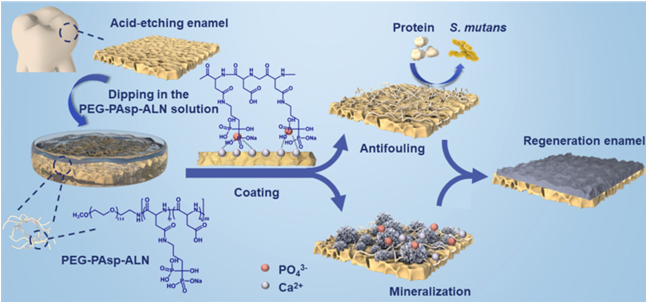
A multifunctional Polymeric Coating with Self Adsorbed, Antifouling and In Situ Remineralization Properties for Caries ManagementIssuing time:2025-04-24 09:45 Wenlin Chu, Haiqin Tang, hiyun Dong, Ailin Hou, Rongmin Qiu, Xinyuan Xu,b Jiaojiao Yang, aLibang He, Jun Luo, Jianshu Lee Dental caries is a biofilm-induced bacterial infectious oral disease, where the early attachment of proteins and pathogenic bacteria to tooth surfaces has been known as the main cause of biofilm formation. Typically, dental caries is commonly accompanied by mineral depletion of enamels, thus causing dental demineralization. Multifunctional materials are highly attractive candidates for treating dental caries. Herein, we successfully synthesized diblock copolymers poly(ethylene glycol)-bpoly(aspartic acid) (PEG-PAsp) and modified them with alendronate sodium (ALN) to serve as bioactive bifunctional coatings (PEG-PAsp-ALN) on teeth. The PEG segments are employed for inhibiting proteins and bacterial adhesion. In addition, due to the presence of both PAsp and ALN, a synergistically strong binding capacity could be achieved with the tooth surface, thus promoting rapid and thorough remineralization in situ, while maintaining excellent safety. The combination treatment can significantly suppress the biofilm formation, which is beneficial for alleviating the demineralization of enamels caused by bacteria, and further, facilitate remineralization in situ. This approach thus demonstrates the potential of the copolymer PEG-PAsp-ALN coating as a multifunctional protecting layer on the tooth surface for high-efficiency prevention and treatment of dental caries. Introduction As one of the three key human diseases presented by the WHO, dental caries has not been a neglected topic for its general incidence in most countries where a vast majority of children and adults are suffering from different degrees of pain brought about by it. The initial extension of the caries lesion hardly leads to pain and discomfort in patients. However, if allowed to develop, the lesion spreading to the dental pulp can induce toothache and serious systemic complications such as local infection and very rarely, tooth loss. Actually, dental caries is a biofilm-mediated multifactorial dynamic disease. The biofilm in the oral cavity, i.e., dental plaque, is the main reason to induce demineralization of enamel and then causes caries.4 In nature, oral proteins and peptides adhere to the dental surface to form a saliva acquired pellicle (SAP) first and then bacteria adhere to the SAP to realize subsequent bacterial biological functions. The formation of dental plaque depends on the successful survival and colonization of opportunistic pathogens on the surface of enamel, and then these pathogens have the ability to develop into biofilms. The bacteria in the biofilms proceed with anaerobic metabolism owing to the absence of oxygen, resulting in producing amounts of lactic acid difficult to be cleared. The local sustaining effect of lactic acid makes pH drop below the critical point, at which the biointerface between the enamel and the biofilm becomes unsaturated and then the surface of enamel demineralizes. Consequently, the prevention and treatment aiming at dental caries focus on two strategies, including inhibition of biofilm formation and remineralization of the defective enamel surface. In recent years, a series of emerging materials bearing different compounds, such as metal ions or nanoparticles, phenols and ammonium compounds, have already been investigated to efficiently kill bacteria to prevent the formation of a biofilm. Despite they have been found to exhibit good antibacterial properties, most of these materials have not widely been accepted due to their intrinsic problems, such as considerable cytotoxicity. Alternative strategy to resist oral biofilm formation is reducing bacterial and protein adhesion in the first place. A number of antibiofouling materials have been developed to suit the urgent need for anti-protein and-bacterial adhesion to dental materials. Among, polyethylene glycol (PEG) is well-known as the gold standard for surface modification to reduce protein and cell adhesion. For instance, it was shownthat grafting PEG coating on dental materials can reduce the adhesion of Streptococcus mutans (S. mutans), the most notably leading acidogenic oral bacteria. However, pure PEG coatings cannot regulate the dynamic equilibrium of the demineralization and remineralization of dental hard tissues. The research to explore the mechanism of biomineralization led to the emergence of a series of remineralized materials such as casein phosphopeptide (CPP), poly(amidoamine) dendrimers and polyacrylic acid (PAA), etc. The presence of carboxyl groups and/or PO4 3 ions in most of these materials made them work by binding with enamels and serving as nucleation sites to facilitate mineral formation. Aspartic acid (Asp) is abundant in non-collagenous proteins (NCPs), which can regulate the amorphous calcium phosphate (ACP) phase transition process and promote mineralization. However, noncovalent interactions may cause unsatisfactory stability of materials during application and short efficacy. Therefore, we have previously designed a system focusing on the synergistic effect of antifouling and remineralization by synthesizing a block polymer PEG-PAsp. The existence of PEG segments endowed the materials with anti-fouling ability and the carboxyl groups of PAsp segments can chelate with Ca2+ ions to induce remineralization. However, the weak binding force between Ca2+ ions and carboxyl groups leads to the low absorption of polymers on the tooth surfaces, thus decreasing the deposition of Ca2+ and PO4 3 ions, which ultimately limits the effect of remineralization. To overcome this drawback, we have proposed a general strategy for a multifunctional coating to realize antifouling performance and highly effective remineralization by polymer postfunctionalization. (Fig. 1). PEG-PAsp was synthesized by ring-opening polymerization to provide antifouling and mineralizing properties. More importantly, as a typical diphosphonate with a high affinity for bone minerals, alendronate sodium (ALN) is chosen to be the bioactive molecule to provide plentiful reactive PO4 3 groups by conjugation onto the polymer for optimizing the stability of materials to enamels and mineralinducing capability. The postfunctionalization coating was expected to possess high antifouling efficiency against proteins and S. mutans as well as enhanced remineralization capacity. The present work provides a promising multifunctional coating to solve the clinical problems of tooth decay.
Fig. 1 Schematic illustration of PEG-PAsp-ALN block polymers to mediate enamel remineralization and resist adhesion of bacteria and proteins. Experimental Materials a-Methoxy-o-amino poly (ethylene glycol) (MeO-PEG45-NH2, Mn =2kDa), b-benzyl-L-aspartate-N-carboxyanhydride (BLA-NCA), alendronate sodium (ALN), 1-ethyl-3-(3-dimethylaminopropyl) carbodiimide (EDC), and N-hydroxysuccinimide (NHS) were purchased from Shanghai Macklin Biochemical Co., Ltd (Shanghai, China). N,N-Dimetylformamide (DMF), chloroform (CHCl3), phosphoric acid, diethyl ether and dimethyl sulfoxide (DMSO) were purchased from Chengdu Chron Chemical Co., Ltd (Chengdu, China) and used without further purification. Phosphate Buffer Saline (PBS), a-Modified Eagle’s Medium (a-MEM), Fetal Bovine Serum (FBS) and Penicillin–Streptomycin solution were all purchased from Bosco Biotechnology Co., Ltd (Chengdu, China). Dialysis tubing (Mw = 3500), fluorescein isothiocyanate (FITC), propidium iodide (PI), rhodaminelabeled phalloidin, fluorescein diacetate (FDA) and 4',6-diamidino-2-phenylindole (DAPI) were purchased from Beijing Solarbio Science & Technology Co., Ltd (Beijing, China). Bovine Serum Albumin (BSA) was purchased from Biofroxx (Germany). Lysozyme (Lys) and Sodium laurylsulfonate (SDS) were purchased from Yuanye Bio-Technology Co., Ltd (Shanghai, China). Brain Heart Extract Broth (BHI) and the Live/dead BacLight Bacterial viability kit were purchased from Thermo Fisher Scientific (USA). Synthesis and characterization of PEG-PAsp-ALN According to the previous literature, PEG-PAsp was synthesized by the ring-opening polymerization (ROP) of BLA-NCA with CH3-PEG-NH2 as the initiator. In brief, BLA-NCA and CH3PEG-NH2 were dissolved in DMF and CHCl3, respectively, and then poured into the reaction flask without water and oxygen. The reaction solution was stirred for 3 days at 35 ℃ in a pure nitrogen atmosphere to react completely. After that, the resulting product PEG-PBLA was purified by dropping the solution into cold diethyl ether to form precipitates and collecting them by centrifugation and subsequently drying in a vacuum. To further access PEG-PAsp, PEG-PBLA needs to be deprotected to remove the benzyl group. First, PEG-PBLA was stirred to dissolve in CHCl3 and subsequently reacted with the NaOH solution which was prepared with a mixed solvent of H2O, methanol, and isopropanol at a volume ratio of 1:2:2. The whole mixing reaction was stirred at 0 ℃ for 30 min. Later, the pHofthe solution was adjusted with glacial acetic acid to 7 and organic solvents were removed by rotary evaporation. The concentrated solution was purified with a dialysis tube for 3 days and lyophilized to get pure PEG-PAsp. As for PEG-Pasp-ALN, it was synthesized by partial modification of PEG-Pasp with alendronate sodium. PEG-Pasp, EDC and NHS were dispersed successively in a mixed solvent of water and DMF at a volume ratio of 3:2 at 4 ℃ for carboxylic group activation, followed by the addition of the alendronate sodium solution. Then the system was stirred for 12 h at room temperature, dialyzed for 3 d and lyophilized to obtain PEGPasp-ALN. The 1H NMRspectra of PEG-PBLA, PEG-Pasp and PEG-PaspALN were recorded using an NMR Spectrometer (AV III HD 400 MHz,Bruker) at room temperature with DMSO-d6,D2O and D2O as solvents, respectively. The spectra of these block copolymers were recorded with an ATR-FTIR system (Nicolet 6700, Thermo Scientific TM) Preparation of tooth enamel slices The periodontal membrane and dental calculus were removed after collecting bovine incisors without lesions and cracks. The tooth slices were prepared with a water-cooled diamond saw (DTQ-5). All the enamel slices were embedded in the acrylic resin and polished with 800, 1200 and 2000-grit carbidepolishing papers in turn. After that, the working surfaces of the samples were covered partly by acid-proof nail varnish, leaving 5 x 5 mm2 operation windows for further research. All enamel samples were divided into two groups according to whether they were acid-etched or not. The samples that need to be acid-etched were immersed in 10 mL of 37% phosphoric acid solution for 45 s. After that, these acid-etched samples were washed with deionized water (DIW) for absorption and remineralization assays. While samples of the other group were used for antifouling assays without any treatment. Finally, all samples were cleaned by ultrasound to remove impurities and stored in 0.05% thymol solution at 4 ℃. Absorption assay of PEG-PAsp-ALN on enamel slices A 100 mL solution of PEG-PAsp-ALN and PEG-PAsp at a concentration of 1.4 mg mL 1 were evenly dropped onto the working windows of acid-etched enamel slices, respectively. At the same time, an equal volume of DIW was pipetted to drop on the enamel surfaces to serve as a control group. All enamel samples were dried thoroughly in air, followed by washing with DIW three times to remove unabsorbed polymers and drying again. Three independent absorption assays were performed. The ATR-FTIR spectra of enamels before and after treatment were recorded to demonstrate successful adsorption. PEG-PAsp and PEG-PAsp-ALN were conjugated with FITC to compare the absorption capability of the two polymers. In brief, FITC was dissolved in ethanol, and equal molar sodium hydroxide and EDC were further added. After that, the prepared polymer solution was dropped into the FITC solution, stirred for three days, dialyzed, and lyophilized to obtain FITC-labelled PEG-PAsp and PEG-PAsp-ALN, respectively. A 100 mL solution of labeled PEG-PAsp-ALN and PEG-PAsp at a concentration of 1.4 mg mL 1 was then dropped onto the working windows of acid-etched enamel slices, respectively. Meanwhile, an equal volume of polymeric solutions without FITC was dropped on the enamel surfaces for comparison. All slices were washed thoroughly and dried in a dark place. Fluorescence images of different slices were recorded using a laser scanning confocal microscope (LSCM, FV3000, OLYMPUS, Japan). A BCA Protein Assay Kit (P0012, Beyotime) was used to quantify the absorption of polymeric coatings. Hydroxyapatite (HA) slices (F 8 2 mm,National Engineering Research Center for Biomaterials, Sichuan University) were used to replace enamel slices for better quantification. The HA slices were immersed into a1mL1.4mgmL 1polymersolutionovernight, and unabsorbed polymers were washed off with DIW. After washing, each HA slice was placed in a tube with 1 mL of 2% SDS eluate. After incubation at 37 ℃ for 2 h on a shaker, the solution in each tube was tested with a BCA Protein Assay Kit to analyze the absorbed polymer concentration. Stability of polymeric coatings FITC-labelled PEG-PAsp and PEG-PAsp-ALN were used to analyze the stability of polymeric coatings. The two polymer-coated enamels were immersed in flowing artificial saliva to explore their dynamic stability. The obtained samples were observed under LSCM on days 1 and 3. Water contact angle measurement Following the method mentioned above, three groups of enamel slices were coated with PEG-PAsp-ALN, PEG-PAsp, and DIW, respectively. The water contact angles were measured using a contact angle meter (DSA25, KRUSS, Germany). The enamel slices were placed on the platform and 1 mL of ultrapure distilled water serving as the working fluid was injected into the surfaces for the measurement of the water contact angle. Each sample was measured at three different points, and three parallel samples were set for each group. Anti-protein adhesion assay BSA andLyslabeled with FITC were chosen to evaluate the antiprotein adhesion ability of polymers. The labelling method was carried out according to the procedure previously reported.38 BSA powders were first dissolved in the Na2CO3 buffer with a concentration of 1 mg mL 1 at room temperature (25 1C). The FITC solution of equal concentration was prepared with DMSO as the solvent. Afterwards, the FITC solution was loaded in a syringe and dripped into the BSA solution by gravity along with constant agitation. The volume ratio of the FITC solution to BSA solution is 1:10. The mixing solution was stirred for 24 h at room temperature. After the reaction, the solution was dialyzed with PBS for 2–3 days to remove the unreacted FITC and BSA in the dark place to obtain FITC-labelled BSA. Likewise, FITC-labelled Lys can be successfully obtained with this method. Enamel slices coated with PEG-PAsp-ALN, PEG-PAsp, and DIW were immersed in 2 mL of FITC-labelled BSA and Lys solutions for 24 h at 37 ℃. Subsequent to the protein adsorption period, the slices were washed with DIW 3 times and dried in a dark place. The fluorescence images of different slices were recorded by LSCM. Salivary pellicle adhesion assay Human oral saliva absorption assays with a BCA Protein Assay Kit (P0012, Beyotime) were designed to explore the application potential of PEG-PAsp-ALN coatings in the oral cavity. Oral saliva was obtained from 3 adults aged between 20 and 30 years with no active caries and instructed not to eat or drink anything except water for 2 h before saliva collection. After collection, the mixed saliva was pooled and centrifuged for 20 min (4 ℃, 4000 rpm) to collect the supernatant. HA slices were immersed into 1 mL of 1.4 mg mL 1 polymer solution overnight and further washed off the unabsorbed polymers with DIW. After washing, HA slices were placed in a 48-well plate and 1 mL of saliva was added per well to submerge the slices. The samples were stored for 24 h at 37 ℃ and again washed off to remove unabsorbed saliva proteins. The final HA slices were placed in tubes and 2% SDS eluate (1 mL per tube) was added. Solutions were collected after incubation at 37 ℃ for 2 h on a shaker and the absorbed saliva protein concentration was analyzed using a BCA Protein Assay Kit. Antibacterial adhesion ability S. mutans UA159 were chosen as models to assess the antibacterial adhesion ability of PEG-PAsp-ALN. S. mutans were cultured in BHI at 37 ℃ for 24 h and the bacterial suspension was diluted with BHI to the same concentration which was confirmed using a microplate reader (KHB ST-360, OD600 = 0.5). The HA slices were sterilized before being placed in a 24-well plate containing sterile PEG-PAsp-ALN, PEG-PAsp solutions and DIW respectively. Then the plate was stored for 24 h at 37 1C with shaking to ensure an adequate coating. Coated samples were transformed into a new 24-well plate containing 1 mL of freshly prepared bacterial suspension in each well to incubate again for 24 h at 37 1C. The final samples were rinsed with PBS three times, stained with SYTO 9 at room temperature for 15 min and observed with an LSCM. Remineralization assay of acid-etched enamels in vitro The working surfaces of enamel slices were acid etched in 37% phosphoric acid for 45 s and then washed with PBS (pH = 7.4) and sonicated for 5 min. Subsequently, enamel slices were coated with 100 mL of 1.4 mg mL -1 PEG-PAsp-ALN solutions overnight. The other two groups coated by equivoluminal PEGPAsp solutions with the same concentration and DIW were set as control groups. After washing, all slices were immersed vertically in 10 mL of 1.5 SBF which need to be changed daily at 37 ℃ to induce remineralization. The preparation of 1.5 SBF was based on the previously reported method.39 After 1, 3, 5, and 7 d, the tooth slices of three groups were collected and then rinsed in DIW to remove excess SBF for further characterization. The morphology of the regenerated enamel surfaces and cross-sections was analysed by SEM (Apreo S HiVoc, Thermo Fisher Scientific). Besides, the morphology of the acid-etched enamel without remineralization was also recorded and the crystal structure of the regenerated mineral was examined by XRD (Ultima IV, Rigaku) with Cu-Ka radiation (40 kV, 110 mA) from 20° to 70°. As for the surface micro-hardness of enamel working windows, it was measured with a Vickers hardness tester (MMT-X7A, Matsuzawa) under a 50 gf load for 10 s. Besides, the surface micro-hardness of healthy enamel and acid-etched enamel without any treatment were also measured as a control. For each sample, the original surface micro-hardness (SMH0), acid-etched surface micro-hardness (SMH1), and remineralized surface micro-hardness (SMH2) were all recorded. The degree of surface micro-hardness recovery was computed as (SMH2 SMH1)/(SMH0 SMH1) 100%. Cytocompatibility test Human oral keratinocytes (HOKs) were used to test the cytotoxicity of PEG-PAsp-ALN and PEG-PAsp. The HOK cells were cultured in a modified a-MEM consisting of 10% FBS and 1% penicillin-streptomycin solution. The cells were seeded in 96well plates with a cell density of 104 in an incubator with 5% CO2 at 37 ℃. After incubating for 24 h, the cells in plates were exposed to 100 mL of PEG-PAsp-ALN and PEG-PAsp solutions for another 24 h. Meanwhile, the cells in the medium without polymers served as the control group. After washing cells with PBS, the well plates were filled with 100 mLofa-MEM containing 10%CCK-8 per well and placed for 3 h at 37 ℃ soastomeasure the optical density (OD) value using a microplate reader (KHB ST-360, China) at a wavelength of 450 nm. Besides, live/dead staining was also recorded with a fluorescence microscope. Cells seeded in 24-well plates at a cell density of 105 were incubated for 24 h. After replacing the medium with 100 μL of fresh culture media containing 1.4 mg mL -1 PEG-PAsp-ALNandPEG-PAsp,thecells werecultured for another 24 h. For live/dead staining, the cells were stained with prepared FDA and PI for 15 min. While the observation of the cellular structural integrity the cells need to be fixed with 4% paraformaldehyde and then stained with rhodamine-labeled phalloidin and DAPI. Statistical analysis All experimental data were expressed as mean standard deviation (SD). One-way analyses of variance (ANOVA) were performed to detect the significant effects. Statistical significance was set once p o0.05.
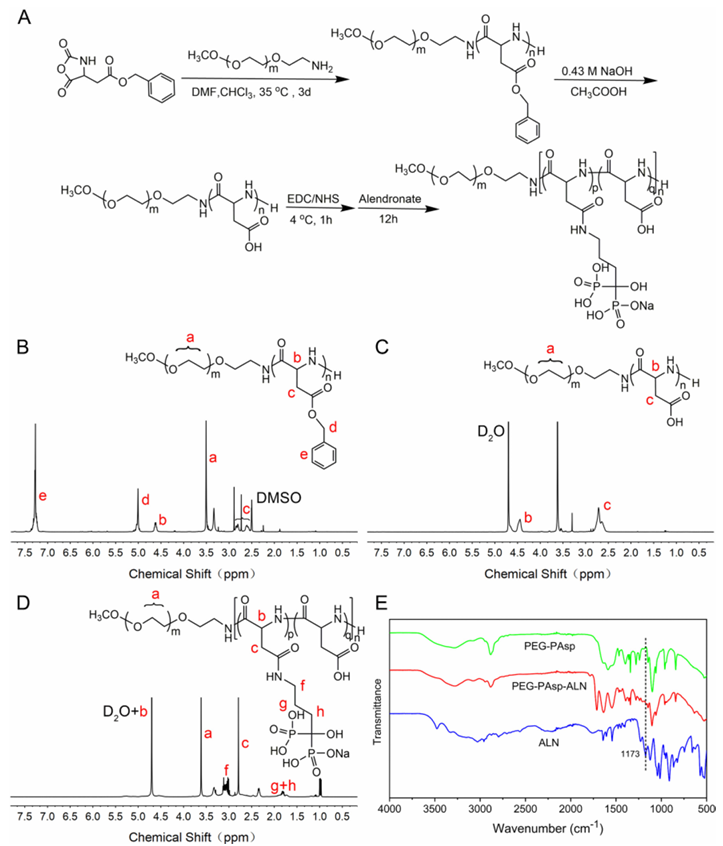
Fig. 2 The synthesis of PEG-PAsp-ALN copolymers. (A) The illustration of the PEG-PAsp-ALN synthetic route. (B–D) The structure and 1H NMR spectrum of PEG-PBLA, PEG-PAsp and PEG-PAsp-ALN. (E) The ATR-FTIR spectra of PEG-PAsp, PEG-PAsp-ALN and the ALN monomer. Results and Discussion Synthesis of the diblock copolymer PEG-PAsp-ALN The synthetic procedure chart of PEG-PAsp-ALN is shown in Fig. 2A. PEG-PAsp-ALN was obtained by sequential ringopening polymerization of BLA-NCA, deprotection of PEGPBLA and modification of PEG-PAsp. The structures of the two synthetic intermediates PEG-PBLA and PEG-PAsp and the final block copolymer PEG-PAsp-ALN, as well as their 1H NMR spectra, are shown in Fig. 2B–D. Based on the previous literature, peak d at 7.3 ppm and peak e at 5.1 ppm in Fig. 2B were attributed to hydrogen protons of the benzene ring and methylene on the benzyl groups. The disappearance of both the peaks means successful deprotection. In addition, the correspondence can be seen that the peaks a and c represent the hydrogen protons of methylene in the PEG segments and methine in the main chains of PBLA (PAsp and PAsp-ALN) segments, respectively. The degrees of polymerization (DPs) of Asp units in PEG-PAsp were calculated to be 32 by comparing the peak intensities between peak a and peak c. Correspondingly, the FTIR spectrum of PEG-PAsp is recorded in Fig. 2E. The appearance of two characteristic peaks at 1637 and 1555 cm1 which can be attributed to the vibration bands of amide I and II demonstrated the formation of amide bonds in synthetic polymers. Meanwhile, the peak at 1100 cm1 corresponded to the stretching vibration peak of C–O bonds in the PEG segments. Besides, the characteristic peak at 2884 cm -1 represents the C–H stretching vibration existing in both the segments. All the results further confirmed the successful synthesis of PEG-PAsp. PEG-PAsp-ALN by the partial modification of PAsp segments with ALN monomers. The peaks f, g, and h appeared in Fig. 2D after the postfunctionalization of ALN, which were attributed to the different methylene groups of ALN monomers. The emerging peaks g and h demonstrated the successful grafting of ALN on the polymer chains. Accordingly, the degree of modification can be calculated to be 34.3% via the comparison of the peak intensity between peak b Journal of Materials Chemistry B and peak g +f. The successful postfunctionalization of ALN can also be verified by the FTIR spectra of PEG-PAsp, PEG-PAsp-ALN and neat ALN (Fig. 2E). The peak at about 1173 cm1 was assigned to the vibration of PQO bonds, which appeared in the FTIR spectrum of PAsp modified with ALN rather than that of PEG-PAsp. Adsorption evaluation of PEG-PAsp-ALN According to the previous data reported, the maximum absorbance concentration to HA of PEG-PAsp powders was 1.4 mg mL -1, which would be the chosen concentration for further research. To verify the adsorption of PEG-PAsp-ALN on tooth enamel surfaces, the surface structures of acid-etched enamels, unwashed enamels coated with PEG-PAsp and PEGPAsp-ALN and washed enamels coated with PEG-PAsp and PEGPAsp-ALN were analysed by FTIR. The resulting FTIR spectra are showninFig. 3AandB. TheFTIRspectraof enamel surfaces coated with PEG-PAsp and PEG-PAsp-ALN showed that many characteristic peaks of polymers appeared in the range of 15001700 cm -1 including vibration peaks of amides I and II at 1637 and 1555 cm -1. After rinsing, the intensity of these peaks decreased but did not disappear completely, indicating that PEGPAsp and PEG-PAsp-ALN can be absorbed on the enamel surfaces. Benefitting from the existence of PO4 3- ions brought about by ALN postfunctionalization, higher absorption should be achieved owing to stronger binding of PO4 3- with the enamel surface, which can be verified by the quantification analysis of the absorbed polymer on substrates. As peptide bonds in the PAsp segment can be detected using a BCA Protein Assay Kit, a quantitative measurement was carried out based on it. As shown in Fig. 3C, under the same processing conditions, the concentration of eluted PEG-PAsp-ALN from substrates was higher than that of PEG-PAsp, indicating the higher absorption of PEG-PAsp-ALN owing to the existence of PO4 3- ions. Furthermore, FITC-labelled PEG-PAsp and PEG-PAsp-ALN were used to investigate the absorption capability of polymers on enamel surfaces. Without FITC labeling, two enamel samples coated with PEG-PAsp and PEG-PAsp-ALN did not show fluorescence. After FITC labeling, the fluorescence intensity of PEG-PAsp-ALN was significantly stronger than that of PEG-PAsp (Fig. 3E), indicating that PEG-PAsp-ALN has stronger adsorption capacity compared with PEG-PAsp. Adsorption stability of PEG-PAsp-ALN The dynamic stability of the polymeric coating was measured under flowing artificial saliva. There is a significant difference Paper in the fluorescence intensity between PEG-PAsp and PEG-PAspALN at day 0, and the significant difference still existed at days 1 and 3, showing little loss of PEG-PAsp-ALN on the enamel surfaces within 3 days (Fig. 3D and E). It can be seen that the interaction between PO4 3- and Ca2+ ions guaranteed the stability of PEG-PAsp-ALN coatings on the substrates.
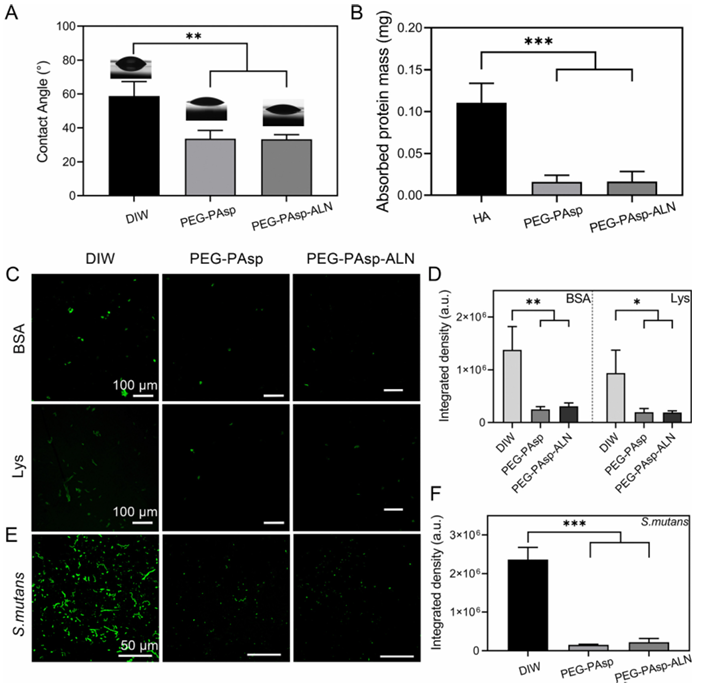
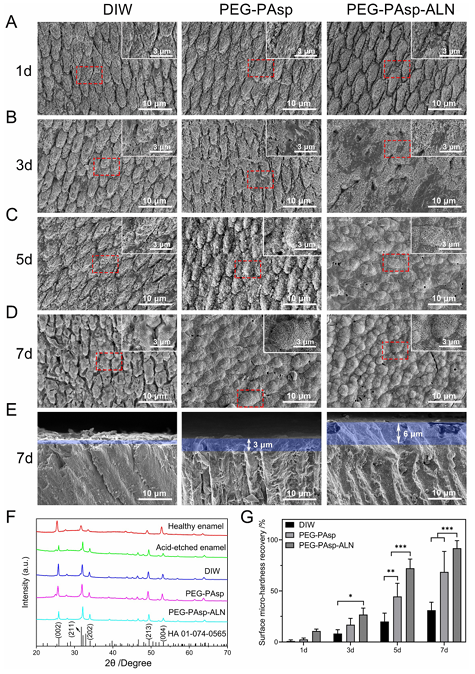
Fig. 4 Theanti-fouling property of PEG-PAsp-ALN. (A) Water contact angles, (B) the mass of saliva protein absorbed on HA substrates. (C) Fluorescence images of BSA, and Lys proteins absorbed on the enamels coated with DIW, PEG-PAsp and PEG-PAsp-ALN and (D) relative calculated fluorescence density. (E) Fluorescence images of S. mutans absorbed on the enamels coated with DIW, PEG-PAsp and PEG-PAsp-ALN and F) relative calculated fluorescence density. *p o 0.01, **p o 0.005, and ***p o 0.001. Anti-fouling effect of PEG-PAsp-ALN The wettability was characterized according to the static water contact angle test by sessile drops of DIW to the surface of enamels coated with different materials (Fig. 4A). It is known that the existence of PEG segments endows PEG-PAsp and PEGPAsp-ALN with stronger hydrophilicity, which is the main source of antifouling property. Correspondingly, the contact angles of enamels coated with PEG-PAsp and PEG-PAsp-ALN were 33.59 4.88° and 33.22 ± 2.71°, respectively, lower than 58.76 ± 8.55° of the DIWgroup. Previous literature reports have stated that the excellent hydrophilicity of PEG segments endows with a larger intermolecular force between materials and water molecules, which would lead to the formation of a hydration layer on the material surfaces to show superb barrier performance. Enhanced hydrophilicity can serve as the basis of materials resisting several bio-pollutants. To demonstrate the anti-fouling effect of materials, protein and salivary pellicle absorption as well as bacterial anti-adhesive tests were carried out. For protein absorption, positively charged BSA and negatively charged Lys conjugated with FITC for visualization using LSCM were chosen as models for the assays, and the corresponding integrated fluorescence density was calculated to demonstrate the universal anti-protein absorption capability of materials (Fig. 4C and D). After 24 h of incubation, both positively charged FITClabelled BSA and negatively charged FITC-labelled Lys were densely distributed on the surfaces of enamels treated with DIW, and a stronger fluorescence signal was observed, which confirmed massive amounts of BSA and Lys absorption. As for the groups treated with PEG-PAsp and PEG-PAsp-ALN, enhanced hydrophilicity inhibited protein absorption. The amounts of protein and the correspondingly calculated fluorescence density decreased significantly, showing less protein existence on the enamel surfaces. Two proteins with opposite charge adsorption assays proved the non-specific anti-adhesioneffectofpolymeric coatings, and its universal anti-fouling ability. In order to explore the potential of our polymeric coatings for oral cavity application, the adsorption of salivary pellicle onthe enamel surfaces was quantitatively determined with BCA (Fig. 4B). The saliva protein content absorbed on HA slices without polymeric coatings was the highest in all three groups. HA slices with polymer coatings significantly reduced protein absorption, indicating good resistance to saliva protein adhesion. Since the saliva acquired pellicle is a breeding ground for bacterial reproduction during biofilm formation, the resistance adhesion properties of our polymeric coatings to saliva are beneficial for inhibiting subsequent bacterial colonization. Since S. mutans is recognized widely as the main cariogenic bacteria of dental caries, inhibiting the adhesion of S. mutans plays a vital role in preventing the occurrence of dental caries. Themeasurement oftheantibacterial activity on the HA surface is conducted to determine whether PEG-PAsp-ALN can inhibit S. mutans adhesion. The LSCMimagesandfluorescence density of bacteria on the coated HA slices were recorded to explain the anti-adhesive effect (Fig. 4E and F). The images showed that S. mutans can attach to the substrates and reproduce on them. Bacteria on HA slices treated with DIW have connected in a long-chain form and exhibited dense distribution resulting from bacterial reproduction on substrates. In the two groups treated with polymers, the improvement of substrate hydrophilicity made the initial adhesion of bacteria difficult. The combined effect of the decrease of initial adhesion and hydrophilicity also weakened the subsequent colonization process. Consequently, their LSCM images showed that bacterial distribution on substrates was sparse and scattered due to the resistance of PEG segments. Meanwhile, quantitative characterization of fluorescence images was conducted and revealed that the bacterial fluorescence intensity of groups treated with polymers was much lower than that of the DIW group, proving that the coatings had excellent resistance to the adhesion of oral cariogenic bacteria. Remineralization capability of PEG-PAsp-ALN To evaluate the capability of PEG-PAsp-ALN to boost remineralization of acid-etched enamels, three groups of enamel samples coated with DIW, PEG-PAsp and PEG-PAsp-ALN, respectively, were immersed in the mineralizing solution. 1.5 SBF solution was chosen as the mineralizing solution because a more distinct comparison can appear within 7 days as post-coated enamels were dipped into SBF solution than simulant saliva due to a more sufficient Ca and P resource. The morphology of new mineral layers at 1, 3, 5 and 7 days was observed by SEM. The acid-etching process changed the regular structure of the enamel surfaces and loosened the HA structure, providing the enamel surfaces with countless tiny pores (Fig. S2, ESI?), which will be beneficial for the deposition of exogenous Ca and P.58 As can be seen from Fig.5AD, the newly formed minerals of all groups treated with DIW, PEG-PAsp and PEG-PAsp-ALN increased with the immersing time going by, indicating that three groups of enamel samples had certain remineralization ability. Despite the low degree of mineralization, pure tooth enamels without polymers can also mineralize within 7 d. The enamel surfaces coated with PEG-PAsp can increase the chelation with Ca2+ ions because of the introduction of carboxyl groups. The COOH–Ca2+ complexes can serve as nucleation sites to initiate the subsequent mineralization process. So the group showed an acceleration than the DIW group. The pores of acid-etched tooth slices can be covered with newly formed minerals initially on day 5 and complete crystals appeared on day 7. Furthermore, the stronger interaction of PO4 3- with Ca2+ ions led to more absorption of PEG-PAsp-ALN, which brought more carboxyl groups and PO4 3- ions more conducive to attracting Ca2+ ion deposition to provide nucleation sites. The mineralization rate of the PEG-PAsp-ALN group was further accelerated. Initial crystallization can be seen on the third day and enamel slices can be covered thoroughly on the fifth day. In addition, the SEM images of the cross sections of the three groups of enamel samples mineralizing for 7 days are also shown in Fig. 5E. The newly formed mineral crystals could be closely bonded to the mineral layer of the enamel itself. Faster mineralization rates lead to thicker mineral formation over the same time. The SEM pictures of three enamel cross-sections on day 7 also demonstrated that the minerals induced by PEG-PAsp-ALN were the thickest compared to the other two groups. To verify the crystal structure of the newly formed mineral, the surfaces of healthy enamel, acid-etched enamel, and enamels mineralized for 7 days were scanned with XRD and the result is shown in Fig. 5F. According to the standard card 01074-0565, the diffraction peaks at 25.88°, 31.77°, 34.06°, 49.49° and 53.22° correspond to the (002), (211), (202), (213) and (004) crystal planes of the HA crystal, respectively. While these diffraction peaks not only appeared in the pattern of healthy enamel, but also existed in that of the three groups of mineralized enamel samples, indicating that the newly formed mineral was the HA crystal rather than amorphous calcium phosphate. In order to investigate the effect of the newly formed HA mineral on the restoration of enamel hardness, a Vickers hardness tester was used to measure the surface microhardness of enamel with different treatments and the degree of recovery was calculated accordingly. The high hardness of healthy tooth enamel mainly depends on an HA inorganic component and their regular arrangement. Consequently, after the crystal structure was destroyed by acid etching, the hardness of enamels will be greatly reduced. The newly generated HA crystals in the mineralization process can make up for the loss of hardness to a certain extent. As shown in Fig. 5G, the hardness of the three groups recovered gradually with the extension of the mineralization time. More importantly, due to the stronger mineralization effect brought by PEG-PAsp-ALN, the structure of the new HA is the most regular and their amount is also the largest. Thus, the strongest hardness repair behaviours can be shown.
Fig. 5 The remineralization capability of materials. SEM images showing the morphology of enamel surfaces coated with DIW, PEG-PAsp and PEGPAsp-ALN after being immersed in SBF for (A) 1 day, (B) 3 days, (C) 5 days, and (D) 7 days, as well as (E) the cross sections for 7 days. (F) XRD spectra of healthy enamel, acid-etched enamel and enamels coated with DIW, PEG-PAsp, and PEG-PAsp-ALN after mineralization for 7 days, respectively. (G) The surface micro-hardness recovery rate of acid-etched enamels coated with DIW, PEG-PAsp, and PEG-PAsp-ALN after mineralization for 1, 3, 5 and 7 days. *p o 0.01, **p o 0.005, and ***p o 0.001. Cytocompatibility of PEG-PAsp-ALN The cytocompatibility of the polymer coatings in the oral cavity was verified by selecting HOK cells as the models. The CCK-8 and fluorescence staining results are shown in Fig. 6. Fig. 6A shows the cytotoxicity of PEG-PAsp and PEG-PAsp-ALN. The cell viability of PEG-PAsp and PEG-PAsp-ALN groups showed no significant difference compared with that of the control group. The cytocompatibility of polymer materials was also determined by HOK live/dead (Fig. 6B) and morphological staining results (Fig. 6C). For both PEG-PAsp and PEG-PAsp-ALN groups, the fluorescence images of live cells (green) in live/dead staining are similar to the control group, and little dead cells (red) were detected. Meanwhile, the morphological staining revealed that there were no differences in the morphology between cells incubated with or without polymers. Conclusions In summary, dual-functional coatings with antifouling and remineralizing properties were successfully constructed for the prevention and treatment of dental caries with a synthetic multifunctional polymer, PEG-PAsp-ALN. A hydrophilic polymer PEG and mineral-inducing moiety PAsp were combined firstly to get PEG-PAsp, and ALN with strong absorption on enamels were readily conjugated with PEG-PAsp to obtain PEGPAsp-ALN. When PEG-PAsp-ALN is applied to enamels, PAsp and ALN synergistically bind with enamels and promote remineralization for their abundant carboxyl groups and PO4 3- in the oral environment. Meanwhile, the hydrophilic interactions between PEG chains and water provide stable hydrogen-bonded hydrated layers on the enamel surface, which resist the attachment of proteins and bacteria. In addition, PEG-PAsp-ALN shows good cytocompatibility. The present work provided a promising strategy for the design and development of a novel multifunctional enamel coating for preventing dental caries and promoting in situ self-healing of tooth decays effectively. Author contributions Wenlin Chu: methodology, formal analysis, investigation, and writing– original draft. Haiqin Tang: formal analysis and investigation. Zhiyun Dong: investigation and writing– original Draft. Ailin Hou: formal analysis and investigation. Rongmin Qiu: formal analysis and writing– review & editing. Jiaojiao Yang: methodology and writing– original draft. Xinyuan Xu: supervision and writing– review & editing. Jun Luo: conceptualization, supervision, and writing– review & editing. Libang He: conceptualization, methodology, supervision, and writingreview & editing. Jianshu Lee: conceptualization, supervision, and writing– review & editing. Conflicts of interest There are no conflicts to declare. Acknowledgements The authors are thankful to the financial support provided by the National Natural Science Foundation of China (grant no. 51925304, 52273135, and 52203180), the Sichuan Science and Technology Program (grant no. 2023NSFSC0999), the Sichuan Regional Innovation Cooperation Project (grant no. 2023YFQ0014), and the Key R & D projects of Guangxi Science and Technology Department ((grant no. 2021AB11097). Source: rsc.li/materials-b |