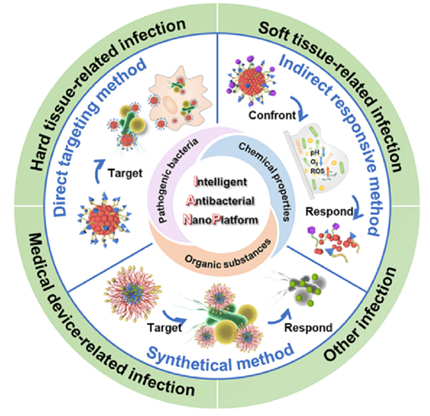
Recent Progress of Intelligent Antibacterial Nanoplatforms for Treating Bacterial InfectionIssuing time:2025-08-01 09:40 Hui Sun, Mingyang Sun, Yuhao You, Jing Xie, Xinyuan Xu, Jianshu Lee Abstract Bacterial infection has been a severe concern of global health which seriously threatens human lives. Particularly, drug-resistant bacteria and protective biofilm pose nonnegligible challenges for traditional antibiotics therapy. Moreover, no newly-developed antibacterial materials entered the clinical trials so far since there is potential biosafety issue associated. Emerging intelligent antibacterial nanoplatforms (IANPs) with targeting or responsive capability have attracted enormous interests due to their favorable biocompatibility and on-demand and precise therapy against bacterial infection. This review firstly introduced the inherent characterizations of bacteria, and organic substances and chemical properties of the bacterial infection microenvironment, which are the origin for the design of IANPs. Then, according to the function process of IANPs, the intelligent antibacterial principles of IANPs are classified into direct targeting strategy and/or indirect responsive method. The construction of IANPs with different materials sources (such as polymer, inorganic materials, biological materials) were highlighted. The applications of IANPs to treat various bacterial infection-related diseases were also summarized, including hard tissue-related infection, soft tissue-related infection, medical devices-related infection, and others. Finally, future perspectives associated with the biosafety, function, technology of IANPs were discussed hoping to advance the development of novel IANPs for treating bacterial infection. 1. Introduction Infectious diseases caused by pathogenic bacteria have caused numerous human deaths in the past decades, severely increasing the burden of public health. A very recent study from the Lancet reported that there were 7.7 million deaths associated with bacterial pathogens out of 13.7 million infection-related deaths and accounted for 13.6% of all global deaths in 2019. Since the birth of penicillin in 1928, antibiotics therapy against bacterial infection has been clinically popularized worldwide and has saved the lives of many people. However, not only certain shortcomings such as non-specific target, short half-life, low solubility, fluctuations in drug concentration greatly limit their further application, but also the emergence and spread of drug-resistant bacteria and biofilm formation pose new nonnegligible challenges. In the past ten years, research fellows have put tremendous efforts to develop novel antibacterial strategies including discovering novel antimicrobial agents, and antibiotics-free approaches (e.g., photothermal therapy (PTT), photodynamic therapy (PDT), chemodynamical therapy (CDT), gas delivery therapy, designable surface strategy, sonodynamic therapy,). Despite the above-mentioned progress, the contradiction between the antibacterial efficiency and biocompatibility is presented and not easily reconciled from the perspective of bacteria-killing mechanism. Due to the certain similarity in chemical composition and structure of bacteria and mammal cells, the majority of antibacterial approaches such as interacting with membrane components or intracellular substances and reducing the proliferation of bacteria by reactive oxygen species (ROS) manifests analogous effect on mammal cells, leading to the risk of biocompatibility and restricting their transformation towards clinical use. Similar to the problem of drug-resistance caused by antibiotics, bacterial resistance against other antibacterial agents (e.g., metal ions) is also increasingly emerging. Therefore, both the bacterial resistance and biocompatibility risk raise urgent requirement on the development of effective and non-toxic antibacterial strategies. Intelligent antibacterial strategies possess great potential in effectively eliminating infection and reducing normal tissue damage. Rational design of antibacterial materials based on the inherent features of bacteria and the microenvironment of bacterial infection has been considered as one of the sally ports to reconcile the demand of bacteria- killing and biocompatibility. Bacteria are generally coated with a cytoplasmic membrane and a cell wall that are decorated with specific biomolecules such as lipopolysaccharide (LPS) and peptidoglycan (PGN). Various biological molecules (e.g., exotoxins, proteins, polysaccharides, nucleic acids) are secreted by the bacteria and the hosts to promote invasion and proceed innate defense respectively. Moreover, the chemical property of bacterial infection microenvironment features in changes of oxygen concentration, pH value and redox potential. On account of these characterizations of bacterial infection, intelligent antibacterial nanoplatforms (IANPs) with adjustable composition, varied structure, and targeting/responsive approaches emerge in the right moment. IANPs are designed to possess specific targeting capability to bacteria/infection sites or bacterial infection-responsive capability and serve as a universal platform for the purpose of precise target of pathogenic bacteria/infection sites and protection of mammal cells and normal tissue. On the one hand, advanced fabrication technology allows the integration of multifunctional organic/inorganic components into the IANPs resulting in the effective antibacterial mechanism. On the other hand, diverse building blocks could be conjugated onto the surface of IANPs or loaded inside the IANPs to realize precise target of specific bacterial infection-related biomolecules or responsive transformation of physicochemical structure of IANPs. Thus, IANPs with self-adaptive antibacterial behavior show high-efficiency elimination of pathogenic bacteria and rare toxicity to host cells. However, few reviews summarize the origin, design principle, and application of IANPs. In this review, the design principles of IANPs were firstly introduced, including target the bacteria, target or respond to the biological molecules by bacteria or hosts, and response to the chemical property of bacterial infection. Then, IANPs with different materials sources were classified into direct target method, indirect responsive method, and synthetical method. At last, the applications of IANPs to treat various bacterial infection-related diseases were summarized. Various reviews have reported the development of responsive antibacterial strategies from different focuses, such as infection-related nanotherapeutic strategies, biofilm-responsive nanoparticles (NPs), nanotechnology-based strategies/agents, targeted delivery of antibiotics by macromolecular approaches, bacteria-responsive materials, etc. This review placed emphasis on the intelligent antibacterial mechanism of integrated nanoplatforms derived from the characterizations of bacteria and infection and paid attention to their biocompatible performance and application to treat bacterial infection- related diseases (Scheme 1). We hope that this review would provide inspirations for the design and development of IANPs to treat bacterial infection in the future.
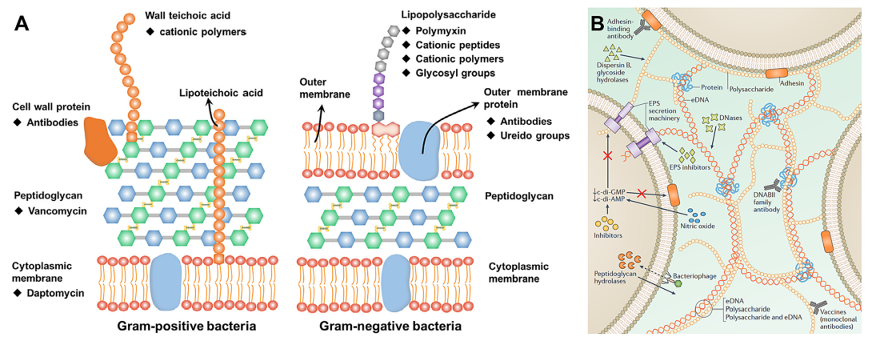
Scheme 1. Overview of IANPs: design origins, intelligent antibacterial methods, and applications. 2. Characterization of infection environment Elaborately manipulated IANPs based on the distinctive features of infection microenvironment have been applied to treat bacterial infection-related diseases due to their intelligent antibacterial mechanism. We divided these features into three aspects: 1) bacteria; 2) organic substances consisting of bacterial secretion and hosts’ immunoreaction and 3) chemical property of infection microenvironment which will be discussed in this section. 2.1. The bacteria Maintaining the integrality of cell wall and cytoplasmic membrane, the protective layers of bacteria, is essential for bacterial survival. As a result, various methods that target and interact with the cell wall and cytoplasmic membrane are highly efficient in destroying them, causing the death of bacteria (Fig. 1A). For Gram-positive (G+) bacteria, the outer layer is the cell wall mainly comprising of PGNs, proteins, and teichoic acids. PGNs are composed of glycan chains and peptide chains, leading to a highly crosslinked network. Vancomycin (Van) was proved to specifically bind the D-Alanyl-D-Alanine dipeptide of peptide chains of PGNs, serving as a classic and effective approach to targeting G +bacteria. There are some proteins on the cell wall of bacteria that could specifically interact with binding fragments of antibodies and that act as target sites on the G + bacteria. For example, bacteriophage SPP1 could specifically bind Bacillus subtilis in an irreversible way with a yueB-coded membrane protein as receptor. In addition, wall teichoic acid, a negatively charged glycopolymer on the bacterial cell surface, accounts for the most abundant substances in bacterial cell envelope. Positively charged materials could bind the G + bacteria via electrostatic interaction with wall teichoic acid. For Gram-negative (G-) bacteria, the bacterial cytoplasm is protected by a sandwich-shaped multilayer structure with the cytoplasmic membrane as outer layer and the cell wall as inner layer. LPS is a characteristic chemical on the outer layer of G- bacteria, consisting of lipid A, core oligosaccharide, and distal polysaccharide. The lipid part penetrates into the cytoplasmic membranes while the polysaccharide part is exposed outside the bacterial cell, which is considered as a specific target of G- bacteria. The phosphoryl substituents in the core oligosaccharide are a common specialty found in G- bacteria, which could be targeted by positively charged polymers such as poly-L-lysine (PLL), chitosan, polyethyleneimine, etc.. Polymyxins could bind with LPS by replacing the calcium ions and magnesium ions of phosphoryl substituent while they are clinically in discontinuation due to the nephrotoxicity and neurotoxicity. The abundant glycosyl groups of LPS are targets on the G- bacteria as well. The glycans could bind LPS through high affinity carbohydrate-carbohydrate interactions and then target the G- bacteria. Moreover, the covalent bond formed between diol structure of LPS and boron dihydroxy group-containing substances is also a common way of targeting G- bacteria. Since LPS is a characteristic endotoxin of G-bacteria, it could be specifically recognized and bound by immune cells through pattern recognition receptor (PRR). In addition, various proteins on the outer layer of G- bacteria can server as target sites and be recognized by specific antibodies. For example, LPS transport proteins which paly important role in the mediation of LPS transportation to the cell surface could be targeted by the hairpin-shaped cationic antimicrobial peptide. Another typical example is the FimH, a bacterial adhesin located at the tip of type I fimbria of Escherichia coli (E. coli), which shows a specific binding capability with mannose. Piliation system is an important protein nanomachine located on the surface of bacteria which paly irreplaceable roles in bacteria survival and pathogenicity. Bacterial flagellum with dozens of microns takes charge of providing moving power for most bacteria. Bacterial pilus with several microns contribute to adhering on cellular surfaces and transferring genetic materials which are lately considered as a target site for targeting bacteria [49]. For example, Wu et al. reported a nanogenerator with the capability of targeting Pseudomonas aeruginosa (P. aeruginosa) though reaction between maleimide and type IV pili which showed highly efficient antibacterial function by NO delivery and ROS generation. Bacterial adhesins on the pili of G + bacteria have also served as targets. Shan et al. synthesized a glycopolymer carrying glucose and mannose residues which exhibited high affinity towards FimH presented on the pili of E. coli.
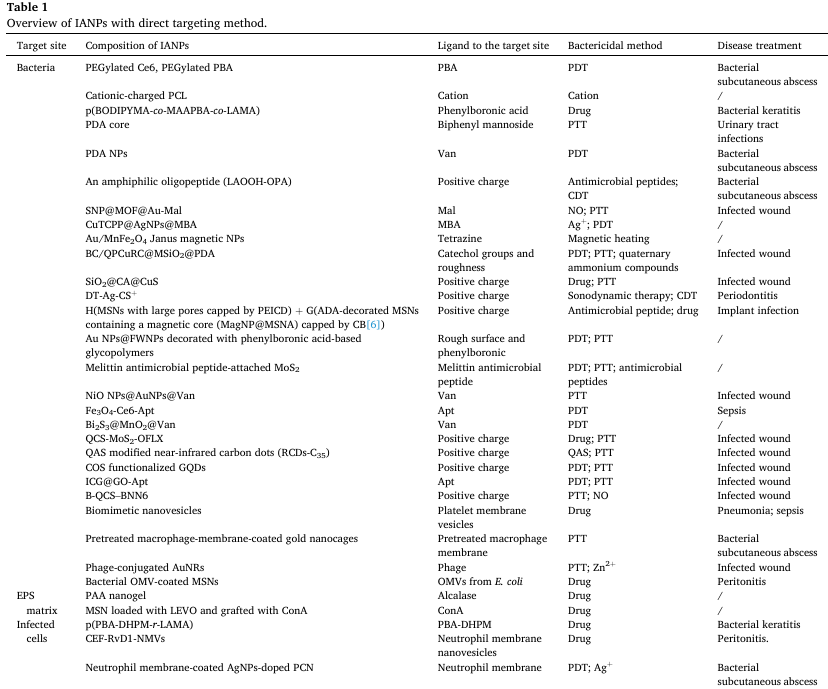
Fig. 1. (A) Structures of bacterial cell envelopes and target sites for IANPs. Reproduced with permission. Copyright 2022, Elsevier. (B) The component of EPS matrix in biofilm. Reproduced with permission. Copyright 2017, Springer Nature. 2.2. The organic substances The organic substances present in the infection microenvironment can be attributed to two sources: bacterial secretory product and host immunoregulatory product. Bacterial secretory products include extracellular polysaccharides, proteins, and nucleic acids, forming a membranous complex, i.e., extracellular polymeric substance (EPS) matrix of biofilm, which is mechanically stability and protects the bacteria from antibiotics and attack of host’s immune system (Fig. 1B). Host immunoregulatory products are a series of substance secreted by the host to clear bacterial infection, including receptors and inflammatory factors. 2.2.1. Bacterial secretory products Extracellular polysaccharides are one of the main bacterial products that contribute to the biofilm formation, water retention and cell protection. The sugar residues and substitutes on the extracellular polysaccharides confer it charged properties. The sugar residues such as glucuronic acid, galacturonic acid and mannuronic acid, and the substitutes of the sugar residues such as carboxyl groups, sulfate groups and phosphate groups endow the extracellular polysaccharides with polyanion nature. Consequently, positively charged IANPs have strong affinity to the EPS matrix due to opposite charge attraction. For example, Cheng et al. reported a guanidinium-rich polymer decorated nanoplatform that is positively charged and shows enhanced penetration into biofilm matrix. In addition, extracellular polysaccharides- degrading enzymes could disperse the polysaccharides matrix which is an effective way of targeting biofilm. Although glycoside hydrolases are secreted by the embedded bacteria and function for regulation of biofilm microenvironment, they are mostly investigated for dispersing extracellular polysaccharides. Among these enzymes, dispersin B (β-N-acetylglucosaminidase) is a widely reported exopolysaccharide-degrading enzyme that could target and hydrolyze the β-(1, 6)-glycosidic bonds of poly-β-(1, 6)-N-acetyl-D-glucosamine (PNGA) produced by various bacteria strains. Alginate is a main component of EPS matrix of P. aeruginosa, a common infection- associated pathogenic bacteria, while alginate lyase found in their biof ilm could degrade alginate and then detach the cells in the biofilm. Recently, more glycoside hydrolases that degrade exopolysaccharides of bacterial biofilm are isolated which could be integrated into the IANPs for specific targeting and removing biofilm. For example, Wan et al. reported an alginate lyase-combined silver nanocomposite that showed enhanced inhibition and disassemble effects on P. aeruginosa biofilm. Proteins are one of the main components of EPS matrix, and even account for more mass ratio than exopolysaccharides in some biofilms. Insoluble amyloids with a repeated β-sheet structure are a common form of proteins in biofilm which serve as effective sites for dispersing EPS matrix. Certain molecules such as polyphenol, antibody, and graphene quantum dot have been found to possess significant disassemble capabilities for amyloid fibers, allowing them to effectively disperse biofilm. The proteins contributed to the structural stability of extracellular deoxyribonucleic acid (eDNA) is called the DNABII family of DNA-binding proteins, which are composed of two kinds of proteins: integration host factor and histone-like protein. The DNABII proteins could bind and bend the eDNA due to their homologous nucleic acid sequence and three-dimensional conformation in the binding region, which shows high affinity and specificity. The absence of DNABII proteins in the biofilm would result in the instability of eDNA structure and disassembly of biofilm matrix. Antibodies against DNABII proteins have been demonstrated to have the ability of collapsing bacterial biofilm in a rapid and broad-spectrum way. Multifarious enzymes are secreted during bacterial metabolism and dynamic variation of biofilm component whose catalytic process shows high selectivity and mildness. Thereby, these enzymes are considered as design origin for bacteria-responsive IANPs. The common bacterial enzymes include lipases, hyaluronidase (HAase), gelatinase, and phospholipase which are used to either digest macromolecules into small molecules for easy internalization or degrade host defense substances for survival and invasion. IANPs containing specific bond or chemicals could response to these enzymes for intelligent and biocompatible antibacterial therapy. ?For example, Liu et al. prepared a pH/ hyaluronidase dual responsive nanocomplex which displayed strong surface adsorption on Staphylococcus aureus (S. aureus) and showed enhanced anti-S. aureus effect through HAase-responsive release of antibiotics. eDNA: The role of eDNA in the nutrition source and structure support of biofilm is similar to that of extracellular polysaccharide, except for that it has the function of gene information transmission. Thus, damaging the structure of eDNA of biofilm is also an important way of disrupting biofilm. Here, we mainly focus on the substances effecting eDNA itself. Deoxyribonuclease I (DNase I) has been proved to destabilize emerging or mature biofilm through degrading eDNA. The phosphoester bonds of eDNA could be cleaved by nucleophiles under the catalytic action of DNase I. Destroying the structure of eDNA not only contributes to dispersing EPS matrix, but also cuts down the communication among bacterial communities, which benefits the removal of biofilm. In addition, the negative charge of eDNA, due to the phosphate backbone and other substitutes of eDNA, facilitates their high affinity with positively charged substances. 2.2.2. Immunoregulatory products of host Inflammation is an adaptive response from host immune system involving infection recognition of phagocytes, secretion of inflammatory mediators, and phagocytizing pathogen of recruited leukocytes. In the process of bacterial infection-caused inflammation, a variety of receptors on cell membrane is highly expressed and all sorts of inflammatory factors are secreted for accelerating sterilization and tissue repair. These features associated with inflammation could act as targets of novel IANPs to precisely treat bacterial infection. PRRs on immune cells are highly expressed when pathogens invade. The clusters of differentiation 44 (CD44) on activated macrophages contributes to mediating the cell migration by acting as the receptor of hyaluronic acid (HA), a component of extracellular matrix (ECM). Similarly, CD206 is also a highly expressed receptor secreted by partial innate immune cells and nonvascular endotheliocytes meanwhile is the receptor of mannose. The expression of intercellular cell adhesion molecule-1 (ICAM-1) on endotheliocytes is promoted to enhance leukocyte migration and activation. ICAM-1 could be specifically bound by its binding molecules to realize infection targeting. Thus, IANPs incorporated with inflammation-targeting molecules have the ability of targeting infection site. For example, Montanari et al. reported a HA- based nanohydrogel targeting intracellular S. aureus through high- affinity HA-CD44 interaction. In addition, various pro- inflammatory cytokines such as interleukin-1 (IL-1), IL-6, and tumor necrosis factorα (TNFα ) are potential targets for IANPs, such as the “cytokine blocker” nanoplatform base on macrophage cell membrane for bone tissue repair. 2.3. The chemical properties Once the pathogenic bacteria find a place of human body to grow and proliferate, the bacterial metabolic products and released toxins and human immune reaction products together form a complex and distinctive microenvironment. These products make the chemical and biological properties of bacterial infection greatly different from that of physiological condition. The chemical properties of bacterial infection microenvironment and their inspiration for designing IANPs will be discussed below. 2.3.1. Hypoxia and pH As mentioned in section 2.2, biofilm provides a compact and protective layer for encapsulated bacteria, where the O 2 increasing with the depth of biofilm. However, O 2 consumption is diffusion is blocked by the compact EPS matrix and the diffusion rate is lower than the consumption rate, which results in the hypoxia condition within the biofilm. The hypoxia condition strengthens the bacterial anaerobic metabolism which produces acid compounds such as pyruvic acid and acetic acid, causing the pH reduction of the microenvironment (pH 5.0–6.0) of infection site. In particular circumstances, such as the oral biofilm, the pH value could reach around 4.5 or even lower with the adequate supply of carbohydrates (e.g., sucrose, starch, etc.). Besides, bacterial retention condition (e.g., lysosome/phagolysosome of intracellular infection) also leads to an acid microenvironment. Hence, hypoxia and pH value lower than that of physiological condition could act as triggers for IANPs respectively or jointly. For example, Xiu et al. reported a MnO 2 responsive release of Mn 2+ nanosheets-based nanoplatform with pH- and oxygen-generating capability to relive hypoxia condition of biofilm. 2.3.2. Oxidative stress Oxidation-reduction (redox) balance in living cells provides a stable microenvironment for the normal function of biological macromolecules and is a significant basis for the normal physiological function of cells. Regulating redox balance involves diverse small and macro molecules, including reactive species such as ROS and reactive nitrogen species (RNS), antioxidant enzymes such as superoxide dismutase (SOD) and catalase (CAT), and redox couples such as glutathione/glutathione disulfide (GSH/GSSG). Once the bacteria invade into the hosts, a series of immune responses will be activated which will disequilibrate the redox balance and cause oxidative stress resulting in the change of redox potential of bacterial infection microenvironment. Excessive ROS level is a representative of oxidative stress such as redundant hydrogen peroxide (H 1 2 O 2 ), singlet oxygen ( (?OH), superoxide anion (O 2O 2 ), hydroxyl radical ), etc., which not only come from the hosts’ immune response but also are generated by the invading bacteria themselves for further infection. Although the bacteria are attacked by the oxidants secreted by phagocytes, they have inherent detoxification methods and inducible systems to limit the oxidant stress [96]. Bacterial secretion of low-molecular-weight thiols (such as GSH) containing a reactive sulfydryl that consumes ROS by thiol-disulfide exchange helps maintain the reduced state of cytoplasm. Although the redox change of bacterial infection microenvironment is a complicated process and varies from pathogenic bacteria species, infection site and progress, and condition of host immune system, these intricacy cues provide potential and extensive space for designing various IANPs. Overall, the oxidative stress caused by excessive ROS level has been served as valuable factors to design IANPs which could either be deconstructed to release payloads or consume redundant ROS to regulate redox balance when exposed to infection microenvironment. For example, Yang et al. reported a one-for-all bactericidal nanoplatform in which GSH is utilized to deplete endogenous H 2 O 2 and generate ?OH for eliminating bacteria and restoring local redox balance. 3. Design principles of IANPs The characteristic microenvironment of bacterial infection provides diverse target sites and responsive methods for designing intelligent antibacterial materials. Various IANPs including polymer, inorganic, organic–inorganic hybrid, and biohybrid materials have been developed based on the microenvironment-inspired design principles. In this section, we will discuss these IANPs with direct targeting method, indirect responsive method, and synthetical method. 3.1. Direct targeting method Direct targeting method refers to that the IANPs could directly interact with bacteria or organic substances mentioned in section 2.1 and 2.2 to realize local enrichment of IANPs. Some recent studies regarding IANPs with direct targeting method are listed in Table 1.
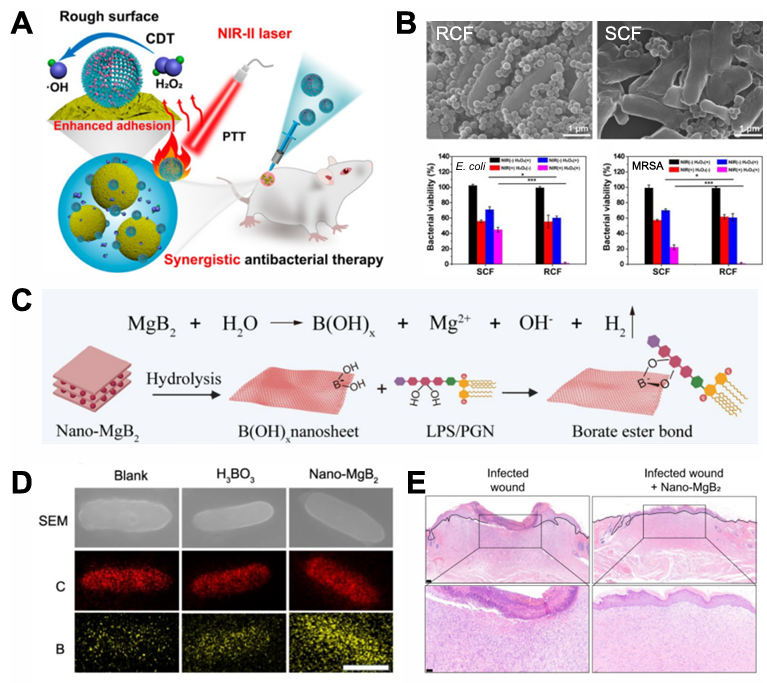
Abbreviations: PEG: polyethylene glycol; Ce6: chlorin e6; PBA: phenylboronic acid; PCL: polycaprolactone; BODIPYMA: boron dipyrromethene monomer; MAAPBA: 3-methylacrylamidephenylboronic acid; LAMA: 2-lactobionamidoethyl methacrylate; PDA: polydopamine; SNP: sodium nitroprusside; MOF: metal–organic framework; Mal: maleimide; CuTCPP: copper-tetrakis-(4-carboxyphenyl) porphyrin; AgNPs: silver nanoparticles; MBA: mercaptophenylboronic acid; BC: bicarbonate; QPCuRC@MSiO2: silica-supported near-infrared carbon dots; CA: cinnamaldehyde; DT-Ag: dendritic large pore mesoporous silica nanoparticles (DLMSNs) followed by deposition of Ag; CS: chitosan; MSN: mesoporous silica nanoparticle; PEICD: β-cyclodextrin -modified polyethylenimine; ADA: adamantane; FWNPs: multifunctional spherical structure; Apt: aptamer; QCS: quaternized chitosan; OFLX: ofloxacin;; QAS: quaternary ammonium salt; COS: chitosan oligosaccharide; GODs: graphene quantum dots: ICG: indocyanine green; GO: graphene oxide; BNN6: N,N’-di-sec-butyl-N, N’-dinitroso-1,4-phenylenediamine; PAA: polyacrylic acid; LEVO: levofloxacin; ConA: concanavalin A; PBA-DHPM: phenylboronic acid-(3,4-dihydropyrimidin-2(1H)-one); AuNRs: gold nanorods; OMV: outer membrane vesicle; CEF: ceftazidime; RvD1: Resolvin D1; NMVs: cell membrane-formed vesicles; PCN: porous coordination network 3.1.1. Polymer-based IANPs Polymers are widely applied to constructing IANPs owing to their adjustable and well-defined architectures and self-assemble/assemble behaviors. Modifying the polymer-based IANPs with a ligand group that enables them to interact with specific target sites is an advantageous feature of polymer-based IANPs. Xue et al. modified PEG with chlorin e6 (Ce6) and phenylboric acid (PBA) to obtain PEG-Ce6 and PEG-B respectively which assembled with NDIA, a photothermal agent, to form NDIA@PEG-Ce6/B NPs [99]. The methicillin-resistant S. aureus (MRSA) were found to be attached with plenty of NDIA@PEG-Ce6/B NPs due to the targeting capability of BA groups and killed by ROS generation and thermal effect under 808 nm laser irradiation. There was no significant cytotoxicity observed when NDIA@PEG-Ce6/B NPs were incubated with human immortalized keratinocyte (HaCaT) cells for 24 h at a high concentration of 400 μg/ml. Furthermore, PBA groups- containing IANPs were proved to possess enhanced ability of epithelial penetration. Shi et al. prepared a E. coli-targeting nanoplatform which could capture E. coli by high-affinity interaction between mannose and FimH on pathogenic E. coli. Besides bacteria themselves, biofilms also serve as the targets of IANPs. Weldrick et al. reported an alcalase-coated polyacrylic acid (PAA) nanogel of which the alcalase could degrade the EPS matrix of G +bacterial biofilm to increase the local concentration of antibiotics. 3.1.2. Inorganic IANPs Various inorganic nanomaterials (such as metal and metallic oxide) have been extensively utilized to fabricate IANPs due to their designable physical and chemical architecture for directly capturing bacteria. It has been found that rough surfaces possessed promoted performance of bacterial adhesion which inspired the development of IANPs with rough surfaces. Song et al. fabricated mesoporous silica nanospheres with rough surfaces which showed enhanced bacteria-capturing capability. Liu et al. fabricated a near-infrared (NIR) light responsive nanohybrid with rough surfaces (RCF) for PDT&CDT synergetic antibacterial therapy (Fig. 2A–C). RCF nanohybrids showed enhanced bacterial adhesion property and antibacterial performance compared with the nanohybrids with smooth surfaces (SCF) meanwhile showed rare cytotoxicity and negligible hemolysis. Owing to the nanoscale hairy surface of bacteria, multiple interaction between IANPs with rough surfaces and bacteria could be induced when the bacteria were exposed to IANPs which led to the local enrichment of bacteria for effective elimination of bacteria. High-affinity interaction between IANPs and bacteria such as covalent bond and electrostatic interaction is a driving force for targeting bacteria. Meng et al. prepared reactive metal boride nanoparticles (MB NPs) which could hydrolyze to generate a local alkaline microenvironment, enhancing the trapping effect of the hydrolysis product i.e., boron dihydroxy groups (Fig. 2D&E) [136]. The hydrolysis reaction of MB NPs could not only induce a high focal concentration of Mg 2+ to disrupt the membrane structure of living bacteria, but also trap dead bacteria-released LPS/PGN to inhibit the LPS/PGN-induced excessive inflammation. Guo et al. developed a positively charged nanoplatform based on copper manganate nanoflakes which could bind with pathogenic bacteria and eliminate them by ROS generation and PT effect.
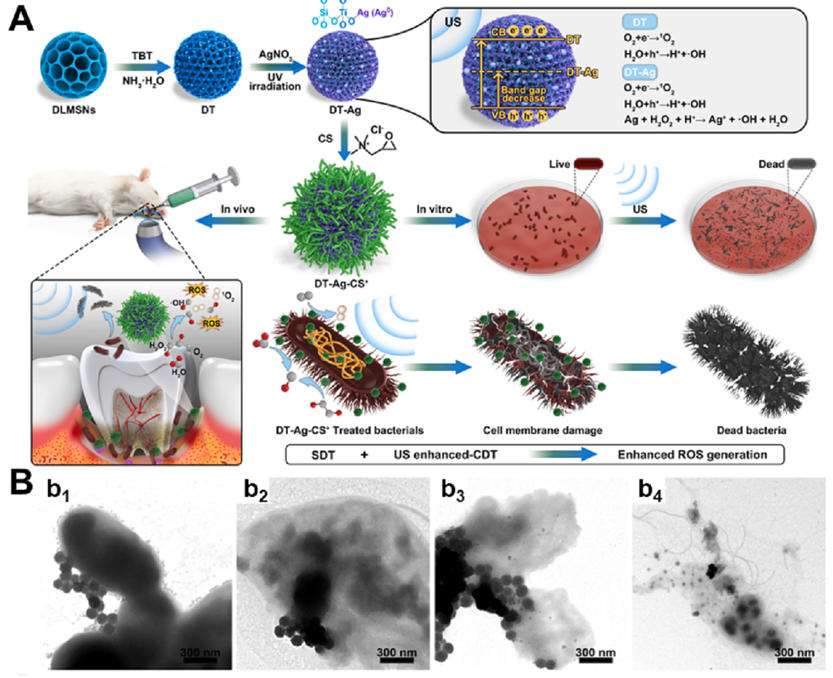
Fig. 2. (A) Schematic illustration of RCF nanohybrid for synergistic antibacterial therapy. (B) SEM images of RCF and SCF adhered on E. coli surfaces and quantitative analysis of the antibacterial ability of RCF and SCF against E. coli and MRSA. Reproduced with permission. Copyright 2021, American Chemical Society. (C) Boron-trapping strategy for bacteria-infected wound healing. (D) Elemental mapping of P. aeruginosa incubated with H 3 BO 3 Scale bar = 500 nm. C, carbon atom. B, boron atom. (E) H&E staining of infected wounds treated with or without Nano-MgB 2 and Nano-MgB 2 for 3 h. at day 12. Scale bar (up) = 100 scale bar (down) = 50 μ m. Reproduced with permission. Copyright 2022, Springer Nature. 3.1.3. Organic-inorganic hybrid IANPs By combining inorganic IANPs with organic components, hybrid IANPs with direct targeting methods can be obtained. Silica NPs combined with functional components to obtain hybrid IANPs are widely explored. Xin et al. developed a novel nano-sonosensitizer (DT-Ag-CS + ) by growing TiO 2 on dendritic large-pores mesoporous silica nanoparticles (MSNs) followed by deposition of Ag and modification of chitosan (Fig. 3). The DT-Ag-CS + nanoplatform possessed positively charged due to the outer layer of chitosan which contributed to penetrating the bacterial cell and then bactericidal 1 O + 2 , ?OH and Ag were produced under ultrasound irradiation to kill pathogenic Porphyromonas gingivalis (P. gingivalis) and treat periodontitis. Liu et al. coated the silica NPs with poly[2-(methacrylamido) glucopyranose] (pMAG) obtaining a E. coli-targeting nanoplatform which simultaneously possesses bacterial targeting, imaging and ablation capabilities. Metal-organic framework (MOF) has been utilized to construct hybrid IANPs as well. Tan et al. modified copper-tetrakis-(4-carboxyphenyl) porphyrin (CuTCPP) MOF nanosheet with mercaptophenylboronic acid (MBA) and silver NPs to obtain CuTCPP@AgNPs@MBA nanoplatform. The CuTCPP@AgNPs@MBA possessed high-efficiency and specific elimination of G + bacteria and enhanced biocompatibility due to the MBA molecules. Other inorganic nanomaterials such as metals and metal oxides have been functionalized with ligand molecules to endow them with direct targeting capability, thereby promoting their bactericidal effects. Zhang et al. fabricated a virus-like-inspired nanoplatform via in situ growth of Au NPs and decoration of PBA-based glycopolymers. The spikey surface topology and chemical modification of surfaces synergistically contributed to the capture and phagocytosis of bacteria which were then eliminated by combination of PDT and PTT. Du et al. reported a self-assemble of NiO NPs and Van-mediated AuNPs which is found to aggerate on the surfaces of MRSA and possess enhanced photothermal bacteria-killing effects by the enrichment of Van and magnetism. Wang et al. functionalized iron oxide magnetic with bacterial species-identifiable aptamers and Ce6 molecules to realize specific identification of S. aureus and photodynamic disinfection respectively. The distinctive fluorescent property of carbon dots (CDs) endowed CDs-based IANPs with bioimaging capability. Chu et al. developed a multifunctional CDs-based nanoplatform i.e., quaternary ammonium salt (QAS) modified near-infrared carbon dots (RCDs-C 35. The RCDs-C 35 ) nanoplatform not only damaged bacterial structure by electrostatic interaction of QAS and PT effect of CDs but also showed f lorescent signal when incubated with living cells which possessed great potential in diagnosis and treatment of bacterial infection.
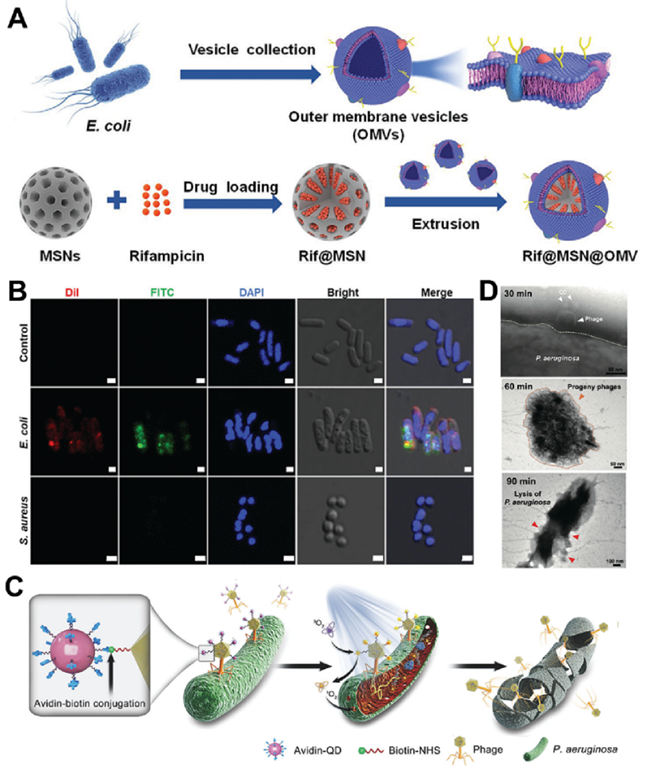
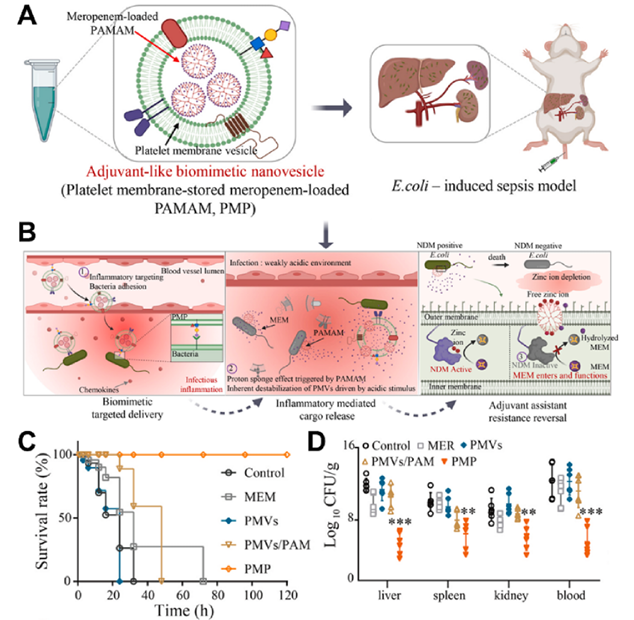
Fig. 3. (A) Schematic illustration for the preparation and the anti-periodontitis mechanism of DT-Ag-CS towards P. gingivalis. TEM images of P. gingivalis incubated with DT-Ag-CS + + NPs. (B) Adsorption and penetration abilities of DT-Ag-CS upon US irradiation for 0 s (b 1 ), 100 s (b 2 ), 200 s (b 3 ), and 300 s (b 4 + ). Reproduced with permission. Copyright 2023, Elsevier. 3.1.4. Biohybrid IANPs Nature created interesting examples for direct target to pathogenic bacteria, among which biological membranes with excellent biocompatibility and biofunctionality have been extensively used in constructing biomedical nanomaterials, especially the materials required for targeting property. Phagocytes are the first line of defense of humans, thus the first to be investigated for constructing IANPs. Thamphiwatanaa et al. demonstrated the macrophage membrane- coated NPs can realize sepsis management by absorbing endotoxins and proinflammatory cytokines. Wang et al. utilized the bacteria- treated macrophage membrane with highly expressed bacterial recognizing receptors to coat on a gold-silver nanocage and the resulted nanoplatform showed effective treatment of bacterial infection in vivo. Except for macrophages, neutrophil membranes have also been used to camouflage nanomaterials due to their inflammation-targeting capability. Hemocytes have inspired the development of IANPs due to their excellent immune evasion, subendothelium adhesion and pathogen targeting properties. The natural red blood cell membrane (RBCM) contributed to escaping from clearance of the immune system and targeting infection sites. Wu et al. reported a biomimetic nanovesicle (PMP) by storing antibiotics into platelet membrane-derived nanovesicles for targeted delivery of antibiotics. Due to the shared characteristics of nanovesicles with platelet membranes, such as immune evasion, subendothelial adhesion, and pathogen targeting, the biohybrid PMP accumulated at infection sites and released the loaded antibiotics resulting in a focal high- concentration of antibiotics. In addition, the outer membrane vesicles (OMVs) derived from G- bacteria have been utilized as immunostimulants for antibacterial or anticancer therapy due to the similar composition and structure with bacterial membrane. Wu et al. constructed a G- bacteria-targeted drug delivery system (Rif@MSN@OMV) on the basis of similar chemical structure between OMVs and G- bacteria (Fig. 4A&B). The Rif@MSN@OMV system was capable of fusing into the sandwich-like structure of G- bacteria and thus promoting the uptake of antibiotics and antibacterial efficiency. Bacteriophage (phage) as a natural enemy of bacteria provides a promising opportunity for developing bacteria-targeting materials owing to their specific bacteria-binding mechanism. Wang et al. combined phages with photocatalytic quantum dots (QDs) by using avidin–biotin bioconjugation and obtained a P.aeruginosa-targeting nanosystem (QD@Phage) (Fig. 4C&D). The QD@Phage first located on the surface of bacteria with the assist of phages and then generated ROS under light irradiation to realize synergistic phage-assisted photocatalytic therapy (PA-PCT) against planktonic bacteria and biofilm.
Fig. 4. (A) E. coli outer membrane vesicles (OMVs) were isolated and coated onto rifampicin-loaded MSNs (Rif@MSN) to ? give the final biomimetic NPs Rif@MSN@OMV. (B) CLSM images of E. coli and S. aureus after incubation with MSN@OMV (DiI (red) labeled OMVs and FITC (green) labeled MSNs) for 3 h at 37 C, and E. coli without treatment of MSN@OMV as control. Scale bar = 1 μm. Reproduced with permission. Copyright 2022, Wiley-VCH. (C) Schematic illustration of phage-assisted photocatalytic therapy against GFP-P. aeruginosa. (D) TEM images revealing PA-PCT processes of QD@Phage: host bacteria binding (30 min), progeny phages releasing (60 min), and lysis of a bacterium (90 min). Reproduced with permission. Copyright 2022, Wiley-VCH. (For interpretation of the references to colour in this figure legend, the reader is referred to the web version of this article.) 3.2. Indirect responsive method Indirect responsive method refers to that the IANPs could respond to the infection microenvironment to realize physicochemical transformation of IANPs. Some recent studies regarding IANPs with indirect responsive method are listed in Table 2.
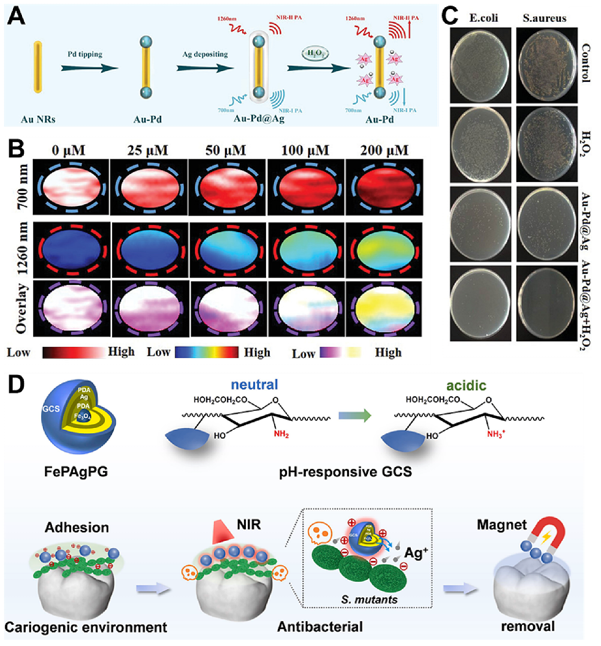
Abbreviations: OEGMA: oligo (ethylene glycol) methyl ether methyl methacrylate; DEAEMA: diethylaminoethyl methacrylate;MPDA: mesoporous PDA; ZIF: zeolitic imidazolate framework; GO: graphene oxide; GODs: graphene quantum dots: CHO-SBA-15: aldehyde-modified mesoporous silica SBA-15; ε-PL: epsilon-poly-L-lysine; AMO: amoxicillin; PANI: polyaniline; GCS: glycol chitosan; FePAgPG: Fe3O4 NPs modified with PDA, silver, PDA, and GCS in sequence; BPY: 2,2′-bipyridine-4-carboxylic; CORM: CO-releasing molecule; NR: nanorod; BIF: boron imidazolate framework; nFMs@Amp: Fe3+-doped MOFs loaded with ampicillin; MSNE: EPR (an endoplasmic reticulum-targeting peptide) grafted MSNs; MEL: melittin; TSPBA: N1-(4-boronobenzyl)–N3–(4-boronobenzyl)-N1, N1, N3, N3-tetramethylpropane-1,3-diaminium; AuMP: mesoporous silica-coated gold nanorod; AuMN: amino- modified AuMP; SGQDs: single-layered graphene quantum dots; TAPP: 5,10,15,20-tetra-{4-[3-(N, N-dimethyl-ammonio) propoxy]phenyl} porphyrin; EGCG: epigallocatechin gallate; PLNPs: persistent luminescence nanoparticles; BSA: bovine serum albumin; Gox: glucose oxidases; MNPs: magnetic nanoparticles; MB: methylene blue; Ca@PDAFe-CNO: Fe2+complexation and surface grafting of cysteine-NO (CNO) on the PAD layers of Ca@PDA NPs 3.2.1. Polymer-based IANPs Polymer-based IANPs with indirect responsive method could be obtained by integrating various chemical bonds or groups that are sensitive to the bacterial infection microenvironment into the polymers. Zhao et al. synthesized a copolymer with a ROS-sensitive bond as linkers which assembled with 5,10,15,20-tetra-{4-[3-(N, N-dimethyl-ammonio) propoxy]phenyl} porphyrin (TAPP) to form a H2O2 and pH dual- sensitive nanoplatform for antibacterial PDT. The copolymer containing arylboronic esters was stable under natural conditions while under weakly acid conditions, the arylboronic esters became sensitive to H2O2 and the TAPP was released and protonated, thus promoting the PDT efficiency. Wu et al. reported a polydopamine (PDA) NPs-based nanoplatform whose outer layer possessed pH-responsive charge switch, resulting in effective binding with bacteria under acid conditions. While under physiological conditions, the nanoplatform was negatively charged which decreased the uptake of mammal cells and reduced the cytotoxicity. Natural polymers such as HA were widely utilized in fabricating responsive IANPs due to their enzyme-responsive degradation. Xiu et al. fabricated an IANP (HCM NP) with cascaded antibacterial property by using metronidazole (MNZ) and Ce6 functionalized HA (HA-Ce6-MNZ). After being delivered into MRSA biofilm infected sites, HCM NPs released Ce6 and MNZ under the catalysis of HAase. Subsequently, the released Ce6 generated 1O2 under 808 nm laser irradiation consuming the oxygen content in biofilm, and the aggravated hypoxia condition activated MNZ to kill the bacteria. 3.2.2. Inorganic IANPs Inorganic nanomaterials showed structural changes when encountered with infection microenvironment which is beneficial for fabricating IANPs. Li et al. coated CuO2 on the surfaces of SiO2 to form a pomegranate-like nanospheres. The CuO2 decomposed to generate Cu2+ and H2O2 under acid conditions and then a Fenton-like reaction took place to catalyze H2O2 into ?OH by Cu2+which showed enhanced antibacterial activity. Importantly, no significant cytotoxicity was observed when the nanospheres were co-cultured with mouse embryo fibroblasts (NIH/3T3 cells) at a high concentration of 100 μg/mL. Imaging capability of organic nanomaterials could be integrated into IANPs to endow them with diagnosis and precise treatment capabilities. Ye et al. developed a silver-coated, palladium-tipped gold nanorod (Au- Pd@Ag NR) nanoprobe that could release bactericidal Ag+under the gentle oxidation of endogenous H2O2 and realize quantitative photoacoustic diagnosis of inflammation by absorption of NIR light (Fig. 5A–C). Due to the etching of H2O2, the nanoprobe can measure local H2O2 concentration by the ratiometric change in photoacoustic signals which provides quantitative data of severity of inflammation.
Fig. 5. (A) Synthetic route of Au-Pd@Ag nanoprobe. The etching and oxidation of the Ag shell by H 2 O . 2 O 2 released the Ag ions which triggered their absorption variation at 700 nm and 1260 nm, and effectively killed bacteria in vivo. (B) PA images at 700 and 1260 nm of Au-Pd@Ag before and after incubated with different concentration of H 2 (C) PA images at 700 and 1260 nm of Au-Pd@Ag before and after incubated with different concentration of H 2 O 2 . Reproduced with permission [158]. Copyright 2020, Wiley-VCH. (D) Schematic illustration of removable photothermal antibacterial “warm paste” target for cariogenic bacteria. Reproduced with permission. Copyright 2022, Elsevier. 3.2.3. Organic-inorganic hybrid IANPs Hybrid IANPs synthesized the responsiveness of organic and inorganic components to improve their intelligent antibacterial performances. Zeolitic imidazolate framework (ZIF) is a classic MOF material with acid pH-responsive disassembly behavior which corresponds with the acid microenvironment of bacterial infection. Peng et al. reported a ZIF-8-coated mesoporous PDA core–shell nanosystem loaded with Pifithrin-μ (PES), a natural inhibitor of heat-shock protein, to realize pH responsive release of Zn2+and PES. Benefitting from the load of PES, the bacterial heat-resistance was suppressed and a biocompatible low-temperature PTT was proceeded inducing effective elimination of bacterial biofilm. He et al. developed a diagnostic and therapeutic nanoplatform based on ZIF-8 which could sense the acidic microenvironment and responsively release zinc ions to eliminate bacterial infection and respond to the oxidative stress of infection site by absorbing excessive ROS of EGCG. Ma et al. designed a pH- responsive O 2 and H 2 O 2 self-supplying nanosystem based on Co- containing ZIF-67 of which the pH-responsive released Co catalyze H 2 O 2 2+ could to generate ?OH for efficient killing bacteria. The released CaO 2 reacted with water to generated H supplied for the CDT process. Metal-based NPs have attracted researchers’ interests due to their distinctive photothermal or photodynamic property while limited efficiency urges their combination with responsive or targeting components. Wang et al. synthesized a mesoporous Pb-Cu nanoalloy loaded with amoxicillin (AMO) which were then coated by ZIF-8 to obtain a dual stimuli-responsive procedural antibacterial system (Pb-Cu/ AMO@ZIF-8). The outer layer of Pb-Cu/AMO@ZIF-8 nanosystem sensed the pH change and split to release the antibiotics AMO when entering bacterial biofilm. The photothermal capability of the inner Pb- Cu nanoalloy under NIR irradiation accelerated the releasing rate of AMO as well as damaged the bacterial cell membrane. In addition, the assemblies of metal ions with other chemicals also provides a versatile platform for fabricating IANPs. Dai et al. reported an acidity-activatable NPs (GB@P) consisting of the self-assembly of Fe3+, polyaniline and bovine serum albumin and the load of glucose oxidases (GOx). The GOx of GB@P NPs could convert the endogenous glucose into gluconic acid (GA) and H2O2 in situ. The former enhanced the acidity of bacterial infection microenvironment and triggered the PT effect. The latter killed the bacteria through increasing oxidative stress and activating the macrophages. Various metal oxides and sulfides have been applied to fabricating IANPs due to their enzyme-mimicking activity or stimuli responsive property. Fe3O4 NPs have been widely studied due to their excellent PT effect, magnetic property, and horseradish peroxidase-like nanozyme activity. Xu et al. developed a removable “warm paste” (FePAgPG) targeting for cariogenic bacteria based on layer-by-layer modification of Fe3O4 NPs (Fig. 5D). The FePAgPG nanoplatform with positive charge induced a strong adhesion with cariogenic bacteria and then, in cariogenic acid environment, Ag-assisted PTT strategy was realized under NIR light irradiation. Finally, the FePAgPG nanoplatforms were removed by the external magnetic field. Gong et al. fabricated a magnetically retained and glucose-fueled hydroxyl radical nanogenerator (pFe3O4@GOx) by GOx-functionalized PAA-coated Fe3O4 (pFe3O4) [168]. The generation of GA catalyzed by GOx and the acid microenvironment of bacterial infection simultaneously promoted the catalytic activity of Fe3O4, thereby decomposing the endogenous H2O2 into ?OH for CDT of bacterial infection. Carbon-based nanomaterials have also been employed for combating bacteria and biofilms, such as CDs and graphene oxides (GO). Liu et al. established a multifunctional antibacterial nanoplatform (SCH) by combining CO-releasing molecule (CORM-401) and HA into single- layered graphene quantum dots (SGQDs). The ultrathin SGQD nanosheets of SCH nanoplatform were exposed and penetrated into bacterial cells after contacting with bacterial-secreted HAase. Subsequently, ROS were generated by SGQD nanosheets under white light irradiation and triggered the release of CO which contributed to the cascade-activated “nanoknife” effect and photodynamic/CO gas therapy. 3.3. Synthetical method Synthetical method refers to that the IANPs simultaneously possess direct targeting and indirect responsive capabilities to realize programmable and intelligent antibacterial activity when confronted with bacterial infection. Some recent studies regarding IANPs with synthetical method are listed in Table 3.
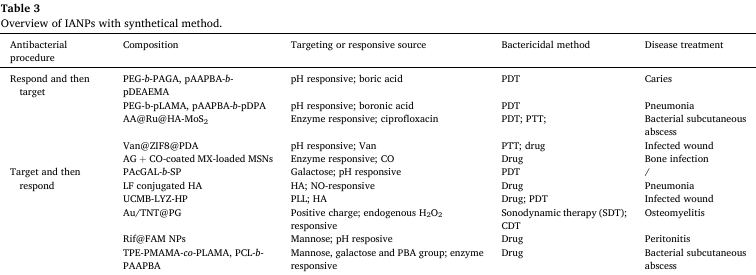
Abbreviations: AGA: 2-acrylamide glucopyranose; AAPBA: 3-acrylamidophenylboronic acid; DEAEMA: DPA: 2-diisopropylaminoethyl methacrylate; AA: ascorbic acid; AG: Arabic gum; CO: colistin; MX: moxifloxacin; AcGAL: 2-O-acryloyloxyethyl-(2,3,4,6-tetra-O-acetyl-β-D-galactopyranoside; SP: spiropyrane; LF: levofloxacin; UCMB: hierarchical coating of dense silica and dendritic mesoporous silica on upconversion nanoparticles (UCNP) loaded with methylene blue (MB); LYZ: lysozyme; HP: HA and PLL; Au/TNT: gold-doped titanate nanotubes; PG: guanidinium-rich polymer; Rif: rifampicin; FAM: mannose-decorated poly(α-N-acryloyl-phenylalanine)-block-poly(β-N-acryloyl-D-aminoalanine); TPE: tetraphenylethylene; MAMA: 2-methacrylamido mannopyranose 3.3.1. Polymer-based IANPs By delicate design and elaborate synthesis, polymers-based IANPs could possess targeting and responsive capabilities concurrently. Yu et al. synthesized polyethylene glycol-b-poly(2-acrylamide glucopyranose) (PEG-b-PAGA) which assembled with poly(3-acrylamide phenylboronic acid)-b-poly(2-(5,5-dimethyl-1,3-dioxan-2-yloxy)ethyl acrylate) (PAAPBA-b-PDMDEA) and co-encapsulated ciprofloxacin (CIP) and IR780 to form a biofilm microenvironment-induced PEG sheddable light-activated nanoplatform (PS-NP@(C +I)) (Fig. 6). The PEG shields contributed to penetrating into biofilm, and then under the acid microenvironment of biofilm, detached to expose boric acid ligands for targeting the pathogenic bacteria. The PS-NP@(C +I) nanoplatform possessed favorable biocompatibility even at 500 μg/ml meanwhile displayed excellent dispersing activity against Streptococcus mutans (S. mutans) biofilms. Ma et al. prepared a P. aeruginosa-targeting nanoplatform by synthesizing spiropyran-conjugated glycoclusters that could specifically bind with lecA, a lectin abundantly distributed on the cellular wall of P. aeruginosa. The glycoclusters and indocyanine green (ICG), a photosensitizer, formed a nanoplatform by self-assembly. The nanoplatform firstly bound to the surface of P. aeruginosa and then disassembled under the acid condition, resulting in the release of ICG. The cell viability of these nanoplatforms reached around 90% even at 1 mg/mL. HA as a typical natural polysaccharide has been employed to establish IANPs with targeting capability taking advantage of its high affinity for receptors of macrophages. Lu et al. reported a NO-sensitive nanomicelle based on levofloxacin (LF) conjugated HA (HA-LF) that could enter infected host cells via the highly-expressed CD44 receptors. The release of conjugated LF was triggered by the presence of endogenous NO, as the covalent linkage between LF and HA utilized NO- sensitive o-phenylenediamine groups.
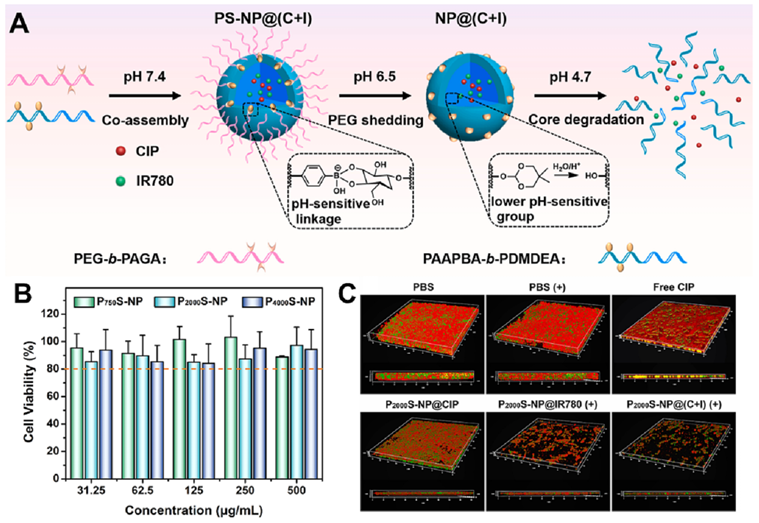
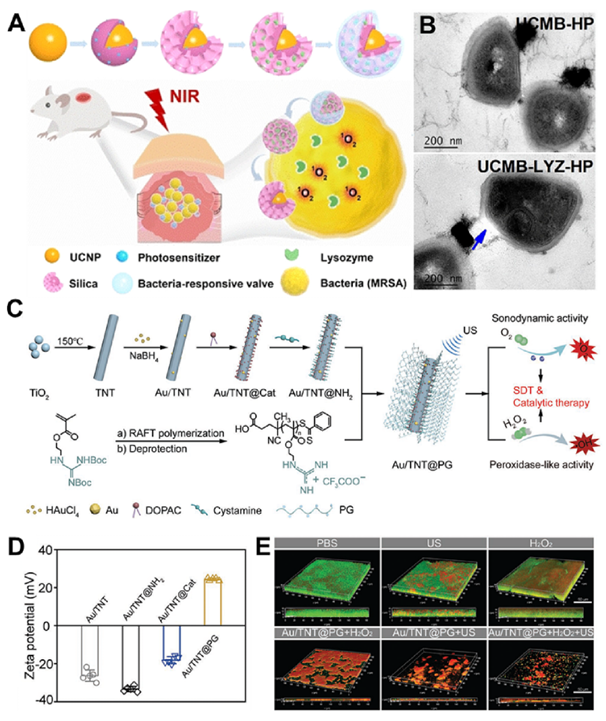
Fig. 6. (A) The formulation of PS-NP@(C + I) to afford a microenvironment-controlled dynamic modulation of nanoplatforms. (B) The cell viability of NIH 3 T3 cells after incubation with P 750 2000 S-NP, and P 4000 S-NP at 37 ℃ for 24 h. (C) CLSM 3D images of S. mutans biofilms in various groups. Red: bacterial cells stained with EB; green: biofilm EPS labeled with FITC-ConA. Reproduced with permission. Copyright 2022, Elsevier. (For interpretation of the references to colour in this figure legend, the reader is referred to the web version of this article.) 3.3.2. Organic-inorganic hybrid IANPs Organic-inorganic hybrid IANPs not only have precisely regulable chemical composition for intelligent antibacterial purpose but also possess ultrasound/photo-responsive bactericidal methods based on inorganic component. Silica NPs have been utilized to constructing IANPs as carriers. Aguilera-Correa et al. prepared moxifloxacin-loaded silica NPs coated with Arabic gum and colistin showing enzyme responsive degradation in E. coli biofilm. After the fall-off of Arabic gum, the exposed colistin targeted and bound with the bacterial surfaces leading to the local release of antibiotics. Moreover, traditional silica NPs could respond to exogenous or endogenous stimuli to realize intelligent dynamic or thermal therapy by combining novel responsive components. To enhance the efficiency against resistant bacteria, Li et al. incorporated lysozymes (LYZ), hyaluronic acids, and PLL with upconversion nanoparticles (UCNPs) to obtain a synergistic lysozyme- PDT antibacterial nanoplatform (UCMB-LYZ-HP) (Fig. 7A&B). The introduction of PLL endowed the UCMB-LYZ-HP nanoplatform with highly positive charges to enhance their adhesion to bacteria. After binding on the surface of bacteria, HA was degraded to realize intelligent release of LYZ. Then the bacteria were synergistically killed by cell wall deconstruction effect of LYZ and ROS attack against bacterial membrane and cytoplasm. Sonodynamic therapy (SDT) could greatly overcome the depth limitation of photo-related therapy. Cheng et al. took advantage of the sonodynamic therapy (SDT) effect of TiO 2 to construct a microenvironment-activable nanoplatform (Au/TNT@PG) whose outer layer was decorated by a guanidinium-rich polymer (PG) in the purpose of penetrating into biofilm (Fig. 7C–E). The Au/TNT@PG nanoplatform generated 1 O 2 under ultrasound irradiation for SDT and catalyzed the decomposition of endogenous H 2 O 2 into toxic ?OH in the acidic infection microenvironment for CDT which synergistically resulted in effective elimination of MRSA biofilm.
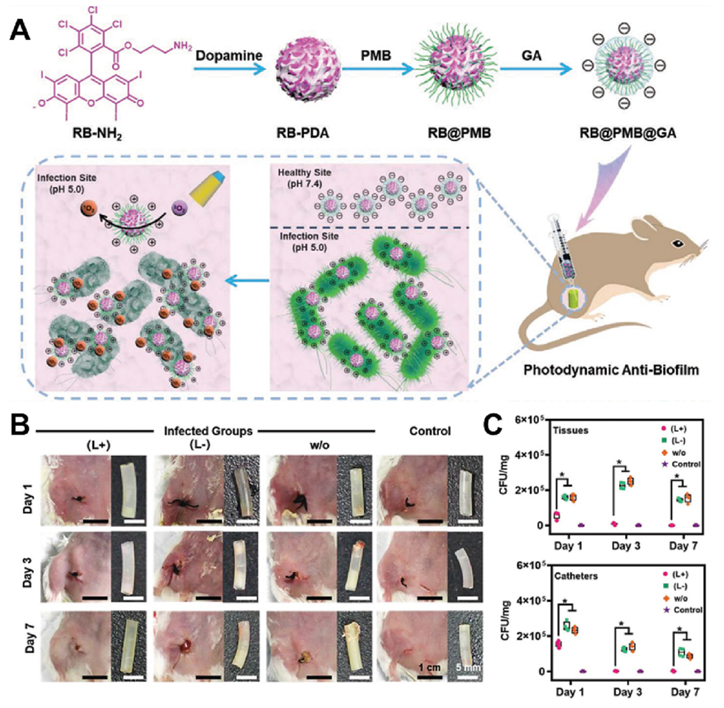
Fig. 7. (A) Schematic illustration for a nanohybrid UCMB-LYZ-HP with features of intelligently bacteria-responsive release of lysozyme and NIR-triggered high production of 1 O 2 for combating deep-tissue infections caused by resistant bacteria. (B) Representative TEM images of MRSA after incubation with UCMB-HP and UCMB-LYZ-HP, respectively. Reproduced with permission. Copyright 2021, Wiley-VCH. (C) Schematic illustration of the construction of Au/TNT@PG nanostructure. (D) Zeta potential of Au/TNT, Au/ TNT@Cat, Au/TNT@NH 2 , and Au/ TNT@PG. (E) Representative CLSM 3D images biofilm treated by various therapies (scale bars: 50 μm). The concentration of H 6 2 O 2 was 50 × 10 M. Reproduced with permission. Copyright 2022, Wiley-VCH. 4. Application of IANPs in treating bacterial infection As mentioned above, IANPs possess favorable biocompatibility and intelligent antibacterial activity offering potential advantages in antibacterial and anti-biofilm application. In recent years, a growing body of research has demonstrated that IANPs can be used to treat a wide range of bacterial infections. In this section, we will focus on the application of IANPs in treating bacterial infections in hard tissue, soft tissue, medical devices, and others. 4.1. Hard tissue-related bacterial infection Osteomyelitis that is clinically hard to handle and easy to relapse after surgery needs effective and thorough elimination of pathogenic bacteria. IANPs with active targeting capability is highly potential in treatment of osteomyelitis. Wang et al reported a pretreated macrophages membrane-coated gold nanocage (Sa-MGSNC) for precise drug delivery of osteomyelitis. The Sa-MGSNCs showed enhanced accumulation at the infected tibia after injection on the tail vein of mice with osteomyelitis. The aggregation of Sa-MGSNCs at infection sites and their affinity to pathogenic S. aureus resulted in a higher drug concentration under NIR light irradiation compared to the nanocages without pretreated macrophages membrane. Furthermore, cell membrane coating significantly benefitted the circulation time and blood retention time of Sa-MGSNCs. The Sa-MGSNCs with enhanced accumulation at infection sites, improved blood circulation lifetime, and promoted photothermal effects possessed high potential in treating osteomyelitis and other localized bacterial infections. Treatment of infected bone defects not only needs thorough elimination of pathogenic bacteria but also pays attention to the bone regenerative process. Furthermore, common nanomaterials cannot persistently function due to easy diffusion resulting in the needs of scaffolds or active targeting capability. By combining bone regenerative components, IANPs offer a promising strategy for improving the treatment outcomes of bone infections. Li et al. reported a bone infection microenvironment-modulating composite by loading endogenous H 2 responsive hollow MnO 2 (hMnO 2 ) O 2- NPs into gelatin methacrylate (GelMA) hydrogels. The hMnO version of H 2 O 2 into O 2 2 NPs not only catalyzed the confor facilitating osteoblasts’ growth but also responded to the ROS change and released the bone morphogenetic protein-2 (BMP-2)-associated peptide. In vivo rat skull defects experiment demonstrated that the excessive H Fig. 7. (A) Schematic illustration for a nanohybrid UCMB-LYZ-HP with features of intelligently bacteria-responsive release of lysozyme and NIR-triggered high production of 1 O 2 for combating deep-tissue infections caused by resistant bacteria. (B) Representative TEM images of MRSA after incubation with UCMB-HP and UCMB-LYZ-HP, respectively. Reproduced with permission. Copyright 2021, Wiley-VCH. (C) Schematic illustration of the construction of Au/TNT@PG nanostructure. (D) Zeta potential of Au/TNT, Au/ TNT@Cat, Au/TNT@NH 2 , and Au/ TNT@PG. (E) Representative CLSM 3D images biofilm treated by various therapies (scale bars: 50 μm). The concentration of H 6 2 O 2 was 50 × 10 M. Reproduced with permission. Copyright 2022, Wiley-VCH. featured in the acid surrounding microenvironment (pH 4.5–5.5) due to the supply of dietary sucrose and other ingredients. Xu et al. prepared a cariogenic bacteria-targeting and pH-responsive nanoplatform with removable performance which was potential for treating dental caries. Yu et al. reported a precise pH responsive nanosystem for caries prevention. The PEG in outer layers contributed to infiltrating into the biofilm and fell off under the weakly acid conditions (pH 6.5) which exposed the boric acid ligands to specifically anchor bacteria. Subsequently, the inner drug-carrying assembly rapidly released the payloads resulting from the transient hydrolysis of DMDEA in the focal acidic microenvironment (pH 4.7) of oral biofilms. Under NIR irradiation, the released drug and photosensitizer synergistically eliminated the oral biofilm. The nanosystem showed excellent biofilm-eliminating performance and biosafety in a rodent model with the features of early childhood severe caries. 2 O 2 in defect site was significantly eliminated and newly formed bone completely covered the defects which revealed favorable outcomes of treating infected bone defect using hMnO 2 /GelMA composites. Dental caries caused by the biofilm of cariogenic bacteria are featured in the acid surrounding microenvironment (pH 4.5–5.5) due to the supply of dietary sucrose and other ingredients. Xu et al. prepared a cariogenic bacteria-targeting and pH-responsive nanoplatform with removable performance which was potential for treating dental caries. Yu et al. reported a precise pH responsive nanosystem for caries prevention. The PEG in outer layers contributed to infiltrating into the biofilm and fell off under the weakly acid conditions (pH 6.5) which exposed the boric acid ligands to specifically anchor bacteria. Subsequently, the inner drug-carrying assembly rapidly released the payloads resulting from the transient hydrolysis of DMDEA in the focal acidic microenvironment (pH 4.7) of oral biofilms. Under NIR irradiation, the released drug and photosensitizer synergistically eliminated the oral biofilm. The nanosystem showed excellent biofilm-eliminating performance and biosafety in a rodent model with the features of early childhood severe caries. 4.2. Soft tissue-related bacterial infection Wound healing is a complicated process involving hemostasis, inflammation, proliferation, and remodeling, meanwhile is open and vulnerable for bacterial invasion which will result in delayed healing and, even serious adverse effects that endangers life. Elimination of bacteria, regulating inflammation of infection sites and promoting tissue repair are fundamental requirements for treating wound bacterial infection. Xie et al. fabricated Janus Ca@PDA Fe-CNO NPs which responded to the low pH value and excessive glutathione of infection sites to generate H 2 O 2 and trigger the release of NO respectively. In the early stage, the ROS generated by the catalysis of Fe 2+ on H 2 O 2 and the RNS converted from NO contributed to effective elimination of bacteria. Then, the long-term slow release of NO promoted endothelial cell proliferation and migration and accelerated the proliferation process of bacteria-infected diabetic wounds. The MRSA-infected wound is a severe threat to human health on which traditional antibiotics have a limited effect. Liu et al. proposed a combination strategy of physical puncture, PDT and CO gas delivery for managing MRSA-infected wound. The sharp structure was exposed under HAase and penetrated into bacterial cells followed by ROS generation and CO release which synergistically contributed to eradicating MRSA. The wound closure rate was around 95% in nine days and the survival MRSA was close to 0 CFU/ mg of tissue revealing the excellent antibacterial and wound healing capability. Although IANPs have been demonstrated to treat wound infection, the lack of moisture preserving and bioliquid regulating properties urges the arise of IANPs-based composite materials for facilitating wound healing. Li et al. fabricated a composite cryogel (CSG- M X ) by grafting nanoenzyme onto dialdehyde chitosan via Schiff base reaction [184]. The CSG-M X cryogel could respond to the acid microenvironment and release endogenous H 2 O 2-responsive nanoenzyme for PDT against bacteria. Due to the great hydrophilicity and macroporous structures, CSG-M X cryogel showed excellent capability of bioliquid management to avoid overhydration in infection sites. Collectively, eliminating bacteria is just one of the procedures of bacteria-infected wound healing. Other performances of the wound healing materials need to be taken into consideration, such as biocompatibility, promotion of tissue repair, moisture and bioliquid management etc. Ocular bacterial infections mainly caused by trauma or intraocular surgical operation require timely and effective treatment and if not, there will be corneal ulcer perforation, even intraocular infection, and eventually eyeball atrophy. Clinical strategy is to frequently administrate antibiotics to meet the therapeutic concentration which increases the risk of drug-resistant bacteria. One of the emerging ways of treating ocular infection is to utilize drug carrier to increase the bioavailability in eyes. Zhang et al. synthesized an amphiphilic glycopolymer containing boron dipyrromethene and boronic acid moieties which could effectively increase bacterial uptake of antibiotics through deeply penetrating epithelial into the cornea and targeting bacterial cell wall. Moreover, they synthesized a ROS-scavenging glyco-nanoplatform by introducing the pendant group of phenylboronic acid-(3,4-dihydropyrimidin-2(1H)-one (PBA-DHPM). In vivo rat model with S. aureus- induced bacterial keratitis could be cured by the glyco-nanoplatform within 5 days through targeted co-delivery of levofloxav cin and chondroitin sulfate and ROS scavenging behavior. Drug-free strategy for treating ocular infections is also being developed. Zhu et al. prepared a PT nanoplatform (EtNBSS NPs) assembled from pH-responsive phenothiazinium dye which possessed charge conversion from negative to positive under acid microenvironment of biofilm. The positive charge and high-efficiency low-temperature PT effect of EtNBSS NPs synergistically contributed to the biofilm eradication which was further validated in treating drug-resistant bacterial keratitis and endophthalmitis of mice. Notably, the high photothermal conversion efficiency of EtNBSS NPs made certain that the laser dose for complete bacterial elimination had no adverse effect on the vulnerable eyes. Oral tissue bacterial infections such as periodontitis seriously affect human health and quality of life. Mechanical debridement combined with antibiotics is clinical first choice of treating periodontitis, whereas certain problems such as irregular shape of periodontium, post- operation pain, drug-resistance and side effects require more effective and painless method. Xin et al. developed a ROS generation nanoplatform based on a nano-sonosensitizer modified with bacteria-targeting CS for treating periodontitis. The nanoplatform could be directly administrated into the gingival sulcus of Wistar rats with periodontitis and generate ROS under ultrasound irradiation to kill pathogenic P. gingivalis to suppress alveolar bone resorption and relieve inflammatory responses. Such noninvasive and bactericidal strategy would guide the development of the next-generation treatment of periodontitis. 4.3. Medical devices-related bacterial infection With the rapid development of biomedical materials, the usage rate of various medical devices such as metal implants, catheters, etc. is increasing. Bacterial invasion and adhesion on the surface of medical devices result in biofilm formation and further lead to medical devices- related infection. Traditional strategies to treat such infection include debridement and antibiotics administration which would make patients suffer from the pain of surgery and the side effects of large dosages of antibiotics. Prevention of medical devices-related infection needs to seal the interface of device and tissue and endows the interface with great antibacterial effects. However, it seems insufficient to treat infection associated with mature biofilm. IANPs showed great potential in treating medical devices-related bacterial infection due to their intelligent bactericidal mechanism and high biocompatibility. Nie et al. reported a bone and bacteria dual- targeting IANPs (MSN@D&U) by modifying bone-targeted peptides (Peptides D 6 ) and bacteria-targeted peptides (UBI 29-41 ) on the surfaces of MSNs. The MSN@D&U nanoplatform loaded with Van possessed superior targeting capability into infection sites and controlled release of Van resulting the enhanced treatment of implant- related bacterial infection. Cai et al. reported a multi-mode antibacterial nanoplatform (ICG&CO@G3KBPY) by encapsulating ICG and manganese pentacarbonyl bromide into a BPY-modified peptide dendrimer-based nanogel. The controlled release of CO gas was driven by endogenous H 2 O 2 and photodynamic and PT effect of ICG to enhance the penetration and ablation function of ROS to biofilm. In vivo biofilm-infected catheter experiment demonstrated the excellent biofilm elimination and anti-inflammation effect of ICG&CO@G3KBPY nanoplatform. In addition to gas permeation, opposite charge attraction is an available way to enhance the penetration of IANPs into biofilm. Wu et al. fabricated a pH-sensitive nanosystem (RB@PMB@GA) by layer-by-layer decoration of polymyxin B and GA on Rose Bengal (RB)-PDA NPs (Fig. 8). The RB@PMB@GA NPs remained negative charged at physiological condition which possessed great biocompatibility, and became positive charged when exposed to acid microenvironment of infection. After surface charge conversion, the RB@PMB@GA NPs targeted and bound to the surface of bacteria and showed effective elimination against in vivo biofilm on catheter via PDT. Similarly, Yu et al. reported an antimicrobial peptides and antibiotics dual-loading system for treating implant infection in which the β-cyclodextrin (β-CD)-modified polyethylenimine (PEICD) possessing highly positive charged rendered the platform with high-efficiency targeting towards P. aeruginosa biofilms.
Fig. 8. (A) Schematic illustration of the preparation process of photodynamic NPs for enhanced penetration and antibacterial efficiency in biofilms. (B) Typical photographs of the incision areas and implanted catheters from mice under different treatments on Days 1, 3, and 7. (C) Quantitative analysis of bacterial colony- forming units obtained from the tissues and catheters in each group. Reproduced with permission. Copyright 2021, Wiley-VCH. 4.4. others Sepsis refers to acute systemic infections caused by pathogenic bacteria that invade the blood circulation, grow and proliferate in the blood, and produce toxins, the mortality of which reaches 30%. Specific recognition and inhibition of pathogenic bacteria and suppress the inflammation are of critical significance for treatment of sepsis. Zhao et al. developed an infection-responsive and macrophage-targeting nanoplatform which responded to the excessive H 2 O 2 of intercellular infection to release antimicrobial peptides and antibiotics. The targeting and responsive release of payload contributed to inhibiting bacterial growth and relieving inflammation which protected the mice with systematic infection from death and organs dysfunction. How to avoid being cleared from immune system and target the pathogenic bacteria are another two problems for treating sepsis while IANPs are designed with targeting capability. Wang et al. fabricated a PEG-Ce6 and bacterial species-identifiable aptamers functionalized iron oxide magnetic NPs (Fe 3 O 4-Ce6-Apt) in which the PEG polymer promoted the compatibility and stability during blood circulation. The Fe 3 O 4- Ce6-Apt nanosystem realized successful diagnosis of sepsis caused by a single or multiple bacterial species owing to the bacteria identification of Apt and magnetic enrichment of Fe 3 O 4 . Wu et al. prepared a platelet membrane vesicles (PMVs)-coated drug delivery system which integrated pathogen targeting and site-specific drug release properties due to the introducing of PMVs (Fig. 9). Under the “guidance” of PMVs, the nanovesicles could target the activated endothelial cells of infection site and bind the bacteria. The loaded drug gradually released triggered by local weakly acid microenvironment to kill the bacteria and diminish inflammation which was demonstrated in model of E. coli- infected mice with sepsis.
Fig. 9. (A) and (B) Representative schematic diagram of adjuvant-like biomimetic nanovesicles reverse carbapenem resistance for tackling New Delhi metallo- β-lactamases (NDMs) producing superbugs infections. (C) Survival curves for the NDM-EC1349 sepsis model. (D) Bacterial loads of infected tissues in the sepsis mice model after different treatments. Reproduced with permission. Copyright 2021, Elsevier. Pneumonia caused by P. aeruginosa, Streptococcus pneumoniae (S. pneumoniae) or S. aureus has been a troublesome issue. Bacterial invasion through the airway epithelial lining implicated the survival pattern transformation and the escape form host immune system which meant traditional antibiotics therapy would bring little effect. In such condition, the factors of microenvironment were overexpressed, such as inflammation factors, pH, toxins, enzymes, etc., that provided opportunities for IANPs to treat pneumonia. For example, various bacterial toxins (e.g., H 2 O 2 and H 2 S) can be secreted by multiple bacterial strains such as P. aeruginosa and S. pneumoniae and inspire the design of IANPs for treating corresponding diseases. Wu et al. developed a H 2 O 3+ 2-reactive MOF-based nanosystem (nFMs@Amp) which produced ?OH via the catalysis of Fe upon H 2 O 2 and decomposed to release the loaded Amp (Fig. 10). Surfactant Pluronic F-127 was employed as a protective layer to decrease the zeta potential of nFMs@Amp and avoid being cleared from immune system. The nFMs@Amp nanosystem accumulated at infected lungs after intravenous injection, evidently eliminated the drug-resistant S. pneumoniae and reduced the lung tissue damage showing great potential in clinically treating pneumonia. Aiming at disassembly the biofilm of P. aeruginosa, Wang et al. prepared an alginate lyase-loading nanocomposite consisting of Ag NPs and a mesoporous organosilica layer. The alginate lyase specifically degrade the alginate of P. aeruginosa biofilm and the antibiotics and silver ions responsively released under the acid microenvironment to synergistically inhibit pathogenic bacteria. In vivo experiments demonstrated that the nanocomposites successfully eradicated P. aeruginosa from the mouse lungs and decreased the lung injuries without deaths or serious side effect. Biofilm-associated infection remains a severe challenge due to the biofilm's recalcitrance to antimicrobials and to the immune system. Nanorobots that could be precisely controlled to move along designed paths have been applied to destroy biofilm structure and expose internal pathogenic bacteria. Hwang et al. reported the catalytic antimicrobial robots (CARs) for highly efficient elimination of biofilm. The CARs not only killed the bacteria and degraded the EPS matrix by their peroxidase-like activity but also could be directed to biofilm locations by magnetic field and scrubbed away dead bacteria and biofilm debris. Sun et al. fabricated a magnetic urchin-like capsule robots (MUCRs) by loading magnetic liquid metal droplets (MLMDs) into natural sunflower pollen. The shape transformation of MLMDs into spheroids and rods with sharp edges was triggered by external magnetic field which contributed to rupturing EPS matrix of biofilm and killing the embedded bacteria in assistance with natural microspikes of MUCRs. Liu et al. reported a NIR- II -driven Janus nanomotor for precise treatment of bacterial infection. Under NIR-II light, a distinct thermal gradient was formed due to the nanomotor’s Janus structure which drove the active motion of the nanomotor. Such autonomous movement of the nanomotors promoted the transdermal penetration and the interaction with pathogenic bacteria. Besides manipulation by external force, autonomous motion could be realized by loading the sources of active motion, such as the enzyme urease.
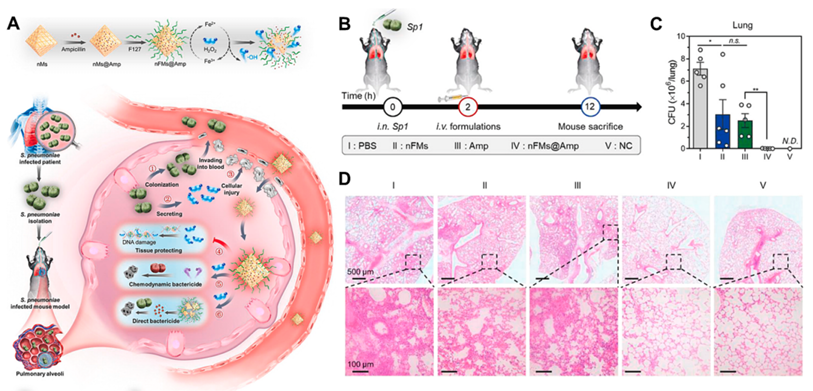
Fig. 10. (A) Schematic illustration showing the working principles of nFMs@Amp to treat hydrogen peroxide secreting S. pneumoniae infection. (B) Schematic illustration of the procedure of therapeutic experiment. (C) The CFU of S. pneumoniae in lungs with different treatments. (D) H&E-stained histological sections of lungs. Reproduced with permission. Copyright 2021, Elsevier. 5. Conclusion and perspectives The main obstacle to deal with bacterial infection lies in the limited bactericidal efficiency of antibiotics resulting from the emerging and spreading drug-resistant bacteria and the protective layer of bacterial community, i.e., biofilm. Meanwhile, there is a contradiction between bacteria-killing and biocompatibility accompanied with traditional antibacterial materials. Antibacterial nanomaterials with responsive or targeting capability have been developed in response to address the issue of bacterial infection in recent years which are defined as intelligent antibacterial nanoplatforms (IANPs) in this review. The IANPs show great biocompatibility in physiological condition due to their specific targeting to bacteria/infection sites or their responsive capability under pathological condition of bacterial infection. This review classified the design origins of IANPs aiming at the treatment of bacterial infection into three parts: targeting the bacteria, targeting the organic matrix, and responsiveness to chemical properties in the microenvironment of bacterial infection. Then, the construction of IANPs using direct targeting and/or indirect responsive method were highlighted and the composition of IANPs contains polymer, inorganic materials, organic–inorganic hybrid materials and biohybrid materials. At last, the applications of IANPs to treat various bacterial infection- related diseases were summarized, including hard tissue-related diseases, soft tissue-related diseases, medical devices-related diseases, and others. Although plenty of exciting progress about the development of IANPs are made, there still remains many challenges with the fabrication and clinical transformation of IANPs. 1) Biological aspect. The toxicity of biomaterials is a necessary question. To render the IANPs with targeting/responsive properties and high-efficiency bactericidal function, all sorts of materials are utilized in their fabrication which increases the risk of biosafety. Furthermore, the laboratory methods to evaluate the toxicity of IANPs subjected to nonstandard experiment model and limited experiment duration. Thus, more investigations about the biotoxicity of IANPs need to carry out in the future. Moreover, the generation of ROS to eliminate the bacteria is popular among the existing researches while ROS might cause toxicity and delay tissue repair whose long-term reliability needs to be investigated. On the other hand, the degradability of IANPs is rarely considered in the existing researches. Whether IANPs will be degraded and produce toxic products in the functioning process need to be taken seriously. The in vivo distribution, metabolism, and toxicity of IANPs or their degradation products should be explored in the future. 2) Functional aspect. Promoting the bactericidal efficiency and specif icity is a significant developing direction of IANPs which not only contributes to avoiding toxicity by reducing the dosage but also protect probiotics and mammalian cells. Targeting/responsive efficiency greatly influences the effects of IANPs which is limited by mononuclear phagocyte system capture, off-target bind with other biomolecules, and the complexity of bacterial infection. Thus, certain camouflaging arrangements and multiple targeting/responsive mechanism should be considered in the design of IANPs. In- depth investigations regarding the bacterial structure and composition and microenvironment of bacterial infection are needed to provide more target sites and responsive factors. Despite the population of light responsive antibacterial nanomaterials, their application in treating deep tissue infection is limited by the penetration depth of light. Moreover, few works attempt to develop smart theranostic nanoplatforms that could automatically detect infection in real time and activate bacteria-killing process while it may be the developing tendency in the future. 3) Technical aspect. Due to the complicated synthesis or the multiple composite process of most IANPs, large-scale production of IANPs with quality assurance and long-term stability remains difficult which is adverse to cost control and clinical transformation. For biohybrid IANPs, certain problems such as immature production technology, demanding preservation condition, limited characterization method etc. are confronted with the animal-derived membranes or vesicles. To boost the clinical transformation of IANPs and benefit the patients, more efforts should be put into developing simple, effective, and universal IANPs. Declaration of Competing Interest The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper. Data availability No data was used for the research described in the article. Acknowledgements This work was supported by National Natural Science Foundation of China (Grant Nos. U22A20158, 52203180, and 52073191) and Sichuan Science and Technology Program (Grant Nos. 2022NSFSC1949). Source: www.elsevier.com/locate/cej |