Recent Advances in Adhesive Materials Used in the Biomedical Field: Adhesive Properties, Mechanism, and ApplicationsIssuing time:2025-09-12 09:48 Yongping Lu,a Xinyuan Xu and Jianshu Lee Abstract Adhesive materials are natural or synthetic polymers with the ability to adhere to the surface of luminal mucus or epithelial cells. They are widely used in the biomedical field due to their unique adhesion, biocompatibility, and excellent surface properties. When used in the human body, they can adhere to an accessible target and remain at the focal site for a longer period, improving the therapeutic effect on local disease. An adhesive material with bacteriostatic properties can play an antibacterial role at the focal site and the adhesive properties of the material can prevent the focal site from being infected by bacteria for a period. In addition, some adhesive materials can promote cell growth and tissue repair. In this review, the properties and mechanism of natural adhesive materials, organic adhesive materials, composite adhesive materials, and underwater adhesive materials have been introduced systematically. The applications of these adhesive materials in drug delivery, antibacterials, tissue repair, and other applications are described in detail. Finally, we have discussed the prospects and challenges of using adhesive materials in the field of biomedicine. 1. Introduction Anadhesive material can join two separated substances together and prevent their separation. As one of the most important auxiliary materials, traditional adhesive materials have been widely used in a variety of industrial production and manufacturing processes, including, but not limited to, packaging operations, automotive manufacturing, and industrial coating or fillers. Their application in the biomedical field is very important. They are widely used in biomedicine due to their unique adhesion, biocompatibility, and excellent surface properties. Biomedical adhesives can be divided into external and internal adhesives according to their applications in the human body. The most important property of an adhesive is its adhesion ability. There are two types of adhesive forces: internal and interfacial. Internal adhesion forces refer to the internal forces of the adhesive material itself and the adhesive strength inside the material, which has nothing to do with the application sites. Interfacial adhesion refers to the action of the forces between the materials and different interfaces to achieve the adhesive effect. The external adhesion force mainly involves the interaction of intermolecular forces, which may involve ionic bonds, covalent bonds, hydrogen bonds, dipole interactions, and van der Waals forces. The interfacial adhesion of general materials ranges from 0.01 to 6 MPa. Due to their strong interface and intrinsic adhesive effects, adhesive materials have attracted a lot of research attention in the biomedical field, especially toward the treatment and repair of physical injuries. When compared with traditional sutures and surgical screws, adhesive materials have many unique advantages in the biomedical field, mainly in the following aspects: (1) simple operation, which can effectively shorten the operation time, (2) minimize the treatment of injury and relieve the pain of patients, and (3) prevent fluid leakage and reduce secondary damage. Currently, 90% of medical adhesives are applied to external wounds in the form of wound excipients. This may be attributed to the functional needs of adhesives in the same industry being similar. In addition, traditional medical adhesives have limited functional requirements and usually only partially satisfy the required mechanical properties, such as bond strength. In particular, adhesives often require a variety of special properties to meet the complex medical requirements of biomedical applications. With the continuous development of synthetic chemistry and biomedical engineering technology, some adhesive materials prepared from the perspective of bionics have shown good biocompatibility and biodegradability, which can expand their application potential in biomedicine. Over the past few decades, although the material and form of adhesive have changed significantly, the essence and most basic requirements for application in the field of biomedicine have not changed significantly, and mainly include the following aspects: (1) physical protection of wounds, (2) accelerated wound healing, (3) provision of physical support, (4) prevention of infection, and (5) filling of cavities. In this review, we systematically discuss the properties and mechanisms of natural adhesive materials, organic synthetic adhesive materials, composite adhesive materials, and underwater adhesive materials used in the design of biomedical materials. In addition, the applications of these adhesive materials in drug delivery, antibacterials, tissue repair, and other areas have been reviewed in detail (Scheme 1). Finally, the perspectives and challenges associated with the clinical application of adhesive materials are discussed in detail in the field of biomedicine. Many reviews of adhesive materials have been published, such as the use of underwater adhesive materials, adhesive materials in drug delivery, and adhesive materials used in antibacterial or tissue repair applications. However, they emphasize only a few adhesive materials and their applications. Ourreviewsystematically discusses a series of adhesive materials and their applications in the biomedical field.
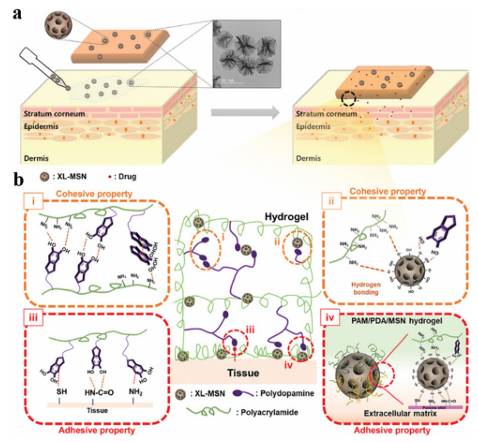
Scheme 1 A systematic overview of adhesive materials and their potential applications in the biomedical field. Reproduced from ref. 32, 39, 43 with permission from American Chemical Society. Reproduced from ref. 33, 36, 41 with permission from Elsevier. Reproduced from ref. 38, 42 with permission from Springer Nature. Reproduced from ref. 34, 35, 37, 40 with permission from John Wiley & Sons. 2. The main classifications of adhesive materials 2.1 Natural adhesive materials Bio-based adhesive materials are natural materials produced by organisms found in nature. According to their composition, they can be divided into protein-adhesive materials and polysaccharide-adhesive materials. In nature, many invertebrates secrete sticky proteins, such as mussel sticky proteins. Theseproteinshavegoodbiodegradability and biocompatibility, achieve successful adhesion between cells and tissues, and have been applied toward the adhesion of skin, mucosa, cartilage, bone, and other wounds, and surgical incisions. Fibrin binding material is a very important protein binding material, which comes from plasma and is mixed with fibrin and thrombin substances. Fibrinogen is cleaved into fibrin peptides A and B, and thrombin activator V3 promotes their cross-linking and the final formation of clots In addition, collagen adhesive materials are an important component in protein adhesive materials, which account for approximately one-third of the total protein content in mammals, and have good biological activity, biocompatibility, and biodegradability, as well as promote wound healing. They are widely used in medical cosmetology, hemostatic repair, tissue stents, drug slow release, etc. However, there have been few reports on their use as a tissue binder. Chitosan (CS) is a conventional polysaccharide-binding material obtained via the deacetylation of chitin, which is widely present in nature. This adhesive material has excellent biocompatibility, biodegradability, and hemostatic and antibacterial properties, and has been widely used in clinical applications as a hemostatic excipient. For example, HemConTM is a hemostatic bandage approved by the Food and drug administration (FDA). Because CS contains a large number of amino groups, it can only exhibit an adhesion effect via a chemical conjugation reaction upon exposing its primary amine groups under alkaline conditions. CS has a positive charge under physiological conditions and exhibits poor adhesion to tissues when used alone. In addition, CS is difficult to dissolve in the humoral environment and therefore, its adhesion in the human body is not high. In addition to CS, alginate (broken bone binder) and starch (tablet binder) have been widely used as biomedical adhesive materials. Gelatin is the degradation product of collagen, which has good biocompatibility and biodegradability, which can form transparent, flexible, and high-strength glue. However, its strong hydrophilicity makes the structure unstable, so it is often necessary to cross-link modified gelatin for use under physiological conditions. Although most bio-based adhesive materials obtained from natural ingredients have good biocompatibility and biodegradability, they also have some drawbacks, such as low yield, high cost, and insufficient adhesive strength. At present, most research on bio-based adhesive materials has focused on improving the performance of natural-based adhesive materials and their availability as adhesive materials by modifying them or combining them with other synthetic materials, such as new superhydrophobic interface materials based on lysozyme phase transition coatings. 2.2 Organic synthetic adhesive materials Most of the earliest chemical adhesive materials synthesized by researchers were polymeric materials, which play a very important role in the adhesion field. For example, polyethylene glycol (PEG) is one of the most commonly used biocompatible adhesives and there are many PEG-based adhesives available on the commercial market. Because the structure of PEG is easy to modify, it can be altered to change its physical properties. PEG is commonly used as a sealant to prevent leakage of liquid and air after surgery. At present, the development of synthetic chemical adhesives mostly involves the modification of existing medical polymeric materials and combining them with different chemical substances to make them have more abundant functionalities. However, when compared to existing chemical polymeric materials, biomaterial-derived materials have better properties and are suitable for application in biomedicine.The combination of chemical polymers and biological components may become the mainstream of the future development of chemical adhesive materials. In the mid-20th Century, Coover developed a cyanoacrylate medical tissue adhesive, also known as Superglue, which is one of the strongest and most versatile adhesives available today. It is a liquid monomer and anionic polymerization occurs in the presence of nucleophiles, especially hydroxide groups including hydrated blood, rapidly in a matter of seconds. The low pre-curing viscosity (easy to spread and low volume per unit area), high bond strength, colorless transparency after curing, and low shrinkage has led to its widely use in emergency rooms, dermatology, and plastic surgery. However, the heat of polymerization generated by the rapid polymerization of this type of organic synthetic adhesive can easily cause secondary damage to the tissue, poor elasticity and flexibility after curing, short storage period, and easy solidification limit its widespread use in biomedicine. 2.3 Compound adhesive materials Natural biological binders are not conducive to application in the biomedical field due to their large individual differences, difficult preparation, high cost, and poor mechanical properties. However, the defects of chemical synthetic adhesives often result in the poor elasticity of the adhesive sites, rejection of living tissues, and potential chemical toxicity, which are not conducive to their application in the biomedical field. Therefore, after modifying some of their functional sites using chemical or bioengineering methods, an increasing number of composite adhesive materials have been prepared, including composites formed between viscous biomolecules, composites between biomolecules, and chemical modification. There are many methods used to prepare composite adhesive materials, which can be roughly divided into physical mixing, modular gene, and chemical grafting methods. 2.4 Underwater adhesive materials Traditional adhesives are usually based on natural biological components or organic solvents, which can only be used in dry or low-humidity environments. However, adhesives often need to be able to bond effectively in an aqueous environment for tissue adhesion in biomedical applications. However, conventional commercially available polymer adhesives are often weakened or destroyed in an aqueous environment. The good wettability of the mucous membrane surface of an organisms tissues makes swellable polymeric materials come in close contact with water; the molecular chain segment of the adhesive material embeds in the cell space or penetrates the viscous chain segment in the mucus through a combination of mechanical mosaics, covalent bonds, electrostatic attraction, van der Waals forces, hydrogen bonds, and hydrophobic bonds, which bind tightly to the mucosa, resulting in biological adhesion, and can be maintained for a considerable period of time. To solve this problem, several researchers have designed a series of underwater binders that mimic naturalbiologicalbindersthat exist underwater. Theadhesivestrength is relatedto the charge density, molecular weight, molecular space configuration, swelling degree, solubility, and concentration of polymeric materials. In addition, the surface polarity of the polymer, flexibility of the polymer chain, pH value of the drug sites, and amount of mucus also have some impact. 3. Application of different adhesive materials in the biomedical field 3.1 Drug delivery At present, research on adhesive materials in the drug delivery phase mainly aims at precise targeting and dosing, and low toxicity in order to achieve the best therapeutic effect. Adhesive materials can provide space and time control for the release of various therapeutic drugs, including small-molecule and macromolecule drugs, and cells. Adhesive materials have the characteristics of adjustable physical properties, controllable degradability to protect unstable drugs from degradation, and the use of a platform for various physical chemistry interactions between the adhesive material and encapsulated drug, which can also reduce the administration frequency for convenient medication. Polyphenols have become important multifunctional materials used in novel drug delivery systems, both as the carrier components for drug delivery and therapeutic agents to intervene in disease progression. Due to their unique molecular structure, polyphenols can bind to proteins, metal ions, polymers, and nucleic acids, which contributes to their use as important functional components in other drug delivery strategies. In addition to being an integral part of the vehicle, polyphenols are also effective in protecting the structural integrity of nanoparticles (NPs) and the biological activity of the medical intervention compounds carried by them, such as by acting as a protective layer to protect nanomedicines from oxidation and improve carbohydrate polymer carriers. As a biological ligand, curcumin (Cur) is a natural polyphenol active molecule. Luo et al. filled Cur into polyvinyl alcohol (PVA) hydrogels containing phytic acid (PA) to prepare wood-derived hydrogel composite membranes, which can not only exhibit an obvious heterogeneous structure and good mechanical properties, but also release the loaded drugs sustainably to obtain long-term biological activity Furthermore, they found that the Cur-PA-loaded composite membranes significantly inhibit bacterial growth and inflammation, and promote adhesion, proliferation, and osteogenic differentiation of bone marrow mesenchymal stem cells. Cheng et al. proposed the delivery of proteins with different molecular sizes and the isoelectric point-specific recognition of natural polyphenols and boric acid-containing polymers. Natural polyphenols tightly bind to various biomolecules, nucleic acids, and cell membranes via hydrophobic/hydrophobic interactions, and the protein molecules are modified by polyphenols via non-covalent hydrophobic/hydrophobic interactions or reversible dynamic covalent bonds. Natural polyphenols effectively increase the binding affinity between proteins and polymers containing boric acid. The pH-sensitive properties of catechol–borate allow for the release of the bound proteins in an acidic environment, thereby greatly improving the efficiency of cytosolic delivery. They demonstrated this by delivering a variety of proteins, including bovine serum albumin, phycoerythrin, and ribonuclease A. This polyphenol-containing polymer platform displays high efficiency and maintains protein bioactivity after its intracellular release. For the adhesion of hydrogels to the skin surface, Kim et al. proposed a tissue adhesive hydrogel comprised of a polyacrylamide/polydopamine (PAM/PDA) hydrogel embedded with super-macroporous mesoporous silicon dioxide NPs (XLMSN). Due to the adhesion of XL-MSN at the interface, the application of XL-MSN at the hydrogel-skin interface can further enhance the adhesion (Fig. 1). They loaded the model drug into a patch-embedded XL-MSN and used the resulting hydrogel patch to demonstrate transdermal drug delivery through pig skin. The results show that this approach is simple and effective for the transdermal delivery of adhesive hydrogel patches that can enhance the strength and adhesion to skin.
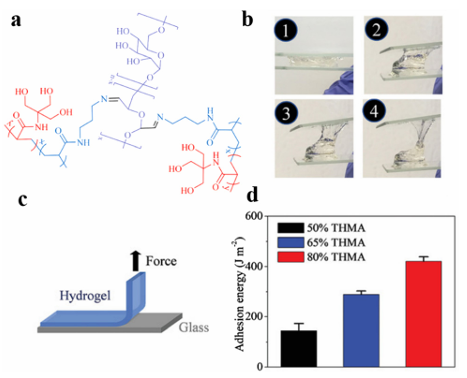
Fig. 1 (a) A schematic representation of an adhesive hydrogel patch with enhanced cohesive and adhesive properties on skin tissue. (b) A schematic representation of the four characteristic molecular interactions associated with enhanced (i and ii) cohesive and (iii and iv) adhesive properties. Reproduced from ref. 35 with permission from John Wiley & Sons. Zhang et al. integrated the molecular structure of mussel adhesion proteins, in which the microscopic structure of octopus tentacles, polymyxa polymyxin, and PDA hydrogel were selected as a flexible base, and a multi-functional microneedle (MN) for practical application was proposed. In addition, due to the PDAhydrogel having a similar molecular structure to that of the adhesion protein, it endows the resulting MN with strong adhesion properties. The results showed that the MN releases the drug rapidly for the first 8 hours, then slows down gradually until it reaches equilibrium after 2 days, accounting for about 80% of the total release, in the process achieving the desired sustained release of the drug at the appropriate release rate and amount. MN obtained from this experiment performed well for continuous drug delivery in a rat osteoarthritis model and can provide new and promising insights into transdermal drug delivery in any part of the body. CS is a valuable drug delivery agent due to its excellent biological and adhesive properties. Due to its advantages of stability, good biodegradability, and low toxicity in the stomach, CS is often used to protect unstable drugs, such as polyphenols, from gastrointestinal enzyme (GIT) degradation in the gastrointestinal tract, and to enhance the absorption of therapeutic agents. CS binds closely to polyphenols via electrostatic interactions, which limits the accessibility of polyphenols in the stomach and small intestine, thereby improving the bioavailability of polyphenols in the presence of low pH and various digestive enzymes. Wen et al. investigated the use of CS–alginate nanofibers constructed using coaxial electrospinning for the colon-specific delivery of quercetin. They first prepared sodium triphosphate ion-cross-linked CS microstructures to encapsulate quercetin. An electrospun fiber mat was then formed by adding sodium alginate. In vitro experiments showed that the electrospun mats degrade the fibers in a controlled manner (73%) via the enzymatic action of bacterial glucosidase in a simulated colon environment. Due to the CS, the delivery system exhibits strong mucosal adhesion properties, which contribute to the prolonged carrier residence time and release of quercetin in specific regions of the GIT. Han et al. developed a series of multifunctional mucosal adhesives based on the borate reaction of chitosan grafting dihydrocaffeic acid and the oxidation of pullulan polysaccharide, and prepared pH-responsive hydrogels as injectable local drug delivery carriers by mixing the solution of chitosan grafting dihydrocaffeic acid and the oxidation of amylopectin.36 When amoxicillin was encapsulated in the hydrogel as a model antibacterial drug, the resulting hydrogel showed a good drug release capacity, effectively killing colon tumor cells (HCT116 cells). In addition to its good mucosal adhesion, the hydrogel exhibited in vitro antibacterial properties against E. coli and Staphylococcus aureus, which has potential use in mucosaltargeted drug delivery systems. Stevenson et al. used genistein-crosslinked catechol-modified CS(Cat-CS) hydrogels for rectal administration to treat ulcerative colitis.130 Catechol can be used to enhance the mucosal adhesion of polymer materials by forming covalent and non-covalent bonds with mucin. Such mucosal adhesion drug delivery systems can adhere to mucosal tissues to extend the local retention time of drugs. They evaluated the efficacy of sulfasalazine (SSZ) loaded with Cat-CS gel in an ulcerative colitis mouse model. When compared with the oral treatment of SSZ, the rectal SSZ/ Cat-CS delivery system is more therapeutic and induces lower plasma concentrations of potentially toxic byproducts. These results suggest that the SSZ/Cat-CS rectal hydrogel was more effective and safer than the oral administration of SSZ. CS derivatives are a new-generation variant of CS. These modified CS derivatives have overcome the limitations of CS and have madegreat progress in the field of drug delivery. As delivery carriers, drug release from CS and its derivatives follows a conventional protocol applicable to CS. They are usually provided in the form of microspheres, NPs, micelles, and gels. Shaw et al. designed CS-coated polylactic acid–glycolic acid copolymer (PLGA) NPs embedded in CS–alginate hydrogels used for the colon delivery of curcumin. The preparation was also prepared with hyaluronic acid and CD98 siRNA (siCD98), which showed therapeutic effects by protecting the mucosa and relieving inflammation. In addition, the drug prepared using CS exhibited sustained- and controlled-release effects, which not only enhance the drug stability, but also extend the drug effect. Tang et al. prepared self-assembled NPs composed of CS, edible polypeptides, and poly LRB-g-glutamic acid (g-PGA) used for the delivery of tea catechins. As a type of adhesive macromolecule, CS can enhance the transport of ( )-epigallocatechin-3-O-gallate (EGCG) and catechin in the jejunum of mice. A sustained free radical scavenging test showed that the NPs retained the antioxidant activity of the tea catechins. NPs with a positive surface charge can instantaneously open tight junctions between caco-2 cells, thus increasing the cellular bypass transport of tea catechins. These results indicate that CS/g-PGA NPs can beeffectively used as oral delivery carriers for catechins with potent antioxidant activity. Chen et al. reported a novel adhesive hydrogel with a high hydrogen bond density that utilizes the load-sharing effect of "triple hydrogen bond clusters" (THBC). When THBC is incorporated into the hydrogel matrix as a side group, it causes the hydrogel to adhere firmly to a range of surfaces, including glass and tissue, even without any chemical reaction. The key ingredient, a copolymer of N-[tris(hydroxymethyl)methyl]acrylamide (THMA) and N-(3-aminopropyl)methacrylamide hydrochloride (APMA), promotes crosslinking and salt displacement (Fig. 2). They demonstrated a biodegradable hydrogel bandage based on this adhesive hydrogel in an in situ liver cancer model that encapsulated serum albumin (HSA)-coupled cisplatin NPs (HCP), which adhered to the liver prior to degradation and continuously delivered directly to the tumor site, and can also be used to enable cell replacement therapy for type 1 diabetes (T1D). The results showed that the use of a "load-sharing" adhesive hydrogel as a tissue bandage for drug delivery results in a significant improvement in the therapeutic efficacy when compared to systemic injection, and the tumors in the orthotopic liver cancer mouse model studied were almost completely suppressed, and the diabetes of chemically induced diabetic mice recovered for one month.
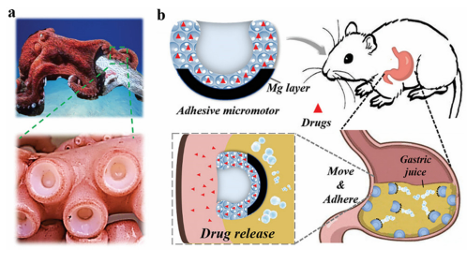
Fig. 2 (a) A schematic representation of p(APMA-co-THMA) cross-linked with dexamethasone (Dex)-CHO used to form a biodegradable adhesive hydrogel. (b) The adhesive properties of the hydrogel between two glass slides. (c) A schematic representation of the peeling test. (d) Average adhesion of hydrogels prepared with different THMA contents to a glass slide.39 Reproduced from ref. 39 with permission from American Chemical Society. In addition, Yang et al. recently reported that a selfexpanding, endoscopically deliverable, and viscous hydrogel can be used to prevent gastric perforation. They found that the silicon dioxide coating significantly increased the adhesive strength of the hydrogel in a simulated highly acidic stomach environment. Acid-resistant, tough, and elastic double-network hydrogels were designed using dimethylacrylamide (DMA) and biodegradable cross-linking agents (N,N0-cysteamine bis (acrylamide) (CBA) and sodium alginate (SA)) used to crosslink calcium ions. The resulting double network hydrogels were cast into thin slices and rolled to construct a multilayer hyperboloid cap-rod device with an onion-like structure. Silicon dioxide NPs were coated onto the external surface of the device to allow for strong and rapid adhesion, and prevent perforation defects. The hyperboloid device continuously released the drug to modulate the gastric pH and promote wound healing after loading with voronazan fumarate (VF) and acidic fibroblast growth factor (AFGF). When applied to acute gastric perforation models in rabbits and miniature pigs, the gel device can be compressed and expanded in a mushroom-like manner. Using the Wei capsule robot to remotely monitor the pH and immunohistochemical analysis, they demonstrated that the compressible hyperbolic gel could stably block the perforation and promote wound healing over a 28 day observation period. Park et al. successfully developed a bifunctional GTA-ob-CD hydrogel by mixing gelatin-tyramine (GTA) with oxidized b-cyclodextrin (ob-CD).116 Their adhesion and mechanical strength were enhanced by the double cross-linking and Schiff base reactions. They found that the optimal composition of the GTA-ob-CD hydrogels was 5 wt% GTA (GTA5) and 1 wt% ob-CD, and their adhesion was 2.3- and 6.2-fold higher than that of the GTA-only hydrogel and fibrin gels, respectively. Due to the extra imine bond, their elastic modulus and degradation rate were 1.8- and 1.5 times higher than those of the GTA hydrogel, respectively. Because of the hydrophobic cavity in ob-CD, the release efficiency of the hydrophobic drug dexmedetomidine was 2.7 times higher than that of the GTA-only hydrogel. With these advantages, the hydrogel has a controllable gelation time, controlled drug release, good cell compatibility, and can improve the uniformity of hydrophobic drug loading. Therefore, it is hoped that it will be a good platform for hydrophobic drug delivery. Wang et al. developed a buccal tissue adhesive made from PVA polymer and mussel adhesion protein 3,4-dihydroxy-Dphenylalanine (PVA–DOPA membrane). Three types of polymers, PEG, PVA, and PDA, were used to assemble core–shell PLGA NPs with different surface coatings, and a combined buccal drug delivery system (PVA–DOPA@NPs film) was constructed (NP refers to PLGA, PLGA–PEG, PLGA–PVA, or PLGA–PDA NPs). This type of composite mucosal adhesive membrane has a strong adhesive force in the oral wet environment, which can leave enough residence time and control, and extend the drug release curve. It also has the potential to be applied via the buccal pathway in other diseases. Lee et al. developed a wearable adhesive skin patch for transdermal delivery using a flexible microneedle, dry adhesive, and frictional electrical energy collector (TEH). The tips of the flexible microneedles were constructed from epoxy SU-8 or maltose. The maltose tip can be transported when it melts after skin penetration, and to prevent the evaporation of water from the solvent from causing the maltose tip to melt, water-soluble pharmaceutical preparations are stored in patches with SU-8 tips. The flexible array of microneedles with a soft base and hard tip can withstand the deformation associated with skin stretching without breaking when applied to skin areas, such as the elbow. A dry adhesive in the skin patch was used to secure the entire device to the skin surface for common adhesion and provide a pulling force that separates the TEH patch from the skin. Wang et al. developed a nanotechnology-based hydrogel barrier consisting of silicate nanoplates and polyethylene glycol that can self-assemble a network of constituents to form a nonNewtonian hydrogel barrier system for frictionless covering of the cardiac outer membrane and self-healing drug delivery. In vitro and in vivo experiments showed that this new nanostructured hydrogel drug delivery system exhibits a certain degree of cell adhesion and can act synergistically to prevent cell infiltration, as well as local immunomodulation to protect the intrapericardial space after surgical intervention. Liao et al. developed an aptamer-functionalized composite nano-object (Apt-ADMC) using small gold NPs (AuNP) as adhesive nanobelts to fold a long string of aptamer-attached tetrahedral DNA pearls (Apt-Nano-Tetra) into a spherical multicenter DNA nanostructure used for the targeted phase drug delivery at the tumor cell nuclei in vivo; the capacity of doxorubicin (Dox) was increased 75–85 times. After Dox-Apt-ADMC specifically enters the target tumor cell, it disintegrates into individual AuNPs surrounded by short Apt-Nano-Tetra, eventually reaching the nucleus and causing cell apoptosis. Experiments demonstrated that Dox-Apt-ADMC treatment using fetal bovine serum for 12 hdidnotimpairthe drugdelivery capacity, contributing to the high functional stability of the circulatory system. Systemic administration of Dox-Apt-ADMC resulted in a 7-fold enhancement in tumor accumulation and almost 100% inhibition of tumor growth without detectable systemic toxicity. Currently, the development of systems with satisfactory adhesion and adequate bioavailability under wet conditions remains challenging. A large number of research studies on bionics, together with mussels, barnacles, octopus, and castle worms, have been carried out on the secretion of similar adhesive substances and marine organisms. Inspired by the Boston Ivy, Zhao et al. proposed a novel particle with outstanding adhesion ability due to the shape of the Boston Ivy tendrils attachment disk and the interlinked surface patterned nanostructure as a drug delivery vector. By replicating silicon dioxide colloidal crystal aggregates assembled in droplet templates after rapid solvent extraction, they produced particles with unique shapes, nanostructures, and viscous hydrogels. The results showed that the new microparticles exhibit remarkable adhesion in a wet tissue environment. When dexamethasone was loaded into these adhesive particles, they were more firmly anchored to the inflamed areas of the body than spherical particles, thus prolonging the drug release process. Therefore, they exhibit a good therapeutic effect on ulcerative colitis. Chai et al. proposed a novel viscous micromotor for drug delivery in the stomach based on the physical structure of an octopus sucker. The micromotor was made using a hydrogel to duplicate the structure of the sucker-like particles, and magnesium (Mg) then loaded on the spherical surface at the bottom to obtain a bubble-powered hydrogel micromotor with adhesive properties (Fig. 3). When exposed to gastric juice, the Mg-loaded micromotors can move spontaneously because of the continuous production of hydrogen bubbles by the Mg/H+ reaction. Due to its unique sucker-like structure and spontaneous movement, the micromotor can effectively move and adhere to tissues to release drugs, thus enabling more efficient drug delivery. In addition, the hydrogel micromotor has a high volume-to-surface area ratio due to its porous structure, which can achieve a high drug-loading efficiency. The results showed that the micro-motor could stay in the stomach for 412 h because of its excellent adhesive properties and showed an ideal therapeutic effect on gastric ulcers.
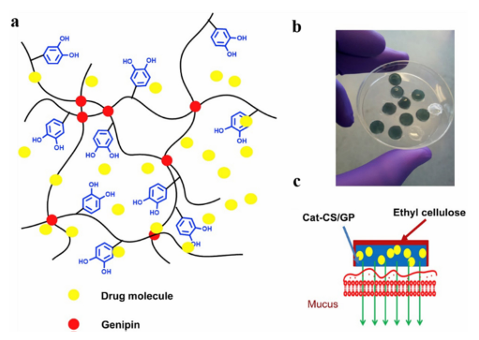
Fig. 3 A schematic representation of micro-motors with adhesive properties. (a) Pictures of octopi and their suction cups. (b) An adhesive micromotor acting as a drug carrier in the stomach.34 Reproduced from ref. 34 with permission from John Wiley & Sons. Gu et al. also developed a natural mussel adhesion protein (NMP) with tumor-environmentally responsive bioadhesion by binding NMP to a phenyl boric acid-containing prodrug, tirazamine, to construct a phototherapy–chemotherapy composite nano-drug delivery system. It is characterized by the efficient encapsulation of the photosensitizer and pre-chemotherapy drug by the structural characteristics of mussel foot silk protein, and the biological activity of the protein was controlled by the drug molecule as the regulatory unit, which can carry drugs to break through the endogenous and pathological barrier, and carry out targeted residence and penetration at the tumor site. On this basis, the tumor environment-responsive release of drugs, activation of prodrugs, and photodynamic and hypoxia-driven chemotherapy play a synergistic therapeutic role. Park et al. have reported a new class of thermosensitive injectable hyaluronic acid (HA)/Pluronic F127 composite tissue adhesive hydrogels. The dopamine conjugated HA (HA–DN) was mixed with a sulfhydryl-modified Pluronic F127 copolymer to form a slightly cross-linked HA pluronic gel structure based on the Mikell 1,2-dihydroxybenzene thiol addition reaction. Rheological studies showed that the sol-gel transition was rapid and reversible in response to temperature. Thus, Ha/Pluronic hydrogels can be injected into the body in the sol-state at room temperature, but become stable in the gel-state immediately at body temperature. Ha/Pluronic hydrogels exhibit excellent tissue adhesion due to the strong adhesion of o-diphenol and curing of o-diphenol-mercaptan via chemical crosslinking. HA/ F127 hydrogels can adhere to serous tissues near the parietal peritoneum, providing more possibilities for bioadhesive materials for drug delivery. Under moist conditions, adhesive materials are often used to hold the lining of the mucosa long enough for the drug to be released and absorbed. Cerutti et al. developed an oral drug delivery system using a novel catechol-functionalized CS (CatCS) hydrogel. The catechol functional groups were covalently bonded to the CS backbone and crosslinked with a non-toxic crosslinker, genipin (GP), to achieve catechol hydrogels with 9 and 19% binding, respectively (Fig. 4). The gelation time and mechanical properties of the Cat-CS hydrogel were similar to those of the CS-only hydrogel, but the cat19-CS hydrogel can still adhere to porcine mucosa after 6 h, whereas the CS-only hydrogels lost contact with the mucosa after 1.5 h. In addition, the hydrogel system can release lidocaine continuously for B3 h due to its close contact with the wet mucosa. These results suggest that the catechol-modified CS hydrogel proposed in the study is a promising oral drug delivery system for mucosal adhesion.
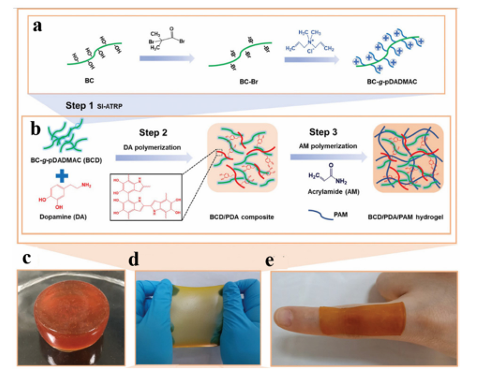
Fig. 4 Cat-CS/GP hydrogel: (a) schematic representation of the Cat-CS hydrogel network comprised of GP crosslinking. (b) Cat-CS/GP hydrogel with ethyl cellulose protective cap. (c) Schematic representation of drug release from encapsulation in the hydrogel on the mucosal surface. Reproduced from ref. 33 with permission from Elsevier. 3.2 Antibacterial applications The development and application of antibacterial materials have been limited by the difficulty of achieving long-lasting antibacterial action and the short duration of antibacterial action in sports wounds. Adhesive materials are widely used to inhibit bacteria in wounds due to their inherent properties, such as biocompatibility and strength. Therefore, they can be used as a carrier for antibacterial agents. Polyphenols inhibit the growth of microorganisms, particularly bacteria. They can cause morphological changes in microorganisms, destroy bacterial cell walls, and affect biofilm formation, resulting in irreversible damage. Polyphenols also affect protein biosynthesis, alter metabolic processes in bacterial cells, and inhibit adenosine triphosphate (ATP) and DNA synthesis. Polyphenol mixtures are more effective than pure compounds. Therefore, current research has focused on the antibacterial properties of polyphenol mixtures rather than single compounds. Wu et al. reported the synthesis of cationic polyelectrolyte brushes grafted with bacterial cellulose (BC) nanofibers in polyacrylamide hydrogels. The PAM component has good biocompatibility and a stable cross-linked structure, so it can be used as ahydrogel scaffold, while the PDA component is rich in 1,2-dihydroxybenzene groups, has good adhesion performance, and can be attached to various surfaces. In particular, there is a need for large-scale movement of particular areas (Fig. 5). More importantly, the nanofiber (BCD) component not only has a rigid BC backbone to enhance the mechanical properties of the hydrogel, but also has a poly(diallyl dimethyl ammonium chloride) (pDADMAC) brush with a broad spectrum of low-toxicity positively charged quaternary ammonium groups, which can provide efficient and durable antibacterial properties. Therefore, the BCD/PDA/PAM hydrogel has excellent mechanical properties, stable adhesion, and long-lasting antibacterial activity, which can be used for antibacterial wound healing in particular areas requiring extensive exercise.
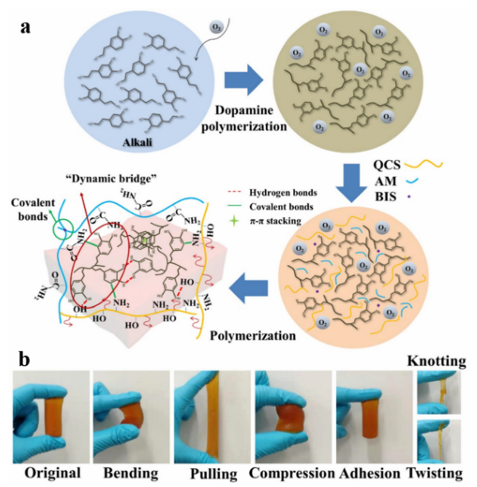
Fig. 5 A schematic representation of an antibacterial hydrogel. (a) Preparation of BCD using SI-ATRP to graft pDADMAC from BC. (b) Formation of BCD/PDA/PAM hydrogel. (c–e) 10% BCD/PDA/PAM hydrogels with different shapes. Reproduced from ref. 37 with permission from John Wiley & Sons. Based on bi-crosslinking and mussel-like chemistry, Chen et al. developed a bi-crosslinked antibacterial hydrogel.156 A series of multifunctional double-crosslinked hydrogels were prepared using a simple method with PDA used as the polyacrylamide of quaternary CS (QCS) and PAM (Fig. 6). Their antibacterial tests showed that the hydrogel killed 99.99% of colon Bacillus and Staphylococcus aureus bacteria. Moreover, the hydrogel loaded with avocado showed a pH-responsive drug release curve, which can promote the proliferation of L929 cells. Compared with pure hydrogels, hydrogels based on double crosslinking and mussel-like chemistry not only improve tissue adhesion, but also have stronger self-healing ability, which has greater application potential in biomedicine and related fields.
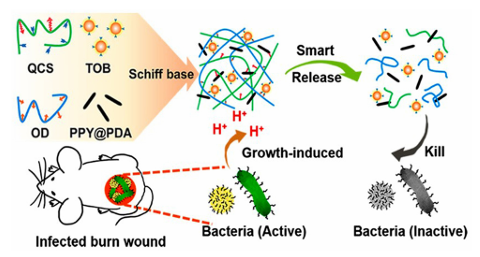
Fig. 6 Aschematicrepresentation of the synthesis of Q-DM hydrogels. (a) Synthetic process and structure representation of the Q-DM hydrogel. (b) The original, bending, pulling, compression, adhesion, knotting, and twisting shapes of the flexible Q4-DM-0.4 hydrogel.156 Reproduced from ref. 156 with permission from Elsevier. Pham et al. reported that AgNPs-gel-PEG-DA hydrogels can improve the adhesion of AgNPs by grafting catechol groups onto the AgNP backbone. At the same time, the hydrogel exhibited excellent antibacterial properties against Grampositive and Gram-negative bacteria. The hydrogel exhibits good adhesion and antibacterial activity. Using a two-step process, Han et al. developed a PDA–clay–PAM hydrogel with strong adhesion and toughness. Free catechin groups were generated in the PDA-inserted clay nanosheets by inserting DA into the clay nanosheets and partial oxidization between the layers. The acrylamide monomers were then added and polymerized in situ to form hydrogels. The hydrogels exhibit a repeatable and durable adhesive force, can adhere directly to human skin without triggering an inflammatory reaction, and are easily removed without causing any harm, which are advantages in surgical applications. Researchers have carried out a large number of antibacterial experiments on CS and their results show that CS has a good inhibitory effect on a variety of coccus, Bacillus, and fungi. During the research and development of antibacterial drugs, CS can be used as an antibacterial raw material and also help the drug to adhere to the required drug delivery site. Hu et al. have prepared a series of EGCG-crosslinked carboxymethyl CS-based hydrogels (EP gels) with inherent antioxidant, bactericidal, and adhesive properties. The hydrogel was constructed by crosslinking the phenyl boronic acid groups in carboxymethyl CS (CMCS-PBA) grafted with PBA with the glycol groups in EGCG and PEG-Glu. The mechanical properties were enhanced by introducing gluconic-acid-capped PEG (PEG-GLU) into the gel network. The resulting hydrogel was excellent at scavenging free radicals and killing Staphylococcus aureus and Escherichia coli. In addition, it has excellent self-healing properties, ideal mechanical and adhesive strength, and inherent ideal properties. Thus, it is expected to be used as a wound dressing. However, the bacteriostasis of CS is closely related to its degree of deacetylation and relative molecular weight, and its applications are limited due to its poor water solubility. Therefore, a series of modified CS derivatives were synthesized by means of alkylation, arylation, salt formation, and splicing. The antibacterial activity of the resulting CS derivatives was enhanced and the stability and bioavailability were also improved. Guo et al. prepared a series of self-repairing hydrogels (QCS/ OD/TOB/PPY@PDA) with good conductivity and antioxidant activity based on QCS, oxidized dextran (OD), and tobramycin (TOB). They first quaternized CS to obtain QCS in order to improve its water solubility and give it the ability to resist drugresistant bacteria (Fig. 7). Their antibacterial results showed that the QCS/OD/TOB/PPY@PDA hydrogel can kill high concentrations of Pseudomonas aeruginosa (PA), Staphylococcus aureus, and Escherichia coli in a short period of time, and exhibit a bactericidal effect on agar for up to 11 d. The plate diffusion test also demonstrated good antibacterial activity in vivo. The Schiff base crosslinks formed between aminoglycosides TOB and OD allow TOB to be released slowly and in response to the pH conditions. Acidic substances during bacterial growth can induce the on-demand release of TOB and avoid the abuse of antibiotics. This intelligent release hydrogel of on-demand TOB is very beneficial for wound healing during bacterial infection.
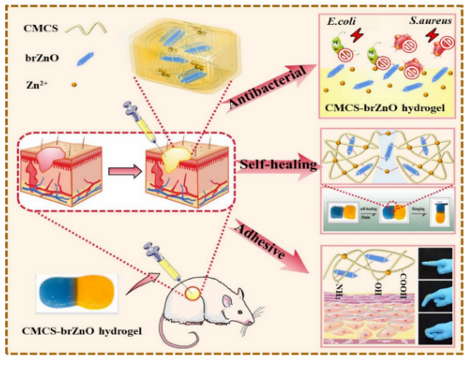
Fig. 7 A schematic representation of the synthesis of the QCS/OD/TOB/ PPY@PDA hydrogel.162 Reproduced from ref. 162 with permission from American Chemical Society. Tang et al. reported a thermosensitive self-healing hydrogel based on carboxymethyl CS and silver-containing polyoxometalates (AgP5W30POM) used as antibacterial coatings. Silver ions were encapsulated in POM cages and exhibit long-lasting antimicrobial activity after repeated exposure to Gram-positive and Gram-negative bacteria, still having antibacterial effects against staphylococcus aureus after 51 days of storage. And the inhibition effect was maintained after 67 h. Their study found a strong interaction between POM and CS, enabling the in situ formation of hydrogels via a thermal response. The adhesion of CSderivatives allows the hydrogel to be used as an antibacterial coating in the desired local environment to combat the growth of a wide range of bacteria, including Staphylococcus aureus, Escherichia coli, and methicillin-resistant Staphylococcus aureus (MRSA). Organic synthetic adhesive materials can be cross-linked by dynamic bonds, showing sufficient mechanical strength and flexibility that can accommodate wound movement at the same time exhibiting an antibacterial effect, thus accelerating wound healing. Pan et al. proposed a novel supramolecular gelatin (GT) hydrogel comprised of a GT-grafted aniline tetramer and quaternary-ammonium CS. The hydrogel was cross-linked using monoaldehyde b-cyclodextrin via host–guest interactions and a dynamic Schiff base. Due to its dynamic properties, the hydrogel exhibits flexibility, self-healing, and tissue adhesion, and is well suited to athletic wounds. The hydrogel has shown antibacterial, antioxidant, and hemostatic properties in sports wound healing experiments with full-thickness Staphylococcus aureus (MRSA) infection and has great potential for wound healing. Inspired by mussels and brown algae, many researchers have introduced catechol groups into hydrogels using organic synthesis reactions to endow them with excellent adhesive properties. Guo et al. prepared a bionic bio-adhesive with antibacterial effects and sufficient adhesive strength via dynamic cross-linking. They cross-linked the hydrogel via Schiff base and catechol–Fe coordination bonds. It also exhibits a temperature-dependent phase transition between GT and SA, which strongly interacts with the resulting hydrogel with sufficient mechanical and adhesive strength. The coordination interaction between ferric and protocatechuic aldehyde gives the hydrogel temperature-responsive mechanical properties, temperature-therapeutic efficacy, and temperature-triggered antibacterial ability, which may participate in the photo-thermal antibacterial process. Lee et al. used trimethylolpropane triacrylate (TMPTA) and gentamicin (GEN) as branching and blocking agents to carry out Michael addition reactions with PEG diacrylate (PEGDA) and dopamine (DOPA), and a mussel-biomimetic hyperbranched polymer coating (TXPDG) was carefully designed and synthesized. By adjusting the molar ratio of the reactants, a series of TXPDG coatings with controllable branching degree (BD) and GEN content were successfully prepared and applied to the interface modification of various substrates using a convenient immersion process. The general adhesion of 1,2dihydroxybenzene to the molecular structures occurs, and the T20PDGcoating s formed firmly on the substrate surface due to the presence of the GEN terminal groups, which exhibits excellent antibacterial activity against both E. coli and Grampositive Staphylococcus aureus. In addition, evaluation of the BSA protein adsorption confirmed that the TXPDG polymer coating also exhibited significant antifouling properties, rendering the implant surface heterogeneous and prevented severe stimulus responses. Xie et al. also prepared a poly(vinyl alcohol)/poly(acrylamideco-[2-(methacryloyloxy)]dimethyl-(3-sulfo-propyl)ammonium hydroxide) (PVA/P(AM-co-SBMA)PPS) hydrogel via UV crosslinking and freeze-thaw cycles for antibacterial use in flexible bio-electronic skin. The PPS hydrogel not only has good mechanical properties, but also a wide range of sensory sensitivity, high adhesion with a variety of substrates, and antibacterial properties. The antibacterial properties of the hydrogel can effectively prevent allergic symptoms of the skin in direct contact with an electronic skin device, prolong the service life of the device, and reduce the replacement frequency of the device. The high adhesion makes the hydrogel exhibit satisfactory adhesion under sweat conditions. Self-adhesive antibacterial self-powered strain sensors made from PPS hydrogels are expected to play an important role in flexible bioelectronics. When compared to synthetic materials, adhesive materials composed of natural polymers have higher biocompatibility and degradability, and composite adhesive materials can overcome the limitations of a single adhesive material, which gives these materials higher application prospects in antibacterial applications. Mogel et al. developed a hydrophobic nanocarrier for use against planktonic bacteria that has been reported to increase the efficiency of the hydrophobic antibacterial agent triclosan by a factor of 1000. Poly(N-isopropylacrylamide-co-N-[3-(dimethylamino)propyl] methacrylamide), P(NIMAM-co-DMAPMA)-based nanogels were prepared via a one-pot precipitation polymerization and quaternized with 1-bromododecane, and hydrophobic domains were formed within the nanogel network via self-assembly within the particles of the aliphatic chain (C12). Triclosan, as a model hydrophobic antibacterial agent, was loaded into the hydrophobic region of the nanogel. Nanogels adhere to bacterial cell walls through electrostatic interactions and induce membrane destruction by inserting fatty chains into the cell membrane. The hydrophobic antibacterial agent triclosan can be actively injected into cells through damaged cell membranes. This method significantly increased the effective concentration of triclosan at the bacterial site. When compared with free triclosan, Staphylococcus aureus and Staphylococcus epidermidis were reduced by three orders of magnitude. Jiang et al. reported an injectable multifunctional hydrogel that was synthesized via the incorporation of spindle-shaped zinc oxide nanorods into carboxymethyl CS (CMCS-brZNO) (Fig. 8). Hydrogels have the advantages of controllable gel time, rapid hemostasis, and good tissue adhesion. In addition, the CMCS-brZnO hydrogel exhibited low minimum inhibitory concentrations (MIC) against Escherichia coli (0.0125 mg mL 1) and Staphylococcus aureus (0.025 mg mL 1), and excellent antibacterial activity. In a full-thickness skin defect experiment, the hydrogel can be directly injected into the wound with an irregular shape and exhibited excellent adhesion to the skin model; the good fitting of the gel to the wound geometry as well as the slow and sustained release of antibacterial Zn2+ significantly promote wound healing and reduce the inflammatory response. These attractive properties give the CMCS-brZnO hydrogel great potential for clinical applications.
Fig. 8 A schematic representation of an injectable CMCS-brZnO hydrogel wound dressing with excellent antibacterial activity, self-healing, and adhesion. Reproduced from ref. 93 with permission from Elsevier. Yao et al. have prepared a new type of ionic conductive multifunctional polyacrylamide (PAAm) CS tannic acid (TA) hydrogel (PCT Hydrogel) using a polyacrylamide chain as the backbone. CsandTAbestowedantibacterial activity (497%) and UV-blocking ability (496%) to the hydrogel, respectively. Simultaneously, the adhesion of the hydrogel allows it to be possibly used as a conductive material attached to the skin. This strategy cleverly utilizes the adhesion of the adhesive material combined with its own antibacterial activity to provide an ideal choice for the production of health monitoring products. Using a nucleophilic substitution reaction between postfibrotic lysozyme (LZMF) and the n-hydroxysuccinimide terminal groups on a four-arm PEG-NHS, Qu et al. designed and synthesized a PEG cross-linked LZMF composite antibacterial hydrogel. The PEG-LZMF hydrogel exhibited antibacterial activity both in vitro and in vivo. The relatively fast cross-linking reaction makes PEG-LZMF injectable and shape-compatible. The simultaneous reaction with tissue-exposed-NH2 or-SH also allows tissue adhesion to occur. In addition, the research group hypothesized that this hydrophobic lysozyme-amyloid fibrillaryintegrated PEG composite hydrogel can efficiently adhere/protect open wounds and internal incisions via a bionic antibacterial mechanism to inhibit pathogen infections. Huang et al. reported tissue adhesive properties by introducing mussel-inspired catechol groups into a hydrogel, which allow the resulting hydrogel to adhere well to the skin underwater and have good shape adaptability under bending and twisting conditions. The mechanical and adhesive properties of the hydrogel were improved by introducing borate/diol interactions into the catechol-modified hydrogel with dynamic Schiff base crosslinking. In addition, they introduced PDA NPs into the hydrogel via a Schiff base reaction between the primary amines in ethylene glycol CS (GC) and quinone groups in the PDA NPs, resulting in potent photothermal antibacterial activity. The PDA NPs were uniformly dispersed in the hydrogel, which showed good cell compatibility and blood compatibility (Fig. 9).
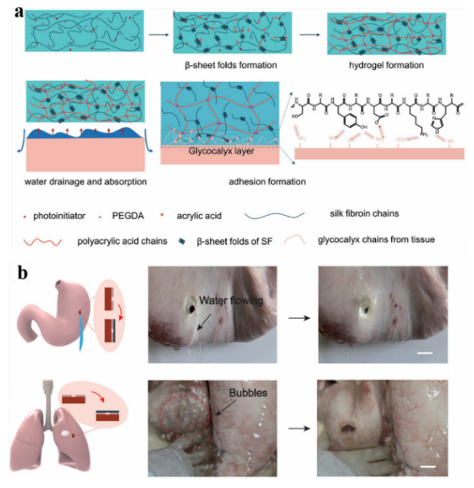
Fig. 9 Tissue adhesion properties of hydrogels. Reproduced from ref. 32 with permission from American Chemical Society. Lee et al. incorporated adhesive catechins into microgels to mimic the strong adhesion of mussel adhesion proteins, which enabled the resulting microgels to bind to various surfaces underwater. Hydrogen peroxide (H2O2) is produced as a byproduct during the autooxidation ofcatechol.Thus,themicrogels can generate H2O2 at millimolar levels upon simply hydrating the microgels in solution at physiological pH. This method has both antibacterial and antiviral effects, and can even inactivate nonenveloped viruses that are more resistant to biocides. These microgels can be produced by repeatedly activating and deactivating H2O2 upon incubating them in solutions with different pH values. This simplicity and recyclability enable the biomaterial to serve as a portable source of disinfectant. Jane et al. developed a new antibacterial family by introducing the antifungal agent 10-undecarbonic acid (UA) into an injectable citrate-based mussel bioadhesive (iCMBAs). The abundant free carboxyl groups from citrate in the polymer may reduce the local pH, inhibit the oxidation of nicotinamide adenine dinucleotide (NADH), and chelate metal ions in the cell wall, which changes the permeability of the cell wall toward the absorption of nutrients, leading to cell damage and subsequent cell death. Therefore, citrate-based polymers exhibit inherent antibacterial properties. By modifying citrate-based polymers with dopamine, the resulting antibacterial agents exhibit strong wet-tissue adhesion. In addition to catechin groups, which are often used, Schneider et al. first identified a peptide derived from mussel foot protein 5, a key protein in mussel adhesion, with antibacterial properties. Based on these properties, they designed a novel peptide-based antibacterial adhesive hydrogel prepared via selfassembly. The hydrogel was active against drug-resistant Grampositive bacteria. The gel exerts two mechanisms of action: (1) destruction of the surface contact film and (2) oxidative killing affected by the H2O2 produced by the materials. By studying the relationship between the amino acid composition and sequence, and the mechanical adhesion, cohesion, and antibacterial activity of the material, they proposed MIKA2 adhesive gel as a material with excellent activity, which has been shown to inhibit the colonization of titanium implants in mice. 3.3 Tissure repair Tissue repair aims to develop biological substitutes for the repair or improvement of defective tissues, organ structures, etc. Therefore, the materials used are required to be highly biocompatible and non-immunogenic, and when used in the body, also require the material to be effective in a wet environment. Due to their good mechanical properties, biocompatibility, and tissue affinity, adhesive materials can play an important role in various aspects of tissue repair. Desirable biological functions can be achieved by combining and utilizing different adhesive materials. CS can promote Schwann cell growth, pelvic floor tissue repair,180 and 3D tissue growth, and is often used to make skin tissue regeneration, antibacterial, and anti-oxidation multi-functional hydrogels. CS-Based multifunctional hydrogels can achieve rapid in situ gelation at the wound site and completely cover irregular wounds, induce skin tissue remodeling, promote vascular repair and hair follicle regeneration, and accelerate wound healing. Wu et al. explored the feasibility of nerve tissue repair using CS composite scaffolds. It was found that a HA-modified CS composite scaffold can improve the adhesion of neurons in vitro and in vivo, and the expression of inflammatory cells and glial fibers was superior to that of PLL-modified CS composite scaffolds and pure CS composite scaffolds. Hamodified CS composite scaffolds have great research and development prospects in nerve tissue repair. Proteins such as gelatin and collagen are used not only for chondrocyte growth and cartilage regeneration, but also promoting cell adhesion to the surface of basic matrix brush-like polymers. Chen et al. developed a nano-micron double polymer brush system consisting of a gelatin-modified poly-LRB-glycidyl methacrylic acid (gelatin-PGMA) brush and p-LRB-ly (n-isopropylacryl) PNIPAAM(PNIPAAm) brush, which provides adequate cell adhesion and allows intelligent isolation of externally stimulated cells for tissue repair. The gelatin-PGMA brush was responsible for the orientation of mouse embryonic fibroblasts (NIH 3T3), whereas the PNIPAAm brush was responsible for cell adhesion. Brush polymers are widely used in tissue repair and have a great influence on cell culture or the production of cell layers, preventing cell or microbe adhesion (antifouling effect). Li et al. developed a bioadhesive hydrogel (GMO) used for conjunctival transplantation based on a semi-interpenetrating polymer network (SIPN) comprised of methacrylate gelatin (Gelma) and oxidized hyaluronic acid (OHA). Gelma was used as a rigid support and OHA was used as the interface initiator. The obtained hydrogels can provide strong adhesion between collagen films; the maximum adhesion strength was 157 17 kPa and the bursting pressure was 357 29 kPa, which is 15 times higher than the human intraocular pressure (IOP). With the help of a genetically modified organism adhesive, collagen scaffolds were seamlessly transplanted into the conjunctiva defect in the body. The seamless transplantation method can promote the repair of scar-free tissue and significantly improve the operation efficiency. It is a promising alternative method for ocular surface reconstruction, which can avoid suture-related complications and improve clinical efficacy. Zheng et al. developed a decellularized tissue (DT) hydrogel equipped with oxidative cross-linking chemistry by combining the catechol moieties with the extracellular matrix in DT, and the catechol-modified DT (DT–CA) was oxidized by a catecholcatechol adduct to construct the hydrogel. When compared to the unmodified DT hydrogel, its mechanical properties were 10 times stronger. This method can improve the machinability and long-term stability of the hydrogel structure, and the modified hydrogel shows enhanced mechanical and adhesive properties. In a mouse model with a critical-sized skull defect, the DT–CA hydrogel showed a strong ability to implant, promote osteogenic differentiation of human stem cells, and accelerate new bone formation. In addition, to specifically promote local wound healing, the group also developed a patch-type DT–CA that promotes wound healing by mediating the efficient local delivery of growth factors. Adhesives that can bind strongly to biological tissues have a broad range of application prospects. In general, strong binding with biological tissue adhesives has a certain degree of biological toxicity. However, Li et al. designed a strong tissue adhesive with a two-layer matrix that was less biologically toxic inspired by the defensive mucus secreted by Mollusca slugs. Theadhesivewas a bionic design comprised of two layers of matrix: adhesive surface and dissipation matrix. The former was attached to the substrate via electrostatic interactions, covalent bonds, and physical interpenetration, and the latter by the hysteresis amplification of energy dissipation. The interaction of the two substrates further enhanced the adhesive ability to adhere to wet surfaces, resulting in a tight bond with pig skin, cartilage, heart, arteries, and liver. It has been reported that this adhesive has successfully achieved the rapid repair of perforated heart tissue in pigs and liver tissue in rats. Therefore, this bionic approach effectively reconciles the issues of tissue adhesion and biotoxicity. 3.3.1 Hemostasis. By adding silk fibroin to a polyacrylic acid network, Yu et al. developed an adhesive hydrogel with excellent bulk strength and interface adhesion. Animal studies showed that the obtained silk fibroin-based hydrogel PSA-2 can stably adhere to rat skin and had a significant effect on the healing of full-thickness skin wounds (Fig. 10). PSA-2 also showed good wet surface adhesion and hemostatic effects in rat models of hepatic and abdominal aortic hemorrhage. During the treatment of traumatic pneumothorax in rabbits, PSA-2 can achieve rapid hemostasis and adhesion, and form a stable adhesion interface. After half a month, the rabbit lung recovered well and there was no significant difference between the rabbit lung and normal tissue. These experimental results showed that the adhesive hydrogel prepared by introducing silk fibroin protein has good application prospects in tissue repair.
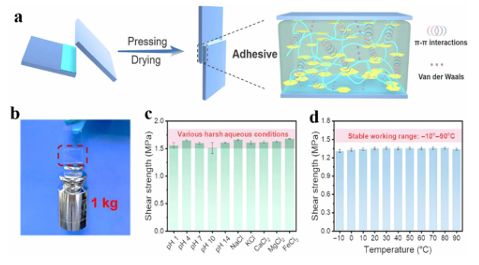
Fig. 10 (a) The preparation and adhesion mechanism of PSA. (b) A schematic representation of sealing a gastric perforation (left) and corresponding ex vivo sealing process by PSA-2. Scale bar: 3 cm.40 Reproduced from ref. 40 with permission from John Wiley & Sons. Aiming at tissue repair in moist environments, Zhao et al. prepared a gallic acid-modified CS-based hydrogel using glow discharge plasma, which was inspired by mussel adhesion proteins. The CS-GA hydrogel with good toughness and adhesive properties was formed via a self-crosslinking reaction in air. They evaluated the ability of the hydrogel to stop bleeding and promote wound healing in in vivo rat models of hepatic hemorrhage and full-thickness skin defects, and found that the properties of the hydrogel were comparable to those of clinically used wound dressings. Cytotoxicity and hemolysis studies also showed that the hydrogel had good blood compatibility and biocompatibility, effectively avoiding the risk of hemolysis caused by contact of the hydrogel with the tissue. Hwang et al. also developed a CS-based wet tissue adhesive with a hemostatic function from the self-repair mechanism of the shell envelope of the sea squirt. By introducing pyrogallol into the CS backbone, the resulting CS-based adhesive exhibited better wet adhesion than commercially available tissue sealant-fibrin glue. At the same time, the combination of CS and primary amine with pyrogallol increased the solubility at physiological pH and improved platelet adhesion and hemostatic function of the adhesive. Due to its rich and easy access to raw materials and low cost, it is a practical and versatile approach with great potential in the biomedical field. 3.4 Others In addition to the above applications in wound dressings, hydrogels made from adhesive materials have attracted considerable attention due to their many compelling properties in favor of wound repair, including the absorption of exudates and maintenance of a moist environment at the wound site. When applied to traumatic anastomoses or surgical incisions, they have excellent adhesion, filling damaged areas to stop bleeding and prevent fluid leakage. Hydrogels formed in situ can be designed to accumulate serum fibrin exudates to form fibrin bridges, where fibroblasts can migrate and secrete collagen to heal injured tissue. Among them, horseradish peroxidase (HRP)-mediated reactions can rapidly and controllably form gels under mild experimental conditions. Using this approach, Pak et al. successfully prepared CS-poly(ethylene glycol)-tyramine (CPT) hydrogels as efficient tissue adhesives. Adhesive materials are widely used as pressure-sensitive adhesives. Acrylate pressure-sensitive adhesives are the most widely used pressure-sensitive adhesives available on the market. They are copolymers comprised of acrylate monomers and other vinyl monomers with no need to add anti-aging agents, good adhesive strength, aging resistance, weather resistance, heat resistance, good transparency, resistance to media, phase-free separation and migration, and other good properties. Polyurethane pressure-sensitive adhesives are characterized by low toxicity and good biocompatibility, and their theoretical research and practical applications have rapidly developed. Polyurethane pressure-sensitive adhesive is a linear multiblock copolymer comprised of alternating glass state or crystalline state hard segments and a highly elastic state soft segment. The soft segments are usually polyethers, polyesters, or polyolefins. The hard segments are usually composed of isocyanate and chain extenders. The adhesive obtained upon solution polymerization is in a state of viscous flow, adding different curing agents. After high-temperature curing, a viscoelastic transition state and good adhesion can be obtained. Excellent adhesive performance is required in the field of intelligent devices. Hydrogels are materials that contain a lot of water, which can provide a transport pathway for conductive ions, and are promising candidates for the manufacture of artificial skin and strain sensors. Most elastomers, which consist of semiconductors and metals, detach from each other under large strain, resulting in the breakdown of the conductive network and loss of conductive stability and sensing properties. Hydrogels with excellent adhesion properties ensure that the electrical signals are converted to the maximum extent possible during application. Yu et al. synthesized a novel double-network hydrogel system based on PVA and the oxidation of polyethylenimine. Based on the properties of the polyethylenimine clusters, the study was carried out by introducing a simple supramolecular crosslinking agent, and a novel in situ oxidation method was developed to fabricate the first double network hydrogel interface sensor based on polymer nanoclusters. The hydrogel has good transparency (490%, 500–800 nm) and high electrical conductivity (1.3 s m1), which can be self-repaired and stretched rapidly in 1 s. The repair efficiency and tensile growth rate of the hydrogel were as high as 98% and7000%, respectively after 60 s. The response time and recovery time to pressure were both o120 ms. It was found that these excellent properties were due to four types of dynamically reversible interactions formed in the double network hydrogels: borate covalent bonds, hydrogen bonds, positive and negative charge interactions, and dipole–dipole interactions between heteroatoms in the polymer nanoclusters. These interactions also allow the hydrogel to reversibly adhere to the skin, wood, and plastic surfaces without causing allergic damage to the skin. Cytotoxicity studies showed that the hydrogel matrix can greatly promote the proliferation of mouse fibroblasts, reaching 682% within 7 day, confirming that the hydrogel had good biocompatibility. Simultaneously, the formation of polymer clusters can improve the conductivity and self-repair ability of the aggregates. As an application, the hydrogel can be used for online, real-time monitoring of large body movements (such as knee, finger, and wrist bends), minor movements (such as speaking, smiling, frowning, etc.), and physiological activities (such as heartbeat and pulse), showing a stable and fast electrical signal output. Zhao et al. prepared a fully recyclable flame-retardant multifunctional adhesive using the polycondensation of phosphorous aryl siloxane derivatives (DPOP-Si). Theadhesivecanbereversibly self-assembled on the substrate surface in an orderly manner driven by p–p stacking. Due to the strong cohesive energy generated by the water-insensitive p–p interactions, the resulting adhesive exhibits a strong and durable adhesive force on various materials. The shear adhesion strength on the surface of a glass substrate was as high as 3.5 MPa and also exhibited good adhesion to steel, wood, and other substrates. Even under harsh conditions in salt/acid/base solutions (pH 1–14) and over a wide temperature range ( 10 to 90 1C), the effect of firm adhesion can be maintained without attenuation for long periods of time (Fig. 11). In addition, the adhesive has high transparency and excellent flame retardancy, which can be completely recovered and reused in response to stimulation with ethanol. The flame retardancy of adhesives is particularly important in many applications. When applied to electronic equipment, a high flame retardancy is often required to reduce the potential fire risk. Therefore, this work will significantly inspire the next generation of versatile and fully recyclable sustainable adhesives.
Fig. 11 The lasting adhesion in various harsh environments according to the adhesion characteristics of different substrates. (a) P(DPOP-Si) adhesion with p–p and van der Waals interactions. (b) Photograph of the glass surface P(DPOP-Si) adhesive. (c) Adhesive stability of P(DPOP-Si) adhesive after 7 d of immersion in various solutions at pH 1–14 and different salt solutions (NaCl, KCl, CaCl2, MgCl2, and FeCl3). (d) The relationship between the shear bond strength of the adhesive and working temperature.43 Reproduced from ref. 43 with permission from American Chemical Society. In order to solve the problems associated with the high strength adhesion of bionic dry adhesion structures on rough surfaces, Tian et al. abandoned the traditional manufacturing idea of "machining" and focused on the growth behavior of organisms evolving from simple structures to complex tissues and organs through cell proliferation and differentiation, cleverly utilizing the electric field law on polymers, and a new forming strategy of the "self-growth" of adhesive structures was proposed. They designed a mushroom-like soft shell/hard core bionic structure and proposed a "from nothing to something" strategy for the electrogenerated autonomous growth of a core–shell structure, and the self-growing core–shell biomimetic adhesion structure showed excellent adhesion characteristics on the surface with roughness on the nanometer, micron or even hundred micron scale. This research work breaks through the application bottleneck of bionic dry adhesion structures in their application on the rough target surface and the durability of the structure, and helps to promote the development of bionic dry adhesion-related structures, devices, and systems. Adhesive materials were introduced into the field and reversible adhesive hydrogels suitable for sampling were designed and synthesized. Based on ion mobility spectrometry, mass spectrometry, infrared spectroscopy, and other trace detection methods, the sampling efficiency of a sample material can directly determine the success or failure of detection. However, the sampling efficiency is o1% because of the weak adhesion and small contact area of the sampled materials. Reversible adhesive hydrogels have a basic three-dimensional skeleton formed by the polymerization of two polymer chains with completely opposite physical characteristics. Polyacrylic polyacrylic acid (PAA) chemically covalently bonded by N,N0methylenebisacrylamide (MBAA) was crosslinked to PVA in a non-covalent polyvinyl alcohol, and borax was introduced to form a dynamic covalent bond with PVA to further improve the cohesion of the hydrogel. PAA, PVA, and MBAA are rich in carboxylic acid, hydroxyl, and amino groups, which ensure the adhesive force of the hydrogel. This type of hydrogel not only has good reversible adhesion, but also has a large contact area with particles due to its wet surface and universal attraction. It can also realize the integration of the sampling and detection of suspicious scenes, which provides a new idea for the development of the field of sampling materials. 4. Conclusion and perspective In summary, this review introduces several types of adhesives that are the most studied and widely used, such as natural polyphenol adhesives and organic synthetic adhesive materials, as well as the applications of composite adhesive materials and underwater adhesive materials in drug delivery, antibacterial, tissue repair, and other fields. Because of their unique biocompatibility, biodegradability, and tissue adhesion, they have great advantages as new medical adhesive materials and will gradually replace traditional adhesive materials in the field of biomedicine, and have been the subject of a wide range of exploration and research studies. However, existing adhesive materials have some limitations in biomedical applications. For example, there is often a conflict between the biocompatibility and mechanical strength of natural adhesives. Natural adhesives often lack sufficient mechanical strength, which limits their practical applications, and when sufficient mechanical strength is achieved, natural adhesives are prone tohavean impact ontheorganismitself. Therefore, some researchers have used composite low-toxicity adhesive materials to improve the mechanical properties of adhesive materials, while minimizing the damage caused by the adhesive itself. However, there is still a need for more in-depth exploration to improve the adhesive properties, such as the discovery of better mechanical properties of the biocompatible elements. Adhesive materials in wet environments have been a hot research topic in recent years. However, adhesion in wet environments has been a major challenge for the application of biomedical materials. It is necessary to simulate different environments to study the application effect of adhesives because the change in the pHandinterfacial interactions between different materials are obstacles to the application of adhesive materials. In addition, existing adhesive materials have many single functions, such as the development of anti-bacterial adhesives that can only be used for this purpose, but the same materials can also be processed for drug release. Apart from their simple mechanical properties, intelligent adhesives have not been extensively studied for specific uses. Now, many researchers are aware of this problem and the development of new adhesive materials will take into account the temperaturesensitivity, pH response, electrical conductivity, and so on to develop multi-functional hydrogels. However, it is worth noting that the development of multi-functional adhesive materials often involves a more complex preparation process, and the cost is higher. Therefore, researchers need to explore more convenient methods to synthesis these materials. Finally, it can be seen that research on adhesive materials is continuously developing and more and more aspects such as the recyclability and moisture retention of materials are being considered. Currently, PVA hydrogels and CS products have been successfully applied in clinical practice with good results being achieved. With the continuous development of science and technology, nanomaterials can be used in more subtle places and injectable adhesive materials can bring greater convenience to the field of medicine, I believe that adhesive materials will be more widely used and play an irreplaceable role in more applications. Author Contributions Themanuscriptwaswrittenthroughcontributionsofallauthors. All authors have given approval to the final version of the manuscript. Source: rsc.li/materials-b |