). 2.3.3 Synthesis of TMAO. DTPA (80 mg) and 30 mL of ultrapure water were added to a round-bottom flask and vigorously stirred for 2 h until the white powder was completely dissolved. The oil bath was heated to 60 °C, and then 3.4 mL of hydrogen peroxide (H 2 O 2 , 30 wt %) was added, followed by oxygen bubbling. Next, 14.4 g of N,Ndimethylacrylamide was dissolved in 10 mL of ultrapure water and slowly added dropwise to the round-bottom flask through a pressure-equalizing funnel at 60 °C. The reaction was carried out for 6 h at 60 °C. The reaction product was extracted multiple times with DCM, and the aqueous phase was collected. Finally, the product was dissolved in a large amount of anhydrous methanol and subjected to vacuum distillation to obtain the oily brownish-yellow product, TMAO, which was stored at ?20 °C in a refrigerator for later use. NMR results are shown in Figure 1c. 1 H NMR(400 MHz, D 2 O) δ: 6.23 (qd, J = 17.1, 5.8 Hz, 2H), 5.77 (dd, J = 9.7, 1.9 Hz, 1H), 3.40? 3.31 (m, 4H), 3.18 (s, 6H), 2.11?2.03 (m, 2H). 2.3.4 Synthesis of the PTMAO Polymer. Twenty-four millimoles of TMAO were dissolved in 20 mL of DMSO, and then 1% 2,2′azobis(isobutyronitrile) (AIBN) and 3.5% 1,4-butanediol diacrylate (BDDA) were added. The mixture was subjected to three cycles of freezing?thawing?vacuuming in a liquid nitrogen environment. The polymerization reaction was carried out at 70 °C in an oil bath for 24 h. After that, 0.2 g of MEHQ was added to terminate the reaction. The resulting mixture was precipitated three times in ice ether and then dialyzed for 3 days before being freeze-dried to obtain the PTMAO polymer and stored at ?20 °C for later use. 2.3.5. Synthesis of the PTCSA Polymer. Ten millimoles of TCSA monomer were dissolved in 20 mL of DMSO, and the PTCSA polymer was synthesized using the aforementioned free radical polymerization method. The polymer was stored at ?20 °C for later use. 2.3.6 Synthesis of PTT Polymers. To optimize the antibacterial and antifouling properties of the PTT polymer, the polymer's performance was optimized by adjusting the ratio of TMAO and TCSA monomers. In simple terms, different mass ratios of TMAO/ TCSA (1:1, 2:1, 4:1, 8:1, 12:1, and 16:1) were mixed and dissolved in 20 mL of DMSO. The same molar ratio of initiators and cross-linking agents was added, and free radical polymerization was carried out at 70 °C for 24 h. Then, 100 mg of MEHQ was added to terminate the reaction. The polymer was precipitated in diethyl ether and dialyzed in ultrapure water for 3 days to obtain PTT polymers with different monomer ratios. The polymers were stored at ?20 °C for later use. 2.3.7 Synthesis of PTTU Polymers. To incorporate 10, 20, and 30% of the UPyMA monomer into the optimal antibacterial and antifouling polymer obtained above, a free radical copolymerization was performed. First, the UPyMA monomer with different weight ratios was dissolved completely in DMSO. Then, it was mixed with the optimal ratios of TMAO and TCSA monomers obtained earlier. The mixture underwent free radical polymerization for 24 h. MEHQ was added to terminate the reaction, and the mixture was cooled to room temperature. The precipitate was collected in diethyl ether, followed by dissolution in chloroform. This precipitation?dissolution process was repeated three times to remove any unreacted UPyMA monomer. Finally, the polymer was dialyzed in ultrapure water for 3 days to obtain PTTU polymers with different UPy contents.
Figure 1 Synthesis and characterization of monomers. (a) 1H NMR spectra of TCS and TCSA. (b) 1H NMR spectra of TMA and TMAO. (c) 1H NMR spectra of UPy, ICEMA, and UPyMA. (d) FTIR spectra of TCSA, TMAO, and UPyMA. 2.4 Antibacterial Performance Evaluation The minimum inhibitory concentration (MIC) and minimum bactericidal concen-tration (MBC) of TCSA, PTCSA, PTMAO, PTT, and PTTU were determined using the broth microdilution method. Since TCSA, PTCSA, PTT, and PTTU are not soluble in water, they were first prepared in BHI (brain heart infusion) solution containing 5% DMSO at specific concentrations. The monomers/polymers were serially diluted in a BHI medium to obtain 100 μL of different concentrations. Then, 100 μL of an S. mutans suspension (10 6 CFU/ mL) was added to each well. The plates were incubated at 37 °C under anaerobic conditions for 24 h. To accurately measure the antibacterial activity of the monomers and polymers, which may cause turbidity and exhibit absorbance when mixed with the BHI medium, a 5% DMSO-BHI solution without bacteria was used as a control. The bactericidal rate (B%) was calculated using the following formula eq 1:
where ODcontrol, ODtest, ODblank, and ODdb represent the optical density values of the bacterial group, experimental group, blank culture medium group, and 5% DMSO-BHI material group, respectively, after incubation under anaerobic conditions for 24 h. 2.5 Lap-Shear Test and Coating Stability The interfacial adhesion performance of all polymers was evaluated according to the ASTM D1002 test standard. In brief, a 10 mg/mL polymer solution was prepared and coated onto a 2.5 × 2.5 cm 2 area of a Ti substrate. Another Ti substrate of the same size was overlaid onto the polymercoated Ti substrate. The bonded assembly was cured, and lap-shear tests were performed by using a universal testing machine at a crosshead speed of 2 mm/min. All lap-shear adhesion tests were conducted at room temperature, and each sample was tested three times. To assess the stability of the antimicrobial coatings, we measured the zone of inhibition for different polymer coatings. A total of 50 μL of S. mutans BHI culture medium was evenly spread on a nutrient agar plate using a sterile swab. Then, Ti substrates with different polymer coatings were inverted and placed on the agar plates. The plates were incubated at 37 °C under anaerobic conditions for 24 h to observe the inhibition zones. The presence or absence of inhibition zones indicated the antimicrobial mode of the polymer coatings and provided insights into the stability of the coatings in a bacterial environment. 2.6 Antifouling Performance The antifouling performance of various polymer coatings against BSA and LZ was evaluated using previous methods. Additionally, qualitative analysis was conducted by capturing fluorescence images of FITC-labeled BSA and LZ adsorbed on the coating surfaces. Furthermore, Ti substrates with different polymer coatings were immersed in 1 mL of an S. mutans bacterial suspension (10 6 CFU/mL) and incubated for 48 h. Subsequently, bacterial viability was assessed on the coating surfaces by staining the bacteria using a bacterial live/dead staining kit and visualizing them using laser confocal microscopy. The bacteria on the Ti surface were fixed with 2.5 wt % glutaraldehyde, dehydrated in a gradient ethanol series for 15 min, dried, and sputter-coated with gold before SEM observation. 2.7 Self-Healing Performance of PTTU Coatings on Dental Materials Modern orthodontic treatment techniques not only effectively correct various dental malocclusions but also provide aesthetic, fast, comfortable, and efficient orthodontic treatments for patients. To explore the feasibility of PTTU polymer coatings on dental materials, the influence of the coatings on the transparency, hydrophilicity/hydrophobicity, and antimicrobial properties of invisible braces, steel 316L, and dental resin was investigated before and after applying the PTTU coatings. Moreover, the variation of surface WCA of the coatings before and after self-healing was investigated to evaluate their self-healing properties on various dental material. Subsequently, cracks of the same size and shape (20 μm wide and 50 μm deep) were introduced on the surfaces of PTTU polymer coatings by using a blade. The samples were then placed in simulated saliva fluid (SSF) containing lysozyme (20 μg/mL) and α-amylase (300 μg/mL) at 37 °C in a shaker to simulate oral environments. The healing performance of the cracks was periodically examined and photographed by using optical microscopy. The PTTU-10% coatings were immersed in FITC-BSA solution (1 mg/mL) for 30 min before and after self-healing, and their surface antifouling capability was examined by fluorescence microscopy. 2.8 Biocompatibility of PTTU The cytotoxicity and hemolytic activity of various polymer coatings were evaluated using the experimental methods described in the previous work. In brief, HOK cells were seeded in 48-well plates at a density of 10,000 cells per well. Each group consisted of six parallel samples. After 24 h of cell seeding, Ti substrates with polymer coatings were placed upside down on the cells and further incubated for 24 and 72 h. After a 2 h coincubation with 10% CCK-8 solution, the absorbance at 450 nm was measured using a microplate reader to determine cell viability. Subsequently, under dark conditions, cells were stained with FDA/PI and TRIIC-phalloidine/DAPI and rapidly observed using an inverted fluorescence microscope. Furthermore, each polymer coating was coincubated with 2% whole blood solution at 37 °C for 2 h. After centrifugation, images were captured, and the hemolysis rate was calculated using eq 2.
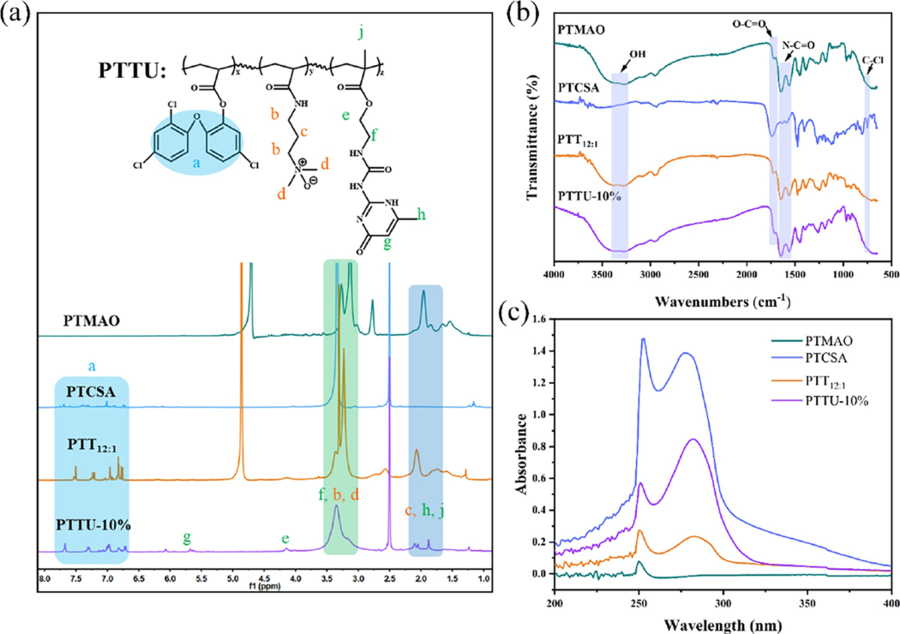
where A, B, and C represent the absorbance of the experimental group, negative control group (diluted with saline), and positive control group (diluted with reverse osmosis water), respectively. 2.9 Biosafety Evaluation In Vivo Animal experiments were conducted with the approval of the Animal Research Committee of Sichuan University (Approval No: 2019065A). In this study, 8-week-old male Sprague?Dawley (SD) rats were selected for the in vivo safety assessment to provide guidance for clinical practices. In brief, 10 SD rats were randomly divided into two groups: control group and experimental group, with five rats in each group. The control group was orally administered with ultrapure water, while the experimental group was orally administered with simulated saliva extract of PTTU 10% polymer coating (250 μL/kg) daily, which was calculated based on the amount of saliva a person secretes daily (1?1.5 L) and the surface area of the braces. The body weight changes of both groups of SD rats were measured and recorded daily. After 7 days, blood samples were collected via the retroorbital plexus for hematological analysis. Furthermore, oral mucosa, dorsal tongue mucosa, and internal organs were collected, fixed, and subjected to hematoxylin and eosin (H&E) staining for histopathological evaluation of biocompatibility. 2.10 Statistical Analysis One-way ANOVA was used for statistical analysis. Data were expressed as mean ± standard deviation (Mean ± SD) and p* <0.05, p** <0.01, indicated the level of statistical difference. 3. Results and Discusstion 3.1 Synthesis and Formulation Optimization of the PTTU To prepare multifunctional self-healing coatings, we first synthesized TCSA, TMAO, and UPyMA monomers with antibacterial, antifouling, and self-healing functionalities (Figure S1). As shown in Figure 1a, the characteristic peaks at δ = 6.65?7.6 ppm correspond to the two benzene rings of TCS. Upon esterification reaction with acryloyl chloride, new peaks at δ = 6.8?7.5 ppm indicative of the benzene rings of TCS were observed, along with distinct peaks at δ = 5.95?6.55 ppm corresponding to the C═C double bonds. The molecular weight of TCSA was determined to be [M + H]?=341.30 which matched the theoretical value (Figure S2). To enhance the antifouling ability of the polymer coatings in a high salt concentration environment in the oral cavity, a novel zwitterionic monomer TMAO with enhanced hydration capacity was synthesized. TMA was converted to the zwitterionic monomer TMAO through catalytic oxidation while retaining the C═C double bonds for subsequent polymerization reactions. The molecular weight of TMAO was determined to be [M + H]+ = 173.13 (Figure 1b, Figure S3), consistent with previous reports. To enhance the adhesion between the polymer and substrate and impart selfhealing properties to the polymer coatings, monomer UPyMA with quadruple hydrogen bonding was synthesized via the addition reaction of ICEMA and UPy (Figure 1c). Highresolution mass spectrometry confirmed its molecular weight to be [M + H]+ = 281.1, consistent with the theoretical value and literature (Figure S4). Furthermore, the FTIR spectrum shown in Figure 1d exhibited characteristic absorption peaks at 1750, 1657, 1632, 1411, 1180, and 752 cm ?1 , corresponding to O?C═O, N?C═O, C═C, C?N, N?O, and C?Cl, respectively. These results indicate the successful synthesis of TCSA, TMAO, and UpyMA monomers. The TCSA exhibited excellent antibacterial performance with both MIC and MBC values of 125 μg/mL (Figure S5). Being a hydrophobic monomer, an increase in the content of TCSA in the polymer chain would decrease the hydrophilicity and biocompatibility of the polymer coatings. To obtain functional copolymer coatings with acceptable antibacterial and antifouling properties, the molar ratio of TMAO and TCSA was turned from 1:1 to 16:1 for the polymerization of PPTx, x is the mass ratio of TMAO to TCSA. The results demonstrated the disappearance of double bond peaks at δ = 5.5?6.25 ppm and the appearance of distinct peaks corresponding to the benzene rings of TCSA, indicating the complete polymerization of TMAO and TCSA. With an increasing proportion of the TMAO monomer, the relative intensity of the methylene (b, c) and methyl (d) characteristic peaks corresponding to TMAO increased gradually (Figure S6a). The infrared spectra also displayed characteristic absorption peaks of each functional moieties, with a decreasing intensity of the C?Cl absorption peak as the TCSA content decreased (Figure S6b). These results confirm the successful synthesis of PTTx polymers with different PTMAO/PTCSA ratios. Subsequently, the antibacterial performance of PTTx polymers and the wettability of the corresponding coatings were evaluated (Table S1). Although PTMAO contains quaternary ammonium ions with positive charges, its overall antibacterial effect was not significant due to the interaction with negatively charged oxygen ions. Despite the acceptable antibacterial performance of TCSA (MBC= 125 μg/mL), the antibacterial effect of PTCSA obtained by free radical polymerization was not remarkable (MBC >500 μg/mL). This could be attributed to the excessively hydrophobic nature and steric hindrance of the entire PTCSA chain, which weakened its interaction with bacteria. PTTx, on the other hand, possesses an amphiphilic structure due to the presence of both hydrophobic benzene rings and hydrophilic zwitterionic groups in its molecular chain. This amphiphilic structure facilitates interaction with bacterial cell membranes and significantly enhances its antibacterial performance (MBC <62.5 μg/mL). Furthermore, the antibacterial activity of PTTx was optimal at a certain ratio of TMAO and TCSA. with an increase in PTMAO content, its antibacterial effect initially increases and then decreases, but the trend is not significant. The hydrophilicity of the corresponding functional coatings also increases with an increase in the PTMAO content. Considering the overall antibacterial performance and hydrophilicity of the polymer coating, PTT 12:1 (TMAO/TCSA in a 12:1 ratio) was optimized for the subsequent copolymerization with UPyMA. Accordingly, different mass ratios of the UPyMA monomer (10, 20, and 30%) were incorporated to obtain PTTU polymers with the self-healing ability and strong interfacial adhesion strength. As shown in Figure 2a, the double bond peak at δ = 5.5?6.25 ppm disappeared after polymerization, and characteristic peaks of TMAO, TCSA, and UPyMA were observed in the NMR spectrum of PTTU-10%, indicating the successful synthesis of PTTU-10%. The final ratios of the components PTMAO, PTCSA, and PUPyMA in PTTU were 86, 5, and 9%, respectively, as calculated by the 1 H NMR spectrum of PTTU-10%, which were basically consistent with the initial feeding ratios. However, the double bond peaks of PTTU-20% and PTTU-30% did not disappear, implying that the monomers were not fully polymerized. With an increase in the UPyMA content, the characteristic peak of UPyMA became more pronounced (Figure S7). This may be attributed to the enhanced intermolecular hydrogen bonding with an increasing UPy content, which inhibited the progress of the polymerization reaction and made it challenging to purify the purification of products. The FTIR spectra of various polymers revealed representative absorption bands of functional moieties in good accordance with three monomers (Figure 2b). Since the functional moieties (OH, NH, Cl, O═N, etc.) and ring peaks (benzene ring of TCS, hexatomic ring of UPy) have distinct absorption peaks at 250 and 250?300 nm, the UV?vis spectral shifts were employed to verify the successful synthesis of individual polymers. Due to the benzene rings and active hydrogens, TCS and UPy exhibit strong absorption in the ultraviolet region. The results showed that PTMAO had an absorption peak only at 250 nm, while PTCSA exhibited strong absorption at 250 nm and in the range of 250?300 nm. PTT 12:1 , which contains a small amount of TCS in the polymer chain, also exhibited absorption in the 250?300 nm range. PTTU-10%, PTTU-20%, and PTTU-30% contained both TCS and UPy, resulting in stronger absorption peaks at 250 and 250?300 nm (Figure 2c, Figure S7c). Subsequently, the effect of UPyMA content on the antibacterial properties of PTTU was used to examine the antibacterial performance of PTTU. The results showed that PTTU maintained good antibacterial activity with the addition of a small amount of UPyMA (PTTU-10%), and MIC and MBC values increased with the increase in UPyMA content (Table S2).
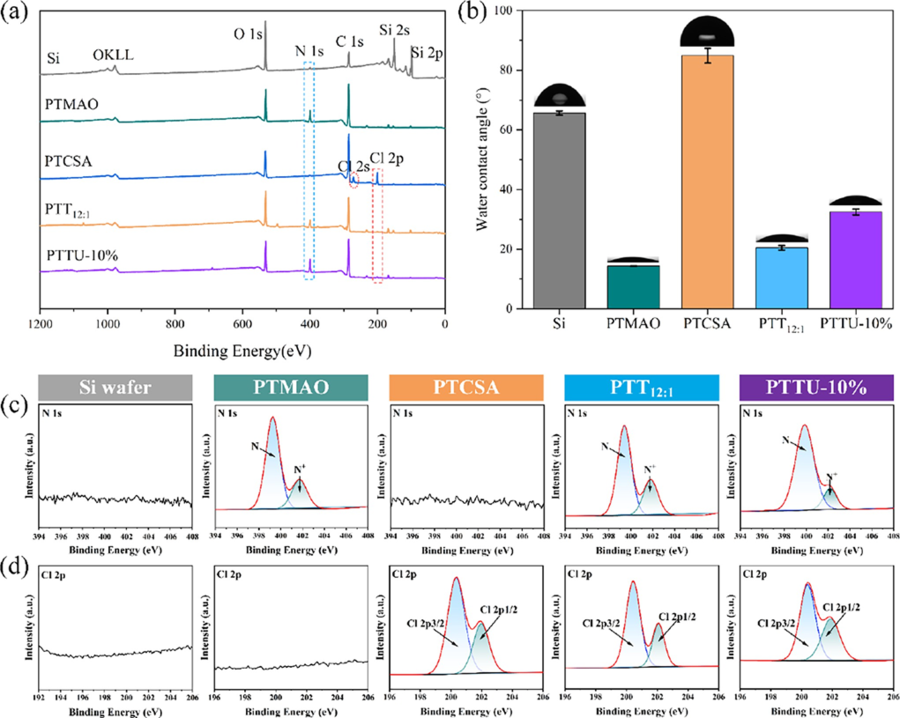
Figure 2 Synthesis and characterization of polymers. (a) 1H NMR, (b) FTIR, and (c) UV?vis spectra of PTMAO, PTCSA, PTT12:1, and PTTU- 10%, respectively. 3.2 Preparation and Characterization of Polymeric Coatings Each polymer coating was prepared by simple spincoating on the Si surface as a model, and the surface composition and morphology were examined by XPS, SEM, and AFM. As shown in Figure 3a, the bare Si wafer exhibited characteristic peaks for C, O, and Si. By contrast, after the deposition of various functional coatings on the surface, intense signals of N and Cl elements were detected, assignable to the introduction of TMAO, UPyMA, and TCS. To further validate the surface elements and their chemical states, highresolution spectra of C 1s, O 1s, N 1s, and Cl 2p were analyzed. In comparison to the Si substrate and PTCSA coating, the PTMAO, PTT 12:1 , and PTTU-10% coatings exhibited a prominent N 1s signal at a binding energy of 400 eV (Figure 3c). Additionally, the PTCSA, PTT 12:1 , and PTTU-10% coatings showed distinct Cl 2p3/2 and Cl 2p1/2 characteristic peaks at binding energies of 200.4 and 202.5 eV, indicating the successful incorporation of the PTTU polymer onto the Si surface (Figure 3d). Moreover, the C 1s high-resolution spectra of the Si wafer was fitted with two peaks representing C?H/C?C (284.8 eV) and C?O/C?N (286.8 eV). The PTMAO, PTCSA, PTT 12.13%, while the PTCSA coating exhibited an increased Cl content of 5.88%. The PTT 12:1 and PTTU-10% coatings contained both nitrogen and chlorine elements. The N content increased from 10.24 to 14.52% after the introduction of UPy units, while the Cl content decreased from 1.09 to 0.56%. Furthermore, the wettability of the polymer coatings was investigated by measuring the water contact angles (WCA). The WCA values of the blank Si, PTMAO, PTCSA, PTT 12:1 , and PTTU-10% coatings are 65.67°, 14.36°, 84.88°, 20.47°, and 32.50°, respectively (Figure 3b). Compared to the original Si substrate, the PTCSA coating exhibited increased hydrophobicity due to the presence of hydrophobic benzene rings on its surface. On the other hand, the PTMAO, PTT 12:1 12:1 , and PTTU-10% can be additional divided into three or four peaks, assignable to N═C?O (286.4 eV), C?Cl (286.9 eV), and O═C?O (288.7 eV), respectively, further confirming the composition of various coatings (Figure S8a). The elemental contents for different polymer coatings are summarized in Table S3. In comparison to the blank Si wafer, the functional coatings exhibited a significant increase in C content and a decrease in O content. The PTMAO coating showed an increased N content of 12.13%, while the PTCSA coating exhibited an increased Cl content of 5.88%. The PTT 12:1 and PTTU-10% coatings contained both nitrogen and chlorine elements. The N content increased from 10.24 to 14.52% after the introduction of UPy units, while the Cl content decreased from 1.09 to 0.56%. Furthermore, the wettability of the polymer coatings was investigated by measuring thewater contact angles (WCA). The WCA values of the blank Si, PTMAO, PTCSA, PTT 12:1 , and PTTU-10% coatings are 65.67°, 14.36°, 84.88°, 20.47°, and 32.50°, respectively (Figure 3b). Compared to the original Si substrate, the PTCSA coating exhibited increased hydrophobicity due to the presence of hydrophobic benzene rings on its surface. On the other hand, the PTMAO, PTT 12:1, and PTTU-10% coatings displayed good surface hydrophilicity due to the integration of zwitterionic functional groups. However, the hydrophilicity of the PTT 12:1 and PTTU-10% coatings slightly decreased due to the influence of the benzene rings and UPy units. These results demonstrate that the PTTU polymer coatings can be easily obtained through simple spin-coating methods.
Figure 3 Surface composition and hydrophilicity/hydrophobicity of polymeric coatings. (a) XPS wide-scan spectra. (b) water contact angles and (c) N 1s and (d) Cl 2p high-resolution spectra of bare Si wafer, PTMAO, PTCSA, PTT12:1, and PTTU-10% coating, respectively. The surface morphology and roughness were further examined. As shown in the SEM images (Figure 4a), similar to the blank Si wafer, all polymer coatings exhibited relatively smooth and uniform surfaces, beneficial for improving the antifouling performance of the substrates. Moreover, the 3D AFM images for various polymer coatings revealed the low roughness value of Ra = 0.37 ± 0.03 nm for Si substrates (Figure 4c). By contrast, the PTCSA polymer coating exhibited the highest surface roughness with a value of Ra = 5.56 ± 0.93 nm. The roughness values for the PTMAO, PTT 12:1 , and PTTU-10% polymer coatings were Ra = 0.27 ± 0.01, 0.54 ± 0.05, and 0.40 ± 0.07 nm, respectively (Table S4). These results show that the PTTU coating should not alter the surface morphology and roughness of the substrate, which is beneficial to maintain the surface activity of the special topographic substrate. The surface element distribution and composition were examined using energy-dispersive X-ray spectroscopy (EDS) mapping. Homogeneous distribution of N, Cl, C, and O was observed for the corresponding polymer coatings, indicating the uniform composition of coatings (Figure 4b and Figure S8).
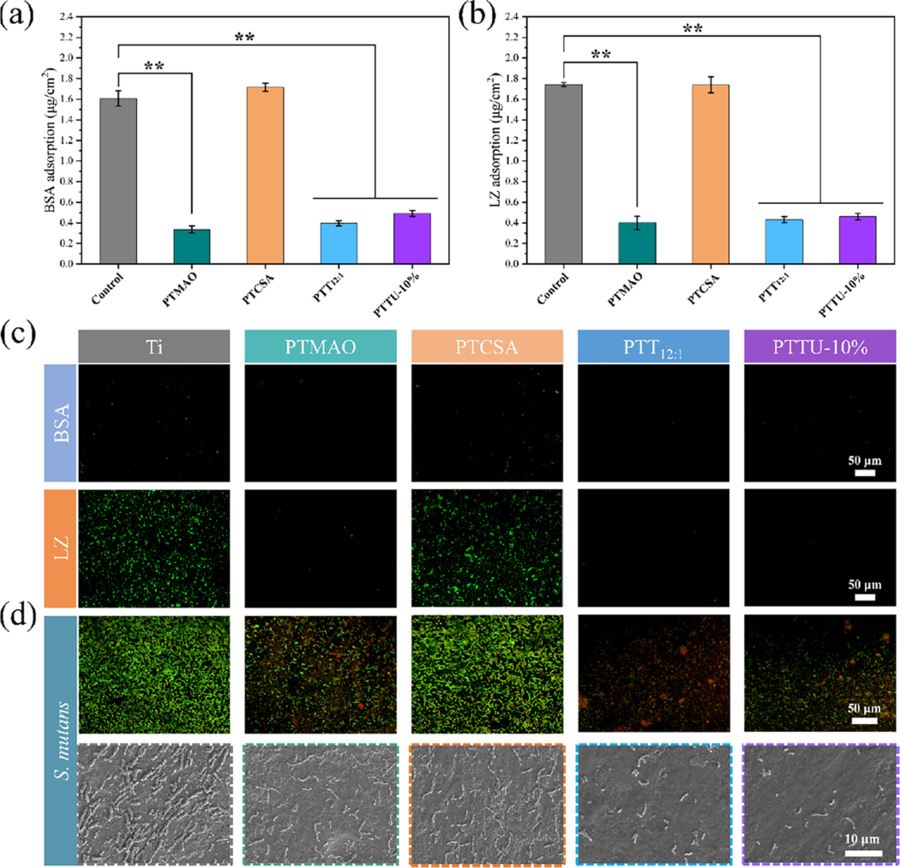
Figure 4 Surface morphology and roughness. (a) SEM images and (b) elemental mappings of N and Cl of bare Si wafer, PTMAO, PTCSA, PTT12:1, and PTTU-10% coating, respectively. (c) 2D and 3D surface profiles of the bare Si wafer, PTMAO, PTCSA, PTT12:1, and PTTU-10% coating, respectively. 3.3 Multifunctionality of PTTU Coating In the complex oral environment, saliva proteins and other molecules rapidly adsorb onto the material surfaces, creating binding sites for nonspecific molecules, including bacteria. This adsorption can interfere with the active groups of the materials, ultimately affecting their efficiencies and longevity. In this sense, the construction of antifouling surfaces is a feasible solution. To verify the versatility of the PPTU polymer coating, the most commonly used implant metal Ti was chosen for modification. The antifouling performance of PTTU coatings was first evaluated by selecting bovine serum albumin (BSA) and lysozyme (LZ) as the model proteins. The results in Figure 5a,b indicate that the surfaces of blank Ti and PTCSA coatings exhibited significant adhesion of BSA and LZ. In contrast, PTMAO, PTT 12:1 , and PTTU-10% polymer coatings, incorporating hydrophilic zwitterionic monomers like TMAO, the protein adsorption decreased more than 70% for both BSA and LZ, which is probably ascribed to the hydration of the zwitterionic polymer as evidenced by the WCA results, which effectively inhibited the adhesion of BSA and LZ on their surfaces. To visually demonstrate the antiprotein adhesion performance of the various polymer coatings, the coatings were immersed in a 1 mg/mL FITC-BSA and FITC-LZ solution. After 2 h of adsorption at 37 °C, followed by three PBS washes, confocal laser scanning microscopy (CLSM) was employed to observe the adhesion of proteins on the surface. All polymer coatings containing hydrophilic zwitterionic monomers, except for the blank Ti and PTCSA coatings, exhibited excellent antiprotein adhesion properties, aligning with the BCA assay results (Figure 5c). Subsequently, the antibacterial performance of the PTTU polymer coatings was investigated. The coatings were immersed in a bacterial suspension and incubated under anaerobic conditions for 48 h. Surface bacterial quantities were evaluated by using CLSM and SEM observations. Figure 5d illustrates that the surfaces of blank Ti and PTCSA coatings accumulated a significant number of S. mutans bacteria by lacking hydrophilic antifouling/antibacterial moieties. Although the PTMAO coating surface exhibited excellent hydrophilicity and antiprotein adhesion properties, it still showed significant bacterial adhesion. This may be due to the absence of bactericidal moieties on the surface of PTMAO coating, which can effectively inhibit the initial adhesion of bacteria, but with the prolongation of time, the bacteria adhering to the surface will secrete some toxic factors to induce more bacteria to adhere to the surface. The PTT 12:1 and PTTU-10% coatings significantly inhibited the number of bacteria adhering to their surfaces compared to the blank Ti sheets due to the synergistic effect of antifouling and bactericidal properties. Collectively, the PTTU polymer coating has good antibacterial and antifouling properties and can effectively inhibit the nonspecific adhesion of oral proteins and bacteria, etc., thereby inhibiting the formation and development of biofilms and preventing biofilm-related oral diseases.
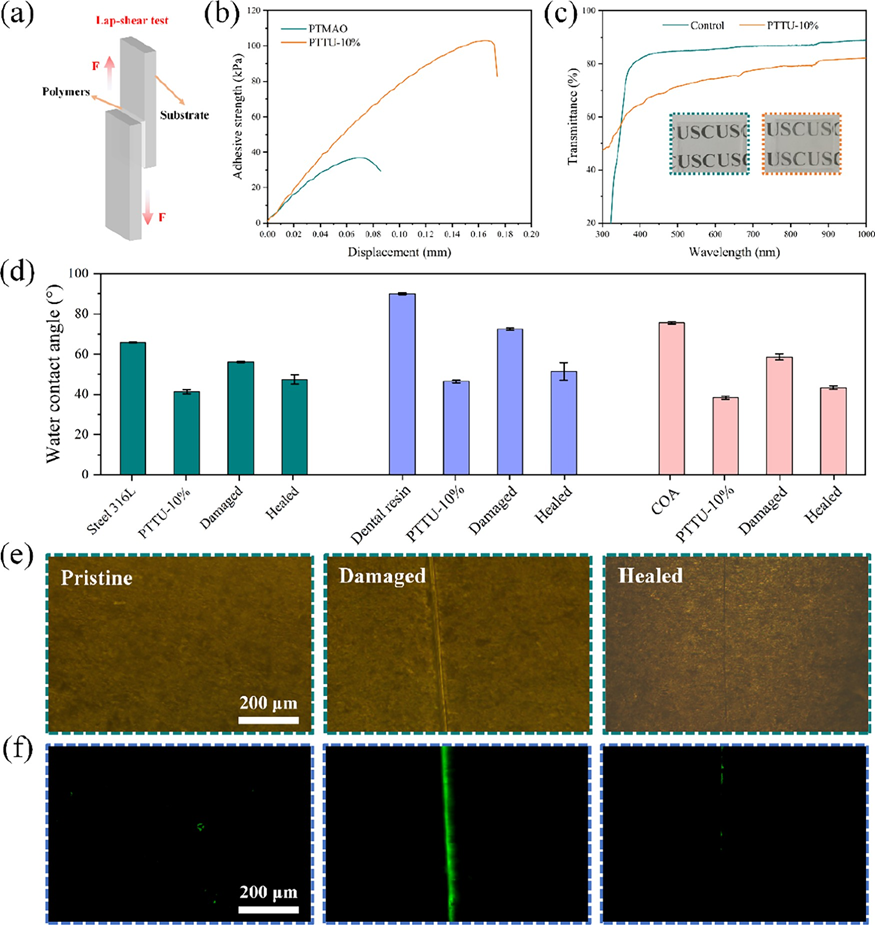
Figure 5 Antifouling property of various coatings. (a) Adhesion of BSA and (b) LZ on the bare Ti sheet and various polymer coatings, respectively. (c) Fluorescence images of FITC-BSA and FITC-LZ adhesion on Ti sheet and various polymer coatings, respectively. (d) Fluorescence images of live/dead staining of bacteria and the corresponding SEM morphology on bare Ti sheet and various polymer coatings. The strong interfacial adhesion of the coating with the dental appliance is essential for the long-lasting functionality. The supramolecular bonds, including hydrogen bonds and ionic bonds, present in the PTTU polymer chains facilitate increased interfacial bonding to the substrate. The adhesion of PTTU-10% to Ti substrates and its stability in a bacterial environment were evaluated by using the ASTM D1002 test standard and antibacterial zone experiments (Figure 6a). The results are shown in Figure 6b, where the shear stress of the PTTU-10% polymer on the Ti substrate was measured to be 102.84 kPa, approximately three times higher than the shear stress of PTMAO on the Ti substrate (36.77 kPa). These results demonstrate the strong adhesion of PTTU to the titanium metal substrates. Additionally, the inverted polymer coatings were placed on agar plates containing S. mutans bacteria and coincubated under anaerobic conditions for 24 h to evaluate their antibacterial activity. No antibacterial zones were observed around any of the coatings, indicating that all of the coatings possessed contact-killing antibacterial properties. This means that they do not release antibacterial agents that could be depleted in the bacterial environment, thus facilitating the maintenance of stable antimicrobial activity on the material surface (Figure S9). Compared to metal and ceramic braces, clear overlay appliances (COA) are highly favored by patients due to their excellent transparency and aesthetics. Therefore, the impact of PTTU coating on the transparency and surface antifouling properties of COA was investigated. As shown in Figure 6c, after application of the PTTU coating on the dental film surface, it maintained good transparency within the wavelength range of 300?800 nm, without compromising its aesthetic appearance. Furthermore, the dental films with PTTU polymer coating were immersed in the BHI culture medium containing S. mutans (1 × 10 6 CFU/mL) for 4 h. The results showed that the blank film surface adhered to a large number of green live bacteria, while the PTTU-coated film exhibited only a minimal amount of bacteria adhesion, demonstrating excellent antibacterial properties (Figure S10). These findings indicate that PTTU polymer coating improves the surface antifouling properties of dental films without compromising their transparent and aesthetic nature, thereby reducing the risk of pathogenic bacteria adhesion and preventing bacterial infectious oral diseases. Molecular chain motility is closely related to the self-healing properties of the polymer network. Therefore, the thermal and crystalline behaviors of various polymer networks were analyzed using differential scanning calorimetry (DSC). The results show that, except for PTCSA, which exhibited a lower melting point (around 50 °C), PTMAO, PTCSA, and PTTU10% displayed melting points in the range of 90?120 °C, indicating their good stability at oral temperature. Furthermore, with the introduction of UPy units, the phase transition temperatures gradually decreased along with a decrease in the total enthalpy. This can be attributed to the amorphous nature of the UPy units, which hindered the crystallization of PTMAO and PTCSA through self-complementary hydrogen bonding interactions. The glass transition temperature (Tg) is the lowest temperature at which molecular chain motion occurs and is closely associated with the self-healing properties of materials. The Tg values of PTMAO, PTCSA, PTT 12:1 , and PTTU-10% were ?7.68, 8.88, ?7.50, and 4.76 °C, respectively (Figure S11). All Tg values were below room temperature, indicating that these polymers may exhibit a favorable self-healing performance under ambient conditions. Subsequently, the polymer coatings were constructed on the surfaces of various dental materials, and their self-healing ability was evaluated by examining the surface pro/hydrophobic properties of the coatings before and after self-healing. The results showed that all three dental materials improved their surface hydrophilicity after modification with PTTU-10%. The hydrophilicity of the coatings is decreased when damaged, and then basically recovered by the interaction of supramolecular bonds in the coating, indicating that PTTU-10% coating possesses a universal self-healing capability (Figure 6d). To visually investigate the self-healing performance of PTTU polymer coatings in the oral environment, the surfaces of PTTU-10% coatings were deliberately scratched by using a scalpel. Subsequently, the samples were immersed in an SSF at 37 °C to simulate the oral environment, and the healing of surface cracks was observed using optical microscopy. As shown in Figure 6e, the pristine PTTU-10% coatings were relatively smooth, while upon gently scratching their surfaces with a scalpel, a noticeable crack appeared. After immersing the samples in SSF for 2 h, the PTTU-10% coating, which contained supramolecular bonds and a lower Tg, exhibited enhanced molecular chain mobility under the 37 °C liquid environment, enabling rapid repair of the surface crack and restoration of its original morphology. Subsequently, these coatings were immersed in FITC-BSA solution for 30 min, and the antifouling capability was examined by fluorescence microscopy before and after self-healing. As can be seen in Figure 6f, the original coating exhibited excellent antiprotein adhesion properties, while the cracks disrupted the surface structure of the coating increasing the surface roughness and decreasing the hydrophilicity, thus adsorbing a large amount of protein. Almost no adherent proteins were found on the repaired coating. The above results demonstrated that the PTTU-10% coating could achieve the recovery of antibiofouling performance in the oral environment.
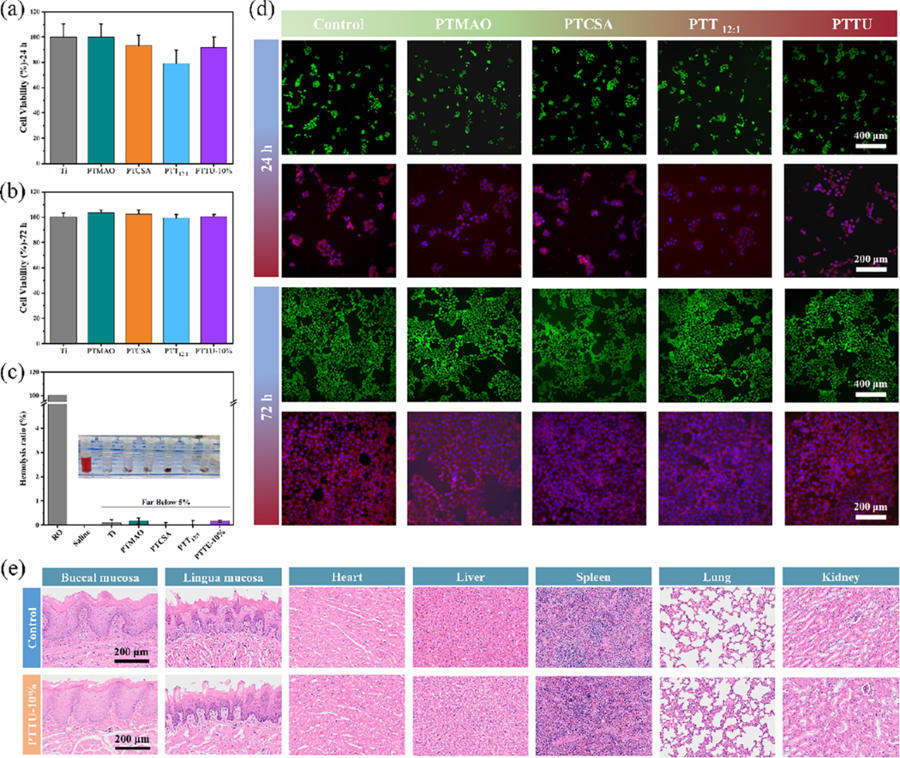
Figure 6 Self-healing property of polymer coatings on a variety of materials. (a) Schematic of the lap-shear adhesion test. (b) Tensile?strain curves of the Ti sheets bonded with PTMAO and PTTU-10%. (c) Transmittance of substrates before and after modification under a wavelength of 300? 1000 nm. (d) Water contact angle of various dental materials before and after self-healing. (e) Optical micrographs showing scratches and (f) BSA adhesion on the PTTU-10% coatings before and after healing in SSF at 37 °C. 3.4 Biocompatibility Evaluation The polymer structure and composition significantly influence cell viability. Therefore, the biocompatibility of polymer coatings is a crucial requirement for their biomedical applications. Initially, human oral keratinocyte (HOK) cells were seeded in a 48-well plate and incubated overnight in a cell culture incubator. Subsequently, the cells were cocultured with various polymer coatings for 24 and 72 h to estimate the cell viability. As shown in Figure 7a,b, the cell viability for all groups was higher than 80%, indicating excellent biocompatibility of PTTU polymer coatings. Furthermore, FDA/PI solution was used for cell viability staining. After washing with PBS for 3 times, the samples were quickly placed under a fluorescence microscope for observation and imaging. The fluorescence images of cells cocultured with various coatings showed predominantly green fluorescence, indicating live cells, with no red fluorescence observed (no dead cells) (Figure 7d). This suggests that all coatings were nontoxic to the cells. Subsequently, cells were stained with FITC-labeled phalloidin (red) for cytoskeletal staining and DAPI (blue) for nuclear staining. The results revealed well-spread cells in all groups, and cell numbers increased gradually with time, indicating the excellent cell compatibility of the PTTU polymer coatings. Furthermore, when the polymer coatings were incubated with a 2% whole blood solution diluted in physiological saline for 2 h, the hemolysis rate of the PTTU polymer coatings was less than 5%, meeting the standards for biomedical materials (Figure 7c). These findings demonstrate the excellent biocompatibility of PTTU polymer coatings, providing assurance of their applications in the field of biomedical materials. Subsequently, we evaluated the biosafety of PTTU-10% in vivo with the previous method. The body weight of SD rats in both the control and PTTU-10% groups increased steadily during the experimental period with no significant differences (Figure S12). No oral buccal/lingual mucosal lesions were found in the H&E tissue staining, and no significant inflammation or other lesions were observed. Moreover, the blood routine indexes of SD rats were within the normal range, and no abnormalities were found in their heart, liver, spleen, lung, and kidney H&E staining, indicating that PTTU-10% has a good biosafety in vivo (Figure 7e, Table S5).
Figure 7 Assessments of the biocompatibility of polymers. Cell viability of human oral keratinocytes cells on blank Ti sheet and various polymer coatings for (a) 24 h and (b) 72 h. (c) Hemolysis ratio and corresponding picture of blank Ti sheet and various polymer coatings. (d) Fluorescence microscopy images of live/dead staining and morphological staining of HOK cells incubated after 24 and 72 h. PTT12:1 and PTTU-10% coating, respectively. (e) The H&E staining images of buccal mucosa, lingual mucosa, heart, liver, spleen, lung, and kidney of rats of the Control and PTTU- 10% groups. 4. Conclusions To inhibit the development of a biofilm resulting from the adhesion of biofouling on the surface of orthodontic appliances, this study developed a self-healing functional polymer coating applicable to various dental appliances by simple spin-coating. The polymer, namely, PTTU, was synthesized by TMAO, TCSA, and UPyMA. The hydrophilic zwitterionic moiety, TMAO, incorporated into the PTTU coating imparted remarkable hydrophilicity, leading to the formation of a hydrated layer that inhibited the adhesion of biofouling (antiprotein adhesion rate above 70%). The presence of the antibacterial unit, TCS, endowed the polymer with outstanding bactericidal performance (MIC = 32.5 μg/ mL). The PTTU coating contained supramolecular bonds, such as hydrogen bonds and ionic interactions, which conferred inherent self-healing capabilities in the oral environment. On basis of this, the coatings could restore its morphology and antibiofouling performance, thus achieving sustainable antifouling properties. Furthermore, the PTTU polymer coating exhibited excellent biocompatibility, making it highly promising for applications in the field of biomedical materials and not merely in dentistry. ■ ASSOCIATED CONTENT Supporting Information The Supporting Information is available free of charge at https://pubs.acs.org/doi/10.1021/acsami.3c09628. Synthesis process and high-resolution mass spectra of TCSA, UPyMA, and TMAO; antibacterial activities of TCSA at different concentrations; 1 H NMR and FTIR spectra of PTTx polymers, PTTU-20% and PTTU-30%; as well as UV?vis spectra of UPyMA, PTTU-20%, and PTTU-30%; C 1s, O 1s high-resolution spectra and elemental mappings of C, O of various coatings; images of inhibition zone development against S. mutans bacteria for various coatings; antifouling properties of PTTU-10% coating on COA; DSC curves of various polymers; the body weights of rats during the experimental period; (Table S1) antimicrobial properties of PTCSA, PTMAO, and PTT polymers and the surface water contact angle of their corresponding polymer coatings; (Table S2) MIC and MBC of PTTU polymers; (Table S3) surface elemental composition and content of various coatings; (Table S4) surface roughness of various coatings; and (Table S5) blood routine analysis of PTTU-10% in vivo (PDF) ■ AUTHOR INFORMATION Corresponding Authors Chunmei Ding ? College of Polymer Science and Engineering, State Key Laboratory of Polymer Materials Engineering, Sichuan University, Chengdu 610065, China; orcid.org/ 0000-0002-7399-3459; Email: dingcm@scu.edu.cn Jianshu Lee ? College of Polymer Science and Engineering, State Key Laboratory of Polymer Materials Engineering, Sichuan University, Chengdu 610065, China; State Key Laboratory of Oral Diseases, West China Hospital of Stomatology and Med-X Center for Materials, Sichuan University, Chengdu 610041, China; orcid.org/0000-0002-1522-7326; Email: jianshu_li@scu.edu.cn ■ ACKNOWLEDGMENTS This work was supported by the National Natural Science Foundation of China (U22A20158, 51973133, 51903175, and 52073191), and the State Key Laboratory of Polymer Materials Engineering (Grant No.: sklpme 2023-2-02). Source: www.acsami.org |