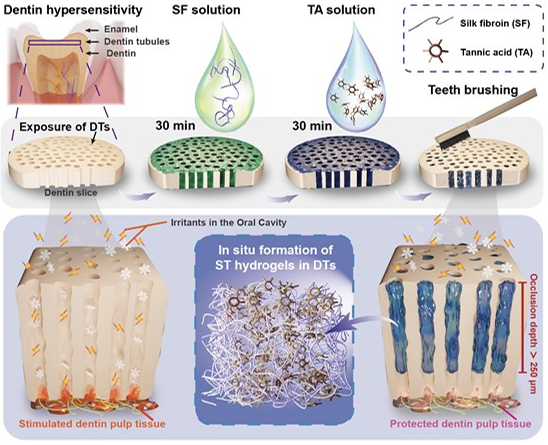
A Drop-by-Drop Self-Assembled All-Natural Hydrogel as a Desensitizer for Rapid and Enduring Management of Dentin HypersensitivityIssuing time:2025-06-26 09:50 Wanshan Gao, Yang Liu, Mingjing Li, Mingming Ding, Li Cheng, Chunmei Ding, Jiaojiao Yang, Jun Luo, Rongmin Qiu, Jianshu Lee Abstract Dentin hypersensitivity (DH) is a prevalent dental condition arising from the exposure of dentin tubules (DTs), leading to discomfort upon external stimuli. However, achieving swift and profound occlusion of these exposed DTs for immediate and enduring relief remains challenging due to the intricate dentin structure and oral environment. Herein, we propose a pioneering and facile drop-by-drop strategy involving an in-situ generated natural supramolecular hydrogel formed by self-assembling silk fibroin (SF) and tannic acid (TA) within the narrow DT space. When SF and TA aqueous solutions are applied successively to exposed dentin, they penetrate deeply within DTs and co-assemble into compact gels, robustly adhering to DT walls. This yields a rapid and compact occlusion effect with an unprecedented depth exceeding 250 μm, maintaining stable occlusion efficacy even under rigorous in vitro and in vivo erosion and friction conditions for no less than 21 d. Furthermore, the biocompatibility and effective occlusion properties are verified through cell studies in simulated oral settings and an in vivo rabbit model. This study, for the first time, demonstrates the translational potential of hydrogel-based desensitizers in treating DH with prompt action, superior occlusion depth and enduring treatment benefits, holding promise as clinical-friendly restorative solutions for delicate-structured biosystems. Keywords: silk fibroin, tannic acid, dentin tubule occlusion, rapid desensitization, long-term stability, clinical-friendly Introduction Dentin hypersensitivity (DH) is a prevalent condition affecting over a third of the population. It is characterized by transient pain arising from the nerve fibers of the pulp cavity underlying exposed dentin tubules (DTs), triggered by various mechanical, chemical, osmotic, and temperature stimuli. Therefore, rapid occlusion of the exposed DTs represents the primary therapeutic approach for DH treatment, effectively shielding the nerve fibers from external stimuli and providing fast alleviation of dental pain. Despite the widespread use of traditional methods like filling restorations, laser photodynamics therapy (PDT), dentin adhesives, and commercial desensitizing medications, their effectiveness is limited by the narrow pore size of DTs (< 3 μm) and the complex physiological conditions within, such as surface affinity. In the dynamic oral environment, where teeth wear ranges from approximately 15 μm y-1 under physiological conditions to 200 μm y-1 under pathological conditions, methods like PDT might offer rapid pain relief but can cause dentinal cracks and collagen fiber degradation. Moreover, they often fail to provide sufficient occlusion depth (typically less than 10 μm from dentin surfaces) necessary to withstand daily erosion and mechanical friction leading to their rapid removal and frequent recurrence of DH symptoms. In situ remineralization has gained recognition as a promising new-generation approach for achieving deep occlusion of DTs by delivering mineral ions and/or mineralization templates into the deep regions of DTs to induce dense mineral formation within DTs, providing longterm relief from DH. However, only a few remineralization strategies have managed to achieve relatively deep and complete DT occlusion, characterized by the dense mineral filling of DTs. For example, the application of lysozyme conjugated with poly(ethylene glycol) resulted in a mineralization occlusion depth of approximately 60 μm after 7 d, while the use of an ultrahighly concentrated polyelectrolyte-calcium suspension followed by phosphate solution achieved an impressive mineralization occlusion depth of 200 μm within 24 h, representing the most profound occlusion reported to date. Despite these achievements, remineralization approaches still face challenges in delivering satisfactory symptomatic relief in clinics due to the confined space within DTs, particularly in the deeper regions, restricting the supply of mineral ions and thus impacting their overall efficiency in achieving prompt symptomatic relief. Moreover, concerns regarding the potential risk of remineralization strategies inducing dental calculus and the safety risks associated with commonly employed synthetic remineralization materials add further apprehensions to the approach. In light of these challenges and concerns, it is imperative to explore alternative strategies that can synergistically achieve rapid, deep, and stable occlusion of DTs, transcending the limitations of the remineralization approaches. To achieve effective occlusion of DTs in DH treatment, hydrogels emerge as a promising solution, bypassing the need for additional chemical reactions and providing immediate DT occlusion. For example, Curodon D’Senz gel, a commercial desensitizer based on a selfassembling peptide matrix, effectively alleviates DH. However, due to its limited liquidity, it struggles to enter DTs, resulting in surface occlusion prone to detachment after friction, leading to a risk of poor long-term effect. Therefore, desensitizer materials must possess two key properties: excellent fluidity to deeply penetrate the confined DTs and the ability to rapidly solidify in situ, forming a compact and enduring structure that can effectively occlude the DTs and withstand external stimuli. A promising option for fulfilling these requirements could be hydrogels with both high fluid precursors during delivery and rapid gelation with remarkable stability upon precise localization. Therefore, in this study, we introduce a pioneering drop-by-drop strategy utilizing an in-situ formed natural hydrogel based on a combination of silk fibroin (SF), a natural protein, and tannic acid (TA), a plant polyphenol, to achieve rapid and profound DT occlusion for DH treatment (Figure 1). Leveraging the remarkable permeability and wetting properties of SF and TA to DTs, these molecules efficiently penetrate into DTs and self-assemble rapidly, forming dense hydrogels (referred to as ST hydrogels) within DTs. Our results demonstrate that ST hydrogels efficiently occlude the DTs, swiftly reaching an unprecedented depth exceeding 250 μm within 1 h. This obviates the need for lengthy remineralization processes or specific environmental conditions, maintaining stable occlusion efficacy even under rigorous in vitro and in vivo erosion and friction conditions for no less than 21 d. Furthermore, we designed a modified transwell dentin disk (modified-TDD) model to affirm that ST hydrogel occlusion within DTs effectively eliminates the risk of pulp cell stimulation resulting from substance exchange associated with DH. The efficacy of occlusion was further confirmed through an in vivo rabbit model. Our findings highlight, for the first time, the exceptional occlusion efficiency achieved through direct DT occlusion using an in-situ formed biocompatible natural hydrogel. It presents a groundbreaking and clinical-friendly alternative direction compared to existing mineralized occlusion treatments, providing nextgeneration solutions for DH management and offering valuable insights for future research on delicate-structured biosystems. Results and Discussion Rapid and profound DT occlusion via drop-by-drop ST Hydrogels Building on our previous findings demonstrating the self-assembly of SF and TA into dense and robust adhesive hydrogels via strong intermolecular hydrogen bonds, we hypothesized that this self-assembly process could be harnessed to occur within DTs, thereby facilitating stable DT occlusion and consequent long-term alleviation of DH. To realize this objective, we devised a drop-by-drop strategy, involving the sequential deposition of SF aqueous solution followed by TA aqueous solution onto the dentin surface. By leveraging the fluid nature of these solutions and the potential affinity between SF and calcium phosphate, we anticipated that these components could rapidly penetrate DTs. Simultaneously, due to strong intermolecular hydrogen bonds, prompt gelation would occur within the narrow DT space. This would effectively block and thoroughly occlude the DTs, providing lasting relief from DH symptoms (Figure 1).
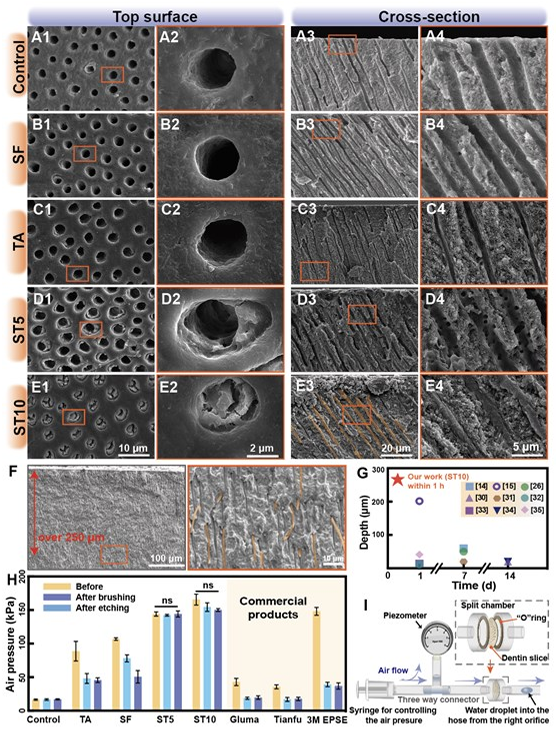
Figure 1. Schematic illustration of the desensitization process facilitated by ST hydrogels within DTs. After etching the dentin slices with 17 wt% EDTA to expose DTs, the procedure involves simply applying a drop of SF aqueous solution followed by a drop of TA solution. The robust interaction between SF molecules and DTs, in conjunction with the interaction between SF and TA, enables the self-assembly of ST hydrogels within DTs, resulting in rapid and in-depth occlusion. To investigate this, after sequential dropwise addition of SF aqueous solution and TA aqueous solution to dentin slices within 1 h, we conducted scanning electron microscopy (SEM) observations to evaluate the occlusion effect. Two concentrations of SF solution and TA solution (5 wt% and 10 wt%), combined in a 1:1 ratio, were employed for comparison, donated as ST5 and ST10, respectively. The control group consisting of ethylenediaminetetraacetic acid-etched (EDTA-etched) bare dentin (used as a model for DH) exhibited enlarged orifices representing exposed DTs (Figure 2A). Similarly, no obvious tubule occlusion was observed in the groups treated solely with TA or SF solution (Figure 2B-C). In contrast, within groups ST5 and ST10, the ST hydrogel underwent in situ formation within the DTs, with dense hydrogel formation observed primarily in the ST10 group (Figure 2D-E). Notably, the ST10 hydrogel exhibited in situ dense gel formation at depths exceeding 250 μm beneath the dentin surface (Figure 2F, S2), which is the maximum depth available in the established literature (Figure 2G), and far exceeds the performance of commercial products like Tianfu?, Gluma? and 3M ESPE Single Bond Universal? (Figure S3). These commercial desensitizers target DT occlusion, and their modes of action are analogous, making them suitable for direct comparison. It is worth noting that the plugs formed by these commercial products are only superficially attached to the dentin and can be easily removed. Considering the dynamic and complex oral environment, achieving rapid and deep tubule occlusion is crucial for long-term relief in DH.
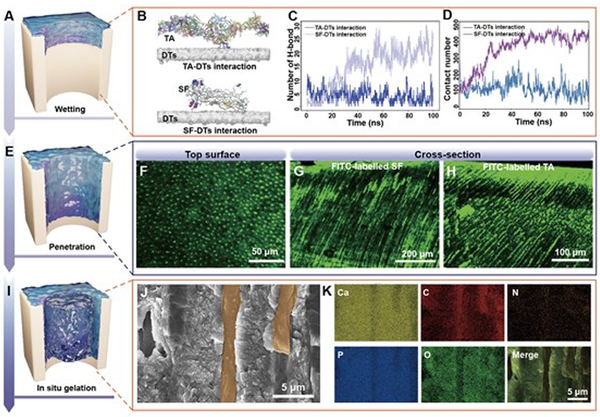
Figure 2. DT occlusion using ST hydrogels. (A-E) SEM images depicting the top surface and cross-section of dentin slices. (F) Cross-section SEM images demonstrate compact occlusion of DTs at a depth of 250 μm. (G) Statistical comparison of time and depth between the ST10 hydrogel and various desensitizing materials form the extant literature. Refs (H) Air pressure graph of dentin slices treated with different desensitizer materials before and after brushing and subsequent etching, using one-way ANOVA test followed by Tukey’s post-hoc test (n = 3, ns = not significant). (I) Schematic diagram of the air pressure test. To further quantitatively assess the tubule occlusion efficacy, a quantitative airtightness test was performed (Figure 2H-I). In comparison to the control group comprising EDTA acidetched dentin slices, all experimental groups showed an increase in airtightness values posttreatment. However, only the ST5 and ST10 groups achieved the maximum range of the piezometer, reaching pressures of approximately 160 kPa. Remarkably, according to the previous research, the airtightness behavior of the dentin slices treated with ST hydrogels remained relatively stable even after the surface of the dentin slice was subjected to mechanical brushing and acid etching with 2 wt% EDTA for 2 min. Possible Processes for In-Depth DT Occlusion by ST Hydrogels We employed molecular dynamics (MD) simulations through the GROMACS 2021 software to investigate the interaction between DTs and the components of ST hydrogels, namely SF and TA molecules. To ensure a fair comparison between the interactions of SF and DTs (SF- DTs) and those of TA and DTs (TA–DTs), we included 40 TA molecules with a molecular weight comparable to that of an SF molecule in the system. Figure 3A-B illustrates SF-DTs interactions, revealing higher contact numbers (Figure 3C) and hydrogen bond numbers (Figure 3D) compared to TA-DTs interactions. Moreover, the flexibility of SF molecules facilitated shape adaptation, thereby boosting the contact area and hydrogen bonding with DTs. Conversely, TA molecules tend to aggregate and engage in self-interactions, resulting in the formation of a notable first peak in the radial distribution function (RDF), as shown in Figure S4. Consequently, the initial application of the SF solution promotes the wetting of the DTs by the SF solution. To further analyze the penetration of ST hydrogels within DTs (Figure 3E), we utilised FITC fluorescent-labelled SF (FITC-SF) and TA (FITC-TA) to trace the distribution of ST hydrogels within DTs. Confocal laser scanning microscopy (CLSM) indicated that the ST hydrogel with FITC-SF infiltrated to depths surpassing 600 μm, while the ST hydrogel with FITC-TA penetrated over 300 μm (Figure 3F-H). These outcomes underscore the exceptional fluidity and viscosity of both SF and TA solutions (Figure S5-7), akin to that of purified water, enabling their effortless flow into DTs. In combination with the outcomes of molecular simulations, which highlight the robust interaction between SF and DT walls, along with the commendable fluidity and low viscosity exhibited by both SF and TA solutions, it becomes evident that these factors synergistically facilitate the profound penetration of SF and TA within DTs. Finally, the in-situ gel formation within DTs is vividly showcased through SEM images and corresponding energy dispersive spectroscopy (EDS) mapping (Figure 3I). The crosssectional SEM view (Figure 3J) illustrates the in-situ formation of ST hydrogel within DTs, achieving compact occlusion. EDS mapping reinforces the role of ST hydrogel in DT occlusion, with enrichments of C, N, and O evident in the mapping image, accompanied by subtle Ca and P element shadows at corresponding locations (Figure 3K). We also investigated alternative hydrogel formations with high fluid precursors, like acryloyl hydrogel. However, these attempts were unsuccessful in achieving robust and stable DT occlusion (Figure S8-S9), attributed to their inability to generate condensed and lasting hydrogel matrices within DTs. In essence, this comprehensive exploration unravels the intricate penetration dynamics of ST hydrogel within DTs, substantiating its potential as a transformative approach for achieving in-situ gel formation and profound occlusion.
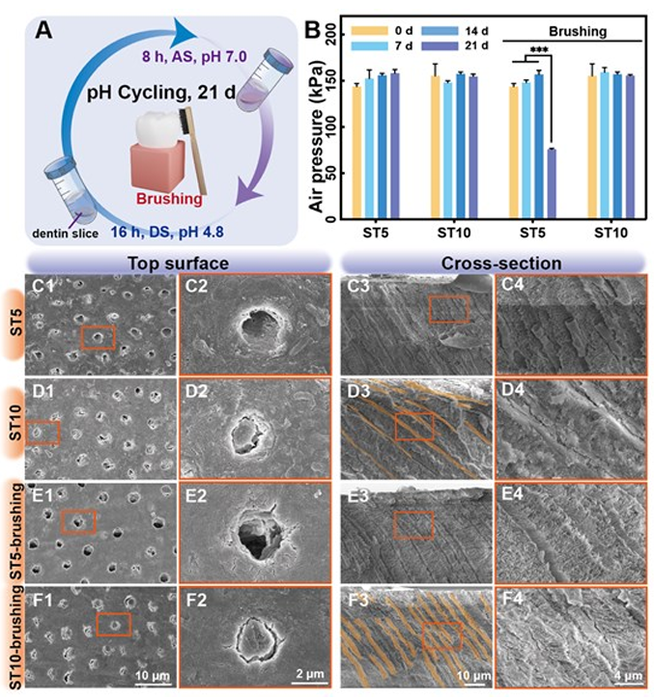
Figure 3. Possible Process for in-depth DT occlusion by ST hydrogels. (A) Schematic illustration of wetting within DTs. (B) Snapshots of TA-DTs and SF-DTs at equilibrium. (C) Hydrogen bond (H-bond) number and (D) contact number as a function of time. (E) Schematic illustration depicting the penetration of SF and TA solutions into DTs. (F) Top-view CLSM images of dentin surface treated with the ST10 hydrogel containing FITClabelled SF. (G) Cross-sectional CLSM images show FITC-labelled SF infiltrating inwardly to a depth exceeding 600 μm and (H) FITC-labelled TA exceeding 300 μm. (I) Schematic illustration of in-situ ST hydrogel formation within DTs. (J-K) Cross-sectional SEM image and corresponding EDS mapping images exemplify compact DT occlusion by ST hydrogels. Toothbrushing and acid attack -- long-term efficacy of DT occlusion in vitro Factors such as mechanical abrasion and acid erosion within the intricate oral environment present a significant obstacle to maintaining the long-term stability of desensitizer materials. Therefore, we employed a pH cycle model to simulate a biomimetic oral environment and evaluate the enduring occlusion effect of hydrogels. To further replicate the complex mechanical disturbances occurring in the oral cavity, we designed an enhanced pH cycling model that incorporates additional mechanical erosion to thoroughly assess the longterm stability of the ST hydrogels. The dentin samples underwent the pH cycling protocol, which involved 8 h of remineralization followed by 16 h of demineralization each day (Figure 4A). Furthermore, to better simulate daily oral habits, two subgroups (ST5-brushing group and ST10-brushing group) were introduced in each experimental group, with daily toothbrushing for 3 min (Figure 4E-F). After 21 d of pH cycling, the airtightness test showed no significant differences among the various procedures in the ST10 group, whereas the ST5-brushing group exhibited a significant decrease compared to the ST5 group (Figure 4B). SEM images revealed that the hydrogel within the DTs was less dense in the ST5 group compared to the ST10 group and couldn’t withstand routine mechanical brushing (Figure 4C-F). Therefore, the ST10 hydrogel demonstrates high suitability for achieving long-term relief in DH treatment.
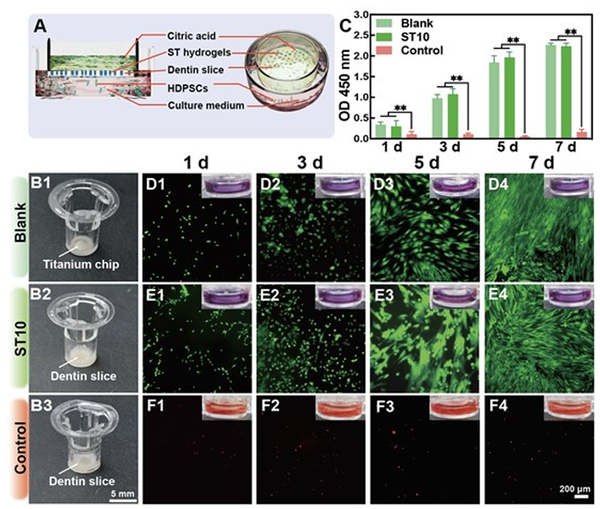
Figure 4. Long-term assessment of DT occlusion efficacy by ST hydrogels. (A) Schematic illustration of the pH cycling assessment of dentin slices treated with ST hydrogels. The model involved 8 h of exposure to artificial saliva (AS, pH 7.0) followed by 16 h in a demineralization solution (DS, pH 4.8) each day. (B) Air pressure changes in the airtightness test over time. The error bars represent the mean ± SD for n = 5, ***p < 0.001. (C-F) SEM images displaying the top surface and cross-section of dentin slices. Cytoprotective effect of DT occlusion against acid attack in vitro Desensitizing materials should not only effectively seal DTs but also shield underlying human dental pulp tissue from external irritants. This protection should mimic the isolation properties of natural dentin covered by enamel. Therefore, we designed a modified-TDD model (Figure 5A) and utilized human dental pulp stem cells (HDPSCs) as a representative of pulp tissue to comprehensively assess both the biocompatibility and the isolating capabilities of the ST hydrogel toward dental pulp tissue within a simulated oral bioenvironment prior to conducting in vivo experiments. Briefly, we replaced the Transwell chamber membrane with a dentin slice and ensured a sealed edge to restrict substance exchange solely on the dentin slice (Figure 5B). The blank group utilised a silicon chip rather than the dentin slice to replicate the full sealed condition (Figure 5B1). Prior to co-incubation with cells, we initially tested the feasibility of the modified-TDD model using an acid-base indicator litmus solution. Both the blank and ST10 groups were able to counteract the effect of citric acid in the upper chamber on the litmus solution in the lower chamber (Figure 5D-F, Movie S1). After being co-cultured with HDPSCs for 7 d, the ST10 group exhibits cell proliferation activity comparable to that of the blank group, indicating that citric acid in the upper chamber has no significant impact on cells located below (Figure 5C). Likewise, live/dead stained results indicated abundant live cells marked with green fluorescence and almost no dead cells marked with red fluorescence in both the blank and ST groups. This can be attributed to the ST hydrogel forming a stable barrier through the 1 mm thick dentin slice, thereby demonstrating excellent cytocompatibility and resistance against cavity irritants (Figure 5D-F). Additionally, it is inevitable for the ST hydrogel to contact with gingival tissue. Therefore, we co-incubated human gingival fibroblasts (HGFs) with ST10 hydrogel-treated dentin, and the results of the Cell counting kit-8 (CCK-8) assay showed favorable proliferative properties of HGFs (Figure S10).
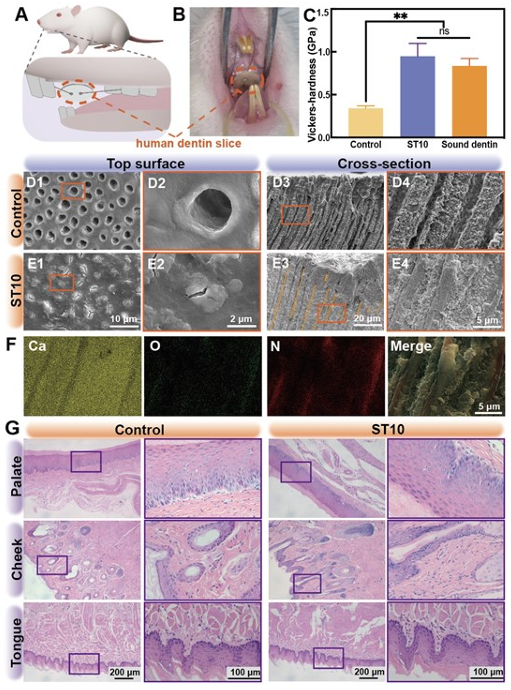
Figure 5. Cytoprotective effect of DT occlusion against acid attack. (A) Schematic illustration depicting the modified Transwell dentin disk (modified-TDD) model employed to detect both the cytocompatibility and cytoprotective effect of the ST10 hydrogel to HDPSCs. In the upper chamber, the conventional Transwell chamber membrane was replaced by our dentin disks, and HDPSCs were cultured in the lower chamber to detect whether the citric acid solution from the upper chamber could traverse the modified chamber membrane and affect cell viability in the lower chamber. (B) Photos displaying the upper chambers of the modified-TDD model, where the Transwell chamber membrane was replaced by (B1) a Titanium chip in the blank group, (B2) a dentin slice in the ST10 group and (B3) an EDTA-etched dentin slice in the control group. (C) Cytotoxicity results for HDPSCs cultured in the modified-TDD model using the CCK-8 assay. The OD values were calculated to evaluate the cytocompatibility using one-way ANOVA followed by Tukey’s post-hoc test (n = 3, ns = not significant, *p < 0.05 against the control group) (D-F) Fluorescence microscopy images of live/dead HDPSCs within the modified-TDD model. The corresponding inset photos display the lower chamber of the modified-TDD, with cells and culture medium replaced by a pH indicator litmus solution to visualize the pH shift from neutral (violet) to acidic pH (red) over time due to citric acid penetration from the upper chamber to the bottom chamber, observed in the control group. In contrast, both the blank group and ST10 group exhibited no obvious colour change during the assessment, indicating their robust isolation effect of the modified transwell chamber membranes in these groups. Biocompatible and long-term DT occlusion via ST hydrogel -- semi-in vivo validation In order to validate the clinical effectiveness of ST hydrogels in achieving long-term DT occlusion within a realistic oral environment, we conducted semi-in vivo animal experiments with human dentin slices. Briefly, EDTA-etched dentin slices and ST10 hydrogel-treated human dentin slices were placed in the palate of Sprague–Dawley (SD) rats to simulate oral challenges (Figure 6A-B). After a 21-d incubation, excellent DT occlusion was observed in SEM images of the ST10 group (Figure 6D-E). The results of EDS mapping demonstrated that ST10 hydrogel was responsible for the DT occlusion, evident from the enrichment of C, N and O in mapping images (Figure 6F, S11-12). This consistency with in vitro experiment results underscores the enduring DT occlusion capability of ST10 hydrogel. Furthermore, the Vickers hardness of dentin surfaces treated with ST10 hydrogel closely resembled that of healthy dentin (Figure 6C). Histological examination of rats treated with ST10 hydrogel revealed no significant inflammatory response or cellular damage in major organs or the oral organ, including tongue, cheek and palate (Figure 6G, S13). Additionally, no significant differences were observed in body weight or hematology parameters between rats in the ST10 hydrogel group and the control group with blank dental flakes (Figure S14-15).
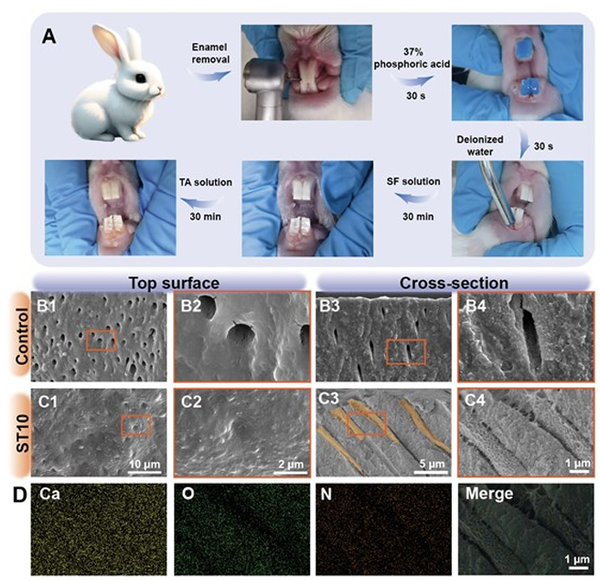
Figure 6. Long-term DT occlusion effectiveness was assessed through semi-in vivo validation. (A) Schematic diagram of the experimental procedure. (B) Photo of a human dentin slice situated within the rat oral cavity. (C) Hardness of dentin slices after cultured in the rat oral cavity for 21 d. The error bars represent the mean ± SD for n = 5, **p < 0.01, ns = not significant. (D-E) SEM images depicting the top surface and cross-section of dentin slices. (F) EDS mapping images corresponding to Figure E4. (G) Histological section images by H&E staining of rat organs, including palate, cheek and tongue. Effective DT occlusion via drop-by-drop ST hydrogel -- in vivo validation Encouraged by the promising outcomes mentioned earlier, we proceeded to evaluate the efficacy of the drop-by-drop ST hydrogel in occluding DTs within the oral cavity of rabbits. To create the DH model, we removed the enamel of the rabbit incisors, exposing the dentin surface, which was subsequently acid-etched (Figure 7A). Subsequently, the ST10 hydrogel treatment was administered directly on the dentin surface by adding 10 wt% SF aqueous solution and 10 wt% TA aqueous solution drop by drop onto dentin slices within 1 h. After 1 d, all the rabbits were sacrificed, and the incisors were extracted for SEM observation. The SEM images of the transverse and longitudinal dentin surfaces in the control groups clearly showed exposed and hollow DTs (Figure 7B-C). In contrast, despite the smaller tubule diameter in rabbit dentin compared to human dentin, the group treated with the ST10 hydrogel exhibited complete occlusion of DTs, further confirmed by EDS analysis (Figure 7D, S16-17). Motivated by exceptional in vitro and in vivo results, our material is advantageous due to its simplicity, offering an easy drop-by-drop application. We anticipate that, following the application of our material, routine clinical procedures like acid etching and resin filling can seamlessly integrate, providing a substantial clinical edge.
Figure 7. Efficacy of DT occlusion evaluated through in vivo validation. (A) Schematic diagram of the experimental procedure conducted in rabbits. (B-C) SEM images illustrating the top surface and cross-section of dentin slices. (D) EDS mapping images corresponding to Figure C4. Conclusion In summary, our study unveils a groundbreaking and clinical-friendly approach for swiftly and comprehensively occluding DTs in DH treatment, achieved through a self-assembled natural hydrogel based on SF and TA. The robust interaction between SF molecules and DTs, coupled with the interaction between SF and TA, culminates in the formation of a dense ST hydrogel that firmly adheres to DT walls. Our comprehensive investigations, encompassing in vitro, semi-in vivo and in vivo experiments, demonstrated the rapid and profound DT occlusion and safeguarding effect on underlying dental pulp cells, further highlighting the long-term treatment efficacy in the face of challenging oral conditions. This pioneering strategy not only presents an innovative therapeutic strategy for effectively addressing DH, but also underscores its potential as a transformative avenue for future dental treatments. Experimental Section Materials TA (analytical reagent) was purchased from Shanghai Aladdin Biochemical Technology Co., Ltd. (Shanghai, China). Sodium hydroxide (NaOH, analytical reagent) was purchased from Tianjin ZhiYuan Reagent Co., Ltd. (Tianjin, China). Lithium bromide (LiBr) was purchased from Aladdin (China). Polyethylene glycol (PEG, Mw = 20000) was purchased from Macklin (China). ethylenediaminetetraacetic acid (EDTA), calcium chloride (CaCl2), disodium hydrogen phosphate (Na2HPO4), hydrogen chloride (HCl) and all salt were purchased from Chron Chemicals Co., Ltd. (Chengdu, China). Fluorescein diacetate/propidium iodide (FDA/PI) and Fluorescein (FITC) were purchased from Solarbio Science & Technology Co., Ltd. (Beijing, China). Phosphate buffered saline (PBS) was purchased from Labgic Technology Co., Ltd. (Beijing, China). Dulbecco’s modified Eagle’s medium (DMEM), fetal bovine serum (FBS), and a penicillin-streptomycin solution were purchased from Gibco Life Technologies Co., Ltd. (Grand Island, USA). Cell counting kit-8 (CCK-8) was purchased from MedChemExpress Co., Ltd. (State of New Jersey, USA), and silica carbide grinding paper was purchased from Kafuwell Industrial Co., Ltd (Hangzhou, China). Preparation of hypersensitive dentin specimens The Ethics Committee of Huaxi Stomatological Hospital of Sichuan University authorized this research project (No. WCHSIRB-D-2021-078). After achieving the informed consent of donors, healthy caries-free premolars and third molars were obtained and cleaned, then stored in PBS solution containing 0.05 wt% thymol at 4°C. Dentin slices with a thickness of approximately 1 mm were prepared using a cutting (DTQ-5, Weiyi, Laizhou, China) and grinding machine (M-1, Weiyi, Laizhou, China). The slices were then polished using 600-, 800-, 1000-, 1500-, and 2000-mesh silica carbide grinding paper. To simulate dentin hypersensitivity (DH), dentin slices underwent an 8-min treatment with a 17 wt% EDTA solution (pH = 7) based on previous research and our pre-experiments.[14,26] Following treatment, slices were cleansed with deionized water using ultrasound for 15 min and dried with nitrogen gas. A subsequent airtightness test ensured the dentin tubules (DTs) were fully open, and slices without complete openness were discarded. Preparation of silk fibroin (SF) solution and tannic acid (TA) solution The purified SF solution was obtained based on a previously reported protocol.[36,37] Bombyx mori cocoons were halved and boiled in a 0.02 M Na2CO3 solution for 1 h, and washed with deionized water. This step was repeated three times to remove sericin. Then the degummed cocoons were dried overnight at 37°C. Next, the dry fibres were dissolved in 9.3 M LiBr solution for 4 h at 60°C. The resulting solution was dialyzed in deionized water for 36 h and then subjected to reverse dialyzed in 10 wt% PEG solution (Mw = 20,000) to obtain 10 wt% SF aqueous solution, which was stored at 4°C for further experiments. The 5 wt% SF solution was prepared by mixing 5 mL deionized water and 5 mL 10 wt% SF solution. To prepare the 10 wt% TA solution, 1.0 g of TA powder was added to 9.0 mL of deionized water.[24] The 5 wt% TA solution was prepared by mixing 5 mL of deionized water with 5 mL of the 10 wt% TA solution. Preparation and characterization of ST hydrogels The ST hydrogels used for FTIR testing were prepared by mixing the 10 wt% TA solution and 10 wt% SF solution in a 1:1 ratio. Since direct investigation of hydrogels within dentin was not feasible using FTIR, the lyophilized powder of this ST hydrogel was analyzed by FTIR (Nicolet 6700, Thermo Scientific, USA) to identify characteristic peaks, and the spectra scanned from 4000 cm-1 to 400 cm-1. DT occlusion via drop-by-drop ST hydrogels In the in vitro and in vivo experiments, the EDTA-etched dentin slices were dried using nitrogen gas, and the untreated side of the teeth was marked. The simple application of ST hydrogels was performed in two steps. First, a drop of SF solution was applied to the tooth slice surface and air-dried in 30 min. Then another drop of TA solution was applied to the same tooth slice surface and air-dried in 30 min. Those who used TA solution or SF solution solely by dropping it on the dentin slice surface and then air-drying in 30 min were the TA and SF groups. Finally, the ST hydrogel-treated dentin slices and the TA solution-, SF solution- treated dentin slice were brushed with an ultrasonic electric toothbrush to remove any remaining on the dentin surface, simulating daily brushing. Additionally, three commercially available products based on the principle of DTs occlusion, namely Gluma?, Tianfu herbal Desensitizer?, and 3M ESPE Single Bond Universal? were applied to the dentin sample according to the manufacturer's instructions.[14,26,38] Each group comprised three samples, with the EDTA-etched group serving as the control. Following goldsputtering, dentin morphology was observed through scanning electron microscopy (SEM, Helios 5 CX, Thermo Scientific, USA) with an accelerating voltage of 5 kV and a working distance of 5.1 mm. Energy Dispersive Spectroscopy (EDS, Helios 5 CX, Thermo Scientific, USA) was employed to analyze chemical elements with an accelerating voltage of 20 kV and a working distance of 5.1 mm. Cross-sectional views of the dentin were obtained by fracturing the dentin slices. Airtightness Test Since only unblocked DT can allow gas flow, the macroscopic occlusion effect can be assessed by evaluating permeability with an air pressure infiltration device. As shown in Figure 2I, the dentin slice is sandwiched between a pair of rubber rings with a central hole and is fixed in the chamber as a whole. After the entire setup is complete, drop a water droplet into the hose from the right orifice. When the gas passes and thus the water droplets start moving inside the tube to the right, we can record the air pressure as the critical air pressure. Each group consisted of three dentin slice samples, and the airtightness test was repeated five times for each sample. To mimic the impact of friction and etching in an oral environment, we subjected dentin slices to a 2 wt% EDTA aqueous solution for 2 min following mechanical friction application.[39] Specifically, the initial airtightness test yielded 15 data points for each group labeled "before" in Figure 2H. Subsequent to mechanical friction on the same samples, the second airtightness test was conducted, providing 15 data points for each group labeled "after brushing" in Figure 2H. This sample batch then underwent 2 wt% EDTA etching, leading to the third airtightness test and generating 15 data points for each group labeled "after etching" in Figure 2H. Fluorescence imaging The FITC labeling of the SF solution was accomplished as follows: 5 mL of SF solution underwent reaction with 5 mg of FITC in 1 mL of DMSO, yielding the FITC-labeled SF solution (FITC-SF solution). Similarly, 10 mL of TA solution reacted with 5 mg of FITC in 1 mL of DMSO to produce the FITC-labeled TA solution (FITC-TA solution). Subsequently, the reaction mixture was dialyzed against deionized water within a 1 kDa dialysis bag (Biosharp, USA) for 12 h in darkness, effectively eliminating any unbound FITC molecules. In the experimental setup, one group was involved in treating EDTA-etched dentin with the FITC-SF solution and non-labeled TA solution, following the previously outlined procedure. In contrast, the second group treated the EDTA-etched dentin with non-labeled SF solution and the FITC-TA solution. The number of samples in each group is 3. The resulting dentin slices underwent examination using a confocal laser scanning microscope (CLSM, Nikon NSIM, Japan) operating under a 488 nm laser, with fluorescence depth measurements conducted in Z-stack mode. pH cycling experiments for long-term measurement In the following 3 w, EDTA-etched and ST hydrogel-treated dentin slices were subjected to pH cycling experiments, in which the dentin incubated in artificial saliva solution (AS: 1.5 mM CaCl2, 0.9 mM KH2PO4,130 mM KCl, 20 mM HEPES buffer and 5 mM NaN3) for 8 h and demineralization solution (DS: 3.0 mM CaCl2, 3.0 mM KH2PO4, and 50 mM acetic acid) for 16 h per day.[40] The AS and DS solutions were refreshed daily and all the samples were kept at 37°C. Each of the four groups (ST5, ST10, ST5 brushing, and ST10 brushing) initially had 10 samples. Within each group, there were two subgroups: One subgroup consisted of 5 samples and was used for dentin airtightness assessment at the designated time points (0, 7, 14, and 21 d). The other subgroup, also containing 5 samples, was designated for SEM observation at 21 d. Molecular dynamics (MD) simulations All simulations were executed using GROMACS 2021 software. TA molecules were simulated employing the CHARMM general force field, while other molecules utilized the CHARMM 36 force field. The temperature was maintained at 298 K using Langevin Integrator. Lennard-Jones interactions were progressively switched off between 10 and 12 ? via a force-based switching function. Long-range electrostatic interactions were computed using the particle-mesh Ewald method with an error tolerance of 10-5. We simulated the SF model and its interaction with TA in a prior publication. DT model parameters followed previous research. Surface models for common (001) facets at pH 5 were employed. Configuration of the TA molecule was optimized using the xTB 6.3 program’s approximate normal coordinate rational function optimizer (ANCopt). For the simulation setup, 40 configurationally optimized TA molecules, SF, and DT were dissolved in ~12,000 TIP3P water molecules. Additionally, 0.15 M of sodium chloride was added for box neutralization. The equilibration phase involved fixing the X and Y dimensions of the simulation box with compressibility set to zero, while pressure in the Z dimension was maintained at 1 bar using the Parrinello-Rahman barostat. Simulations spanned over 30 ns until the Z dimension of the box stabilized. The production run employed constant NVT dynamics with a 2-fs time step for more than 200 ns, discarding the first 5 ns for subsequent analysis. In vitro biocompatibility and cytoprotective effect of ST hydrogels The human dental pulp stem cells (HDPSCs) were extracted from third molars, caries-free and of adults between 18- to 25- years old. The isolation and culture of HDPSCs were conducted according to previous studies.[48,49] Initially, the molars were preserved and flushed with phosphate-buffered saline (PBS) containing 2% penicillin-streptomycin. Under the protection of a double layer of sterile gauze, the pulp chamber was crushed with a hammer to expose the pulp tissue. The pulp tissue was then removed with forceps and rinsed several times, the pulp tissue was then cut into 1 mm cubes and transferred to a T25 flask containing a DMEM solution supplemented with 20% fetal bovine serum (FBS) and 1% penicillinstreptomycin. The flask was inverted and incubated at 37°C in a humidified atmosphere of 5% CO2 for 2 h, after which it was gently returned to an upright position. The cultured medium was changed every 2 days. When the cell grows fusion degree to 80~90%, the cells were digested and cultured for serial passage. Passage 3-6 HDPSCs were conducted for cytotoxicity and biocompatibility tests. The biocompatibility and DT sealing ability of ST10 were assessed by CCK-8 assay and live/dead cell staining with a modified transwell dentin disk (modified-TDD) model.[29] Figure 5A illustrates the experimental setup, where a 1 mm thick dentin slice replaced the Transwell chamber membrane (24-well). The edge between the dentin slice and the chamber was sealed with fluid resin. The groups were as follows: the blank group (Titanium chip mimicking complete DT sealing dentin), the ST10 group (ST10-treated dentin specimens) and the control group (EDTA-etched dentin specimens). To preliminarily evaluate the sealing effectiveness, 100 μL of 1 wt% citric acid aqueous solution was added to the upper chamber, while 500 μL of 1 wt% litmus aqueous solution was added to the lower chamber to visualize the pH shift from neutral (violet) to acidic pH (red) over time. In each well, 100 μL of 1 wt% citric acid aqueous solution was added to the upper chamber, while a total of 10,000 cells were seeded in the lower chamber with DMEM containing 10% FBS and 1 wt% fetal bovine serum as the medium. At different time points (1, 3, 5 and 7 d), 500 μL DMEM medium containing 10% CCK-8 solution was added into lower chambers per well. The number of samples in each group is 3. After 3 h of incubation, the optical density (OD) value at 450 nm was measured by a Microplate Reader (KHB ST-360, Kehua Bio-Engineering, China). Additionally, the live/dead cells of HDPSCs were evaluated by a fluorescence microscope. At different time points (1, 3, 5 and 7 d), HDPSCs in each well were washed with PBS, stained with 500 μL of FDA/PI for 15 min, and washed with PBS five times. The number of samples in each group at each checkpoint time is 3. The stained HDPSCs were observed by an inverted fluorescence microscope (IX71S1F-3, OLYMPUS, Japan). Animal experiments The semi-in vivo animal study was approved by the Animal Ethics Committee of Chengdu Dossy Experimental Animals Co., Ltd., China (No: Dossy20230508001). Male SpragueDawley (SD) rats, aged 8 w and weighing 220-300 g, were selected for the study, with ten rats assigned to each group (control and ST10 group). The human dentin specimens were prepared as oval slices with a diameter of 4 mm and a thickness of 1 mm, for which two holes were punched in the distal-middle section on both sides of the slices for fixation in the rat's cavity. A 0.50 mm stainless steel ligature wire was inserted between the first and second molars and through the anterior teeth of the rat, securely fixing the dentin piece to the palate. Any excess wire was trimmed after twisting. Daily measurements were taken to monitor the weight of each rat. After 21 d, the rats were euthanized and the dentin slices were collected for further measurement by SEM and Vickers hardness test. Five samples in each group were subjected to Vickers hardness testing using a microhardness tester (TMVS-1, Times High Tech, China) at 25 g load for 10 s at 5 random locations on each dentin slice specimen.[50] Additionally, major organs including the heart, liver, spleen, lung, and kidney, as well as oral organs such as the tongue, cheek, and palate, were collected for histological staining. Fresh blood samples were also collected for hematology parameter analysis. A volume of 1 mL of blood was collected from each rat to perform the complete blood panel assay, which encompassed the analysis of parameters including white blood cells (WBC), red blood cells (RBC), mean corpuscular volume (MCV), platelets (PLT), mean corpuscular hemoglobin (MCH), hemoglobin (HGB), hematocrit (HCT), and mean corpuscular hemoglobin concentration (MCHC). Following recent research, we employed an in vivo animal study involving three healthy male New Zealand white rabbits, aged 12 w and weighing 2.1-2.5 kg. Anaesthesia was induced using 10 wt% chloral hydrate, after which labial enamels of maxillary and mandibular incisors were carefully removed using high-speed turbine handpieces with water spraying. This procedure effectively exposed the dentin, simulating the DH model. To further mimic DTs exposure, the exposed dentin underwent a 30 s etching with 37% phosphoric acid, followed by thorough rinsing with deionized water. In the experimental group, the demineralized maxillary incisors were treated with the ST10 hydrogel, while the mandibular incisors served as the control group. After a period of 1 h, the rabbits were humanely sacrificed, and both maxillary and mandibular incisors were extracted for SEM analysis. The in vivo animal study was approved by the Animal Ethics Committee of Chengdu Dossy Experimental Animals Co., Ltd., China (No: Dossy20230716001). Statistical analysis The data are presented as mean ± standard deviation (SD) from a minimum of three repeated experiments. After confirming homoscedasticity assumptions, we conducted one-way ANOVA followed by a Tukey post-hoc test across groups at = 0.05. Statistical analyses were performed using GraphPad Prism 9.0 and Origin 2021 software (OriginLab, USA). Statistical significance is denoted as ns (not significant), *p < 0.05, **p < 0.01, ***p < 0.001. Acknowledgements The authors would like to thank Dr Chao He from the College of Polymer Science and Engineering, Sichuan University for the SEM micrographs. We are grateful for the financial support of the National Natural Science Foundation of China (Nos. U22A20158, 52273135, and 51973133), Sichuan Science and Technology Program (No. 2023NSFSC0999), the Fundamental Research Funds for the Central Universities, Sichuan University (No. 2022SCU12015), Key R & D projects of Guangxi Science and Technology Department (No. 2021AB11097), and Science and Technology Plan Project of Qingxiu District, Nanning City, Guangxi (No. 2020039). Conflicts of interest The authors declare that they have no known competing financial interests or personal relationships that could have appeared to influence the work reported in this paper. Data Availability Statement The data that support the findings of this study are available from the corresponding author upon reasonable request. Source: doi: 10.1002/adhm.202303153 |